Abstract
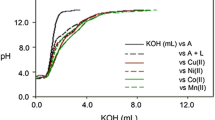
Thermodynamic parameters for dissociation of 3-nitro-1,5-diphenylformazan and its complexation by some divalent metal ions were determined in a 50%(v/v) dioxane–water mixture at constant ionic strength (0.1 M KCl) using an automatic potentiometric technique. The changes in the standard Gibbs energy ΔGo and enthalpy ΔHo accompanying the complexation were found to decrease with increasing metal ionic radius and to increase with the electronegativity, the ionization enthalpy, and the enthalpy of hydration. The order of −ΔGo and −ΔHo values were found to be Mn2+ < Fe2+ < Co2+ < Ni2+ < Cu2+ > Zn2+, in accordance with the Irving–Williams order. The complexes were stabilized by both enthalpy and entropy changes and the results suggest that the complexation is an enthalpy-driven process. The transition-series contraction energy Er(Mn–Zn) and the ligand field stabilization energy δ H were calculated from the enthalpy changes.
Similar content being viewed by others
REFERENCES
B. N. Barsoum, S. K. Khella, H. M. Elwaby, A. A. Abbas, and Y. A. Ibrahim, Talanta 47, 1215 (1998).
P. M. Debnan and G. Shearer, Anal. Biochem. 250, 253 (1997).
Y. B. Liu and D. Schubert, D., J. Neurochem. 69, 2285 (1997).
Y. Kayamori, Y. Katayama, T. Matsuyama, and T. Urata, Clin. Biochem. 30, 595 (1997).
Z. Yuzhen and L. Dongzhi, Dyes and Pigments 29, 57 (1995).
V. S. Misra, S. Dhar, and B. L. Chowdhary, Pharmazie 33, 790 (1978).
D. D. Mukerjee, S. K. Shukla, and B. L. Chowdhary, Arch. Pharmacol. 314, 991 (1981).
D. D. Mukerjee and S. K. Shukla, Bokin Babai 9, 521 (1981); Chem. Abstr. 96, 65524 (1982).
D. Palut and Z. Eckstein, Bull. Acad. Polon. Sci. Ser. Sci. Chim. 12, 41 (1964).
Z. R. Bulkin, L. S. Pupko, E. F. Gavlbenko, M. N. Danchenko, and P. S. Pelkis, Fiziol. Akt. Veshchestva 2, 91 (1971); Chem. Abstr. 73, 12657 (1970).
H. M. N. H. Irving, Dithizone, (Chemical Society, London, 1977).
G. Iwantscheff, Das Dithizon und Seine Anwendung in der Mikro-und Spurenanalyse, 2nd edn., (Verlag Chemie, Weinheim, 1972).
A. M. Kiwan and A. Y. Kassim, Anal. Chim Acta 88, 177 (1977).
A. M. Kiwan and G. A. Wanas, Anal. Chim. Acta 144, 165 (1982).
A. M. Kiwan, F. M. Hassan, W. Hamdan, A. A. Bahajaj, and M. Y. Khaled, J. Coord. Chem. 22, 59 (1990).
I. Shehatta, M. N. H. Moussa, and M. A. Hafez, Thermochim. Acta 219, 121 (1993).
A. A. El-Bindary, I. Shehatta, and E. M. Mabrouk, Monatsh. Chem. 125, 373 (1994).
I. Shehatta, Z. Phys. Chem. 187, 109 (1994).
I. Shehatta and A. A. El-Bindary, Thermochim. Acta 237, 357 (1994).
A. A. El-Bindary and I. Shehatta, Monatsh. Chem. 125, 841 (1994).
I. Shehatta, A. M. Hassanein, and E. M. Mabrouk, Monatsh. Chem. 126, 851 (1995).
I. Shehatta, Functional React. Polym. 28, 183 (1996).
L. G. Van Uitert and C. G. Hass, J. Amer. Chem. Soc. 75, 451 (1953).
H. Irving and H. S. Rossotti, J. Chem. Soc., p. 3397 (1953).
F. J. C. Rossotti and H. S. Rossotti, Acta Chem. Scand. 9, 1166 (1955).
G. A. Sereda, R. I. Ogloblina, V. N. Podchanova, and S. P. Bednyaagina, Zhur. Anal. Khim. 23, 1220 (1968).
Y. M. Issa, N. T. Abdel-Ghani, and O. E. Sherif, Bull. Soc. Chim. Fr. 128, 627 (1991).
N. T. Abdel-Ghani and O. E. Sherif, Thermochim. Acta 156, 69 (1989).
J. B. Gill, H. M. N. H. Irving, and A. Prescott, J. Chem. Soc. Perkin Trans. 2, 1683 (1977).
H. Irving and J. P. Williams, J. Chem. Soc., p. 3192 (1953).
M. T. Beck and I. Nagybal, Chemistry of Complex Equilibria (Wiley, New York, 1990).
S. Murakami and Y. Yoshino, J. Inorg. Nucl. Chem. 43, 2070 (1981).
P. George and D. S. McClure, Progress in Inorganic Chemistry, F. A. Cotton, ed., Wiley (Interscience), New York, (1959).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shehatta, I., Kiwan, A.M. Thermodynamics of Dissociation and Metal Complexation of 3-Nitro-1,5-Diphenylformazan. Journal of Solution Chemistry 30, 389–400 (2001). https://doi.org/10.1023/A:1010383324660
Issue Date:
DOI: https://doi.org/10.1023/A:1010383324660