Abstract
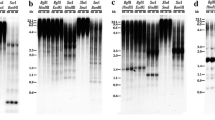
RAPD markers generated by mixtures of two different primers were developed for octoploid × Tritordeum (amphiploid Hordeum chilense × Triticum aestivum) and its parents. Addition lines were used to identify 21 specific RAPD markers for the H. chilense chromosomes detectable in a wheat background. Ten RAPD bands were selected and eight of them were converted into dominant SCAR markers by direct sequencing of the RAPD products, avoiding the costly and time-consuming cloning step. The methodology overcomes some of the pitfalls associated with the election of the right clones when developing SCARs from RAPD markers. The SCARs generated have maintained both the chromosome specificity and the possibility of detection in a wheat background. This strategy provides a rapid method for the characterization of RAPD markers and for the development of PCR-based markers for both the characterization of the introgression of H. chilense in bread and durum wheat, as well as the efficient and reliable screening of tritordeum lines.
Similar content being viewed by others
References
Adam-Blondon AF, Sévignac M, Bannerot H, Dron M: SCAR, RAPD and RFLP markers linked to a dominant gene (Are) conferring resistance to anthracnose in common bean. Theor Appl Genet 88: 865–870 (1994).
Bodenes C, Joandet S, Laigret F, Kremer A: Detection of genomic regions differentiating 2 closely-related oak species Quercus petraea (Matt) Liebl and Quercus robur L. Heredity 78: 433–444 (1997).
Cheng FS, Weeden NF, Brown SK: Identification of codominant RAPD markers linked to fruit skin color in apple. Theor Appl Genet 93: 222–227 (1996).
Deng Z, Huang S, Xiao S, Gmitter F G: Development and characterization of SCAR markers linked to the citrus tristeza virus resistance gene from Poncirus trifoliata. Genome 40: 697–704 (1997).
Devos KM, Gale MD: The use of random amplified polymorphic DNA markers in wheat. Theor Appl Genet 84: 567–572 (1992).
Gu WK, Weeden NF, Yu J, Wallace DH: Large-scale, costeffective screening of PCR products in marker-assisted selection applications. Theor Appl Genet 91: 465–470 (1995).
Hernández P, Rubio MJ, Martin A: Development of RAPD markers in tritordeum and addition lines of Hordeum chilense in Triticum aestivum. Plant Breed 115: 52–56 (1996).
Kleppe K, Ohstuka E, Kleppe R, Molineux L, Khorana HG: Studies in polynucleotides XCVI. Repair replication of short synthetic DNA's as catalyzed by DNA polymerases. J Mol Biol 56: 341–361 (1971).
Lario A, González A, Dorado G: Automated laser-induced fluorescence DNA sequencing: Equalizing signal-to-noise ratios significantly enhances overall performance. Anal Biochem 247: 30–33 (1997).
Martín A: Tritordeum: the first ten years. Rachis 7: 12–15 (1988).
Martín A, Martín LM, Cabrera A, Ramírez MC, Giménez MJ, Rubiales D, Hernández P, Ballesteros J: The potential of Hordeum chinense in breeding Triticeae species. In: International Triticeae Symposium (Aleppo, Syria), pp. 377–386, (1997).
Martín A, Martínez-Araque C, Rubiales D, Ballesteros J (1996): Tritordeum: triticales's new brother cereal. In: Guedes-Pinto H, Darvey N, Carnide VP (eds) Triticale: Today and Tomorrow, pp. 57–72. Kluwer Academic Publishers, Dordrecht, Netherlands (1996).
Miller TE, Reader SM, Chapman V: The addition of Hordeum chilense chromosomes to wheat. In: Broertjes C (ed), Proceedings International Symposium Eucarpia on Induced Variability in Plant Breeding, pp. 79–81. Pudoc, Wageningen, Netherlands (1982).
Mullis KB, Faloona FA: Specific synthesis of DNA in vitro via a polymerase-catalyzed chain reaction. Meth Enzymol 155: 335–350 (1987).
Panet A, Khorana HG: Studies on polynucleotides. The linkage of deoxyribopolynucleotide templates to cellulose and its use in their replication. J Biol Chem 249: 5213–5221 (1974).
Paran I, Michelmore R: Development of reliable PCR-based markers linked to downy mildew resistance genes in lettuce. Theor Appl Genet 85: 985–993 (1993).
Saiki RK, Bugawan TL, Horn GT, Mullis KB, Erlich HA: Analysis of enzymatically amplified β-globine and HLA-DQα DNA with allele- specific oligonucleotide probes. Nature 324: 163–166 (1986).
Saiki RK, Scharf SJ, Faloona F, Mullis KB, Horn GT, Erlich HA, Arnheim N: Enzymatic amplification of β-globin genomic sequences and restriction site analysis for diagnostic of sickle cell anemia. Science 230: 1350–1354 (1985).
Sanger F, Nicklen S, Coulson AR: DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74: 5463–5467 (1977).
Torres AM, Weeden NF, Martín A: Linkage among isozyme, RFLP and RAPD markers in Vicia faba. Theor Appl Genet 85: 937–945 (1993).
Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Fritjers A, Pot J, Peleman J, Kuiper M, Zabeau M: AFLP: a new concept for DNA fingerprinting. Nucl Acids Res 23: 4407–4414 (1995).
Welsh J, McClelland M: Fingerprinting genomes using PCR with arbitrary primers. Nucleic Acids Res 18: 7213–7218 (1990).
Williams JGK, Rubelik AR, Livak K J, Rafalski A, Tingey SV: DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucl Acids Res 18: 6531–6535 (1990).
Xu H, Wilson DJ, Arulsekar S, Bakalinsky AT: Sequencespecific polymerase chain-reaction markers derived from randomly amplified polymorphic DNA markers for fingerprinting grape (Vitis) rootstocks. J Am Soc Hort Sci 120: 714–720 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hernández, P., Martín, A. & Dorado, G. Development of SCARs by direct sequencing of RAPD products: a practical tool for the introgression and marker-assisted selection of wheat. Molecular Breeding 5, 245–253 (1999). https://doi.org/10.1023/A:1009637928471
Issue Date:
DOI: https://doi.org/10.1023/A:1009637928471