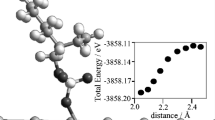
Abstract
Experimental cathodic polarization curves, obtained on a mercury electrode in 0.33 mM K3[Fe(CN)6] + 0–1.5 mM KCl solutions, are analyzed quantitatively. On the basis of quantum-chemical calculations of the geometry of species [Fe(CN)6]3– and [Fe(CN)6]4– and charge distributions in them, it is shown that the species interaction with the EDL field is equivalent to a repulsive interaction between effective point charges localized near anion centers in the diffuse layer. The effective Born radius of ferricyanide anion and the solvent reorganization energy are calculated, and inner-sphere constituents of the energy are estimated. These parameters are used for computing theoretical dependences of the transfer coefficient α on the overvoltage via the equation of the quantum-mechanical theory of elementary act. The value of α, determined from corrected Tafel plots, is shown to substantially depend on the assumptions adopted when analyzing the system in the framework of the classical slow-discharge theory; it is close to the theoretical value only if the participation of anion–cation associates formed in the bulk solution is taken into account. Such an approach explains the weak temperature dependence of the process rate. The experimental facts do not contradict theoretical prediction that the reaction occurs in the vicinity of the activationless region.
Similar content being viewed by others
REFERENCES
Khoshtaria, D.E., Dolidze, T.D., Krulic, D., et al., J. Phys. Chem., 1998, vol. 102,p. 7800.
Marecek, V., Samec, Z., and Weber, J., J. Electroanal. Chem., 1978, vol. 94,p. 169.
Kitamura, F., Nanbu, N., Ohsaka, T., and Tokuda, K., J. Electroanal. Chem., 1998, vol. 456,p. 113.
Frumkin, A.N. and Florianovich, G.M., Dokl. Akad. Nauk SSSR, 1951, vol. 80,p. 907.
Frumkin, A.N., Petrii, O.A., and Nikolaeva-Fedorovich, N.V., Dokl. Akad. Nauk SSSR, 1959, vol. 128,p. 1006.
Petrii, O.A. and Nikolaeva-Fedorovich, N.V., Zh. Fiz. Khim., 1961, vol. 35,p. 1999.
Levich, V.G., Dokl. Akad. Nauk SSSR, 1949, vol. 67,p. 309.
Vorotyntsev, M.A., Itogi Nauki Tekh., Ser.: Elektrokhimiya, 1979, vol. 14,p. 62.
Tsirlina, G.A., Kuznetsov, A.M., Petrii, O.A., and Kharkats, Yu.I., Elektrokhimiya, 1999, vol. 35,p. 938.
Tobe, M., Inorganic Reaction Mechanisms, London: Thomas Nelson, 1972. Translated under the title Mekhanizmy neorganicheskikh reaktsii,Moscow: Mir, 1975.
Nikolaeva-Fedorovich, N.V., Petrii, O.A., Damaskin, B.B., and Furazhkova, G.A., Vestn. Mosk. Univ., 1962, no. 3,p. 40.
Clack, D.W. and Monshi, M., Mol. Phys., 1976, vol. 31,p. 1607.
Sano, M., Adachi, H., and Yamatera, H., Bull. Chem. Soc. Jpn., 1981, vol. 54,p. 2898.
Becke, A.D., J. Chem. Phys., 1992, vol. 98,p. 1372.
Becke, A.D., J. Chem. Phys., 1992, vol. 98,p. 5648.
Foresman, J.B. and Frisch, A., Exploring Chemistry with Electronic Structure Methods, Pittsburgh: Gaussian, 1996.
Hay, P.J. and Wadt, W.R., J. Chem. Phys. 1985, vol. 82,p. 270.
Dunning, T.H. and Hay, P.J., in Modern Theoretical Chemistry, vol. 3: Methods of Electronic Structure Theory, Schaefer, H.F., III, Ed., New York: Plenum, 1977,p. 1.
Hehre, W.H., Ditchfield, R., and Pople, J.A., J. Chem. Phys., 1971, vol. 56,p. 2257.
Lever, A., Inorganic Electronic Spectroscopy, Amsterdam: Elsevier, 1984. Translated under the title Elektronnaya spektroskopiya neorganicheskikh soedinenii, Moscow: Mir, 1987, part 2.
German, E.D. and Kuznetsov, A.M., Itogi Nauki Tekh., Ser.: Kinet. Katal., 1982, vol. 10,p. 115.
Tomassi, J. and Persico, M., Chem. Rev., 1994, vol. 94,p. 202.
Foster, J.P. and Weinhold, F., J. Am. Chem. Soc., 1980, vol. 102,p. 7211.
Reed, A.E., Curtiss, L.A., and Weinhold, F., Chem. Rev. 1988, vol. 88,p. 899.
Nazmutdinov, R.R., Tsirlina, G.A., Kharkats, Yu.I., and Petrii, O.A., J. Phys. Chem. B, 1998, vol. 102,p. 677.
Gorel'sky, S.I., Kotov, V.Yu., and Lever, A.B.P., Inorg. Chem., 1998, vol. 37,p. 4584.
Delahay, P., Double Layer and Electrode Kinetics, New York: Interscience, 1965.
Damaskin, B.B. and Polyanovskaya, N.S., Elektrokhimiya, 1984, vol. 20,p. 90.
Kravtsov, V.I., Ravnovesie i kinetika elektrodnykh reaktsii kompleksov metallov (The Equilibrium and Kinetics of Electrode Reactions Involving Metal Complexes),Leningrad: Khimiya, 1985,p. 76.
Fuoss, R.M., J. Am. Chem. Soc., 1958, vol. 80,p. 5059.
Dogonadze, R.R. and Kuznetsov, A.M., Itogi Nauki Tekh., Ser.: Kinet. Katal., 1978, vol. 5,p. 5.
Tsirlina, G.A., Petrii, O.A., Kharkats, Yu.I., and Kuznetsov, A.M., Elektrokhimiya, 1999, vol. 35,p. 1377.
Damaskin, B.B., Safonov, V.A., and Fedorovich, N.V., J. Electroanal. Chem., 1993, vol. 349,p. 1.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tsirlina, G.A., Titova, N.V., Nazmutdinov, R.R. et al. Electroreduction of [Fe(CN)6]3– on a Mercury Electrode: Substantiating Activationless Character of the Process at High Overvoltages. Russian Journal of Electrochemistry 37, 15–25 (2001). https://doi.org/10.1023/A:1009067309773
Issue Date:
DOI: https://doi.org/10.1023/A:1009067309773