Abstract
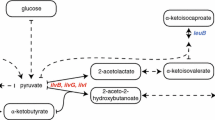
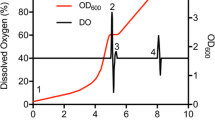
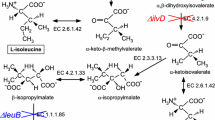
A selective protonation strategy is described that uses [3-2H] 13C α-ketoisovalerate to introduce (1H-δ methyl)-leucine and (1H-γ methyl)-valine into 15N-, 13C-, 2H-labeled proteins. A minimum level of 90% incorporation of label into both leucine and valine methyl groups is obtained by inclusion of ≈100 mg/L α-ketoisovalerate in the bacterial growth medium. Addition of [3,3-2H2] α-ketobutyrate to the expression media (D2O solvent) results in the production of proteins with (1H-δ1 methyl)-isoleucine (>90% incorporation). 1H-13C HSQC correlation spectroscopy establishes that CH2D and CHD2 isotopomers are not produced with this method. This approach offers enhanced labeling of Leu methyl groups over previous methods that utilize Val as the labeling agent and is more cost effective.
Similar content being viewed by others
References
Bax, A. (1994) Curr. Opin. Struct. Biol., 4, 738–744.
Constantine, K.L., Mueller, L., Goldfarb, V., Wittekind, M., Metzler, W.J., Yanchunas, J., Jr., Robertson, J.G., Malley, M.F., Friedrichs, M.S. and Farmer, B.T.I. (1997) J. Mol. Biol., 267, 1223–1246.
Delaglio, F., Grzesiek, S., Vuister, G.W., Zhu, G., Pfeifer, J. and Bax, A. (1995) J. Biomol. NMR, 6, 277–293.
Eisenstein, E. (1991) J. Biol. Chem., 266, 5801–5807.
Gardner, K.H. and Kay, L.E. (1997) J. Am. Chem. Soc., 119, 7599–7600.
Gardner, K.H. and Kay, L.E. (1998) Annu. Rev. Biophys. Biomol. Struct., 27, 357–406.
Gardner, K.H., Zhang, X., Gehring, K. and Kay, L.E. (1998) J. Am. Chem. Soc., 120, 11738–11748.
Gottschalk, G. (1986) In: Bacterial Metabolism, 2nd ed., Springer-Verlag, New York, NY, p. 359.
Grzesiek, S., Anglister, J., Ren, H. and Bax, A. (1993) J. Am. Chem. Soc., 115, 4369–4370.
Grzesiek, S., Wingfield, P., Stahl, S., Kaufman, J. and Bax, A. (1995) J. Am. Chem. Soc., 117, 9594–9595.
Leimer, K.R., Rice, R.H. and Gehrke, C.W. (1977) J. Chromatogr., 141, 121–144.
Massey, L.K., Sokatch, J.R. and Conrad, R.S. (1976) Bacteriol. Rev., 40, 42–54.
Metzler, W.J., Wittekind, M., Goldfarb, V., Mueller, L. and Farmer, B.T. (1996) J. Am. Chem. Soc., 118, 6800–6801.
Muhandiram, D.R., Xu, G.Y. and Kay, L.E. (1993) J. Biomol. NMR, 3, 463–470.
Rosen, M.K., Gardner, K.H., Willis, R.C., Parris, W.E., Pawson, T. and Kay, L.E. (1996) J. Mol. Biol., 263, 627–636.
Santoro, J. and King, G.C. (1992) J. Magn. Reson., 97, 202–207.
Shan, X., Gardner, K.H., Muhandiram, D.R., Rao, N.S., Arrowsmith, C.H. and Kay, L.E. (1996) J. Am. Chem. Soc., 118, 6570–6579.
Smith, B.O., Ito, Y., Raine, A., Teichmann, S., Ben-Tovim, L., Nietlispach, D., Broadhurst, R.W., Terada, T., Kelly, M., Oschkinat, K., Shibata, T., Yokoyama, S. and Laue, E.D. (1996) J. Biomol. NMR, 8, 360–368.
Venters, R.A., Metzler, W.J., Spicer, L.D., Mueller, L. and Farmer, B.T. (1995) J. Am. Chem. Soc., 117, 9592–9593.
Vuister, G.W. and Bax, A. (1992) J. Magn. Reson., 98, 428–435.
Yamazaki, T., Lee, W., Arrowsmith, C.H., Muhandiram, D.R. and Kay, L.E. (1994) J. Am. Chem. Soc., 116, 11655–11666.
Zwahlen, C., Gardner, K.H., Sarma, S.P., Horita, D.A., Byrd, R.A. and Kay, L.E. (1998a) J. Am. Chem. Soc., 120, 7617–7625.
Zwahlen, C., Vincent, S.J.F., Gardner, K.H. and Kay, L.E. (1998b) J. Am. Chem. Soc., 120, 4825–4831.
Author information
Authors and Affiliations
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Goto, N.K., Gardner, K.H., Mueller, G.A. et al. A robust and cost-effective method for the production of Val, Leu, Ile (δ1) methyl-protonated 15N-, 13C-, 2H-labeled proteins. J Biomol NMR 13, 369–374 (1999). https://doi.org/10.1023/A:1008393201236
Issue Date:
DOI: https://doi.org/10.1023/A:1008393201236