Abstract
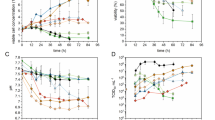
Disabled Infectious Single Cycle (DISC) HSV-2 has been cultured in the complimentary cell line CR2 to provide high titre bulk material suitable for the purification of the virus as a live viral vaccine. CR2 cells are cultured on the microcarrier Cytodex-1 at 5 g l-1 in small scale (1 l) and larger scale (15 l) reactors. The cells are infected at an MOI of 0.01 pfu cell-1 and the culture harvested 60–72 h later. The infected cells are removed from the microcarriers by the addition of a hypotonic saline and the virus released by low-pressure disruption techniques. Virus titres achieved are compared to the standard roller bottle process. The resulting material is the starting point for the purification of the DISC-HSV virus.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baijot B, Duchene M and Stephenne J (1987) Production of Aujeszky vaccine by the microcarrier technology (from the ampoule to the 500 l fermentor). Dev Biol Standards 66: 523-530.
Boursnell MEG, Entwistle C, Blakeley D, Roberts C, Duncan IA, Chisholm SE, Martin GM, Jennings R, Challanain DNi, Sobek I and McLean CS (1997) A genetically inactivated Herpes Simplex virus type 2 (HSV) vaccine provides effective protection against primary and recurrent HSV disease. J Infect Dis 175: 16-25.
Corey L (1993) Herpes simplex virus infections during the decade since the licensure of acyclovir. J Med Virol Suppl 1: 7-12.
Degouys V, Montoisy C, Wanderpepen M-P, Togasaki RK, Surzycki SJ, Segault V and Miller, AOA (1997) 'Bionebulisation': an alternative to Dounce homogenisation for the preparation of mammalian cells nuclei in bulk. In: Carronda MJT, Griffiths B and Moreira LP (eds) Animal Cell Technology (From Vaccines to Genetic Medicine), (pp. 271-275) Kluwer Academic Publishers Dordrecht, Holland.
Dougherty (1964) Animal Virus Titration Techniques in Techniques. In: Harris RJC (ed) Experimental Virology, Academic Press Ltd, London.
Fabry L, Baijot B, D'Hondt E and Duchene M (1989) High density microcarrier cell culture for viral vaccine production. Advances in animal cell biology and technology for bioprocesses. Spier RE, Griffiths JB, Stephenne J and Crooy PJ (eds) (pp. 361-365) Butterworths, Sevenoaks, UK.
Farrell HE, McLean CS, Harley C, Efstathiou S, Inglis SC and Minson AC (1994) Vaccine potential of a herpes simplex virus type 1 mutant with an essential glycoprotein deleted. J Virol 68: 927-932.
Giard DJ, Thilly WG, Wang DIC and Levine DW (1977) Virus production with a newly developed microcarrier system. Appl Environ Micro 34(6): 668-672.
Griffiths B, Thornton B and McEntee I (1980) Production of Herpes viruses in microcarrier cultures of human diploid and primary chick fibroblast cells. Eur J Cell Biol 22: 606.
Griffiths B, Thornton B and McEntee I (1982) The development and use of microcarrier and glass sphere culture techniques for the production of herpes simplex viruses. Develop Biol Standard 50: 103-110.
Hu W-S, Giard DJ and Wang DIC (1985) Serial propagation of mammalian cells on microcarriers. Biotech and Bioeng 28: 1466-1476.
Lesko J, Veber P, Hrda M and Feketevova M (1993) Large scale production of infectious bovine rhinotracheitis virus in cell culture on microcarriers. Acta Virol 37: 73-78.
McLean CS, Erturk M, Jennings R, Ní Challanaín D, Minson AC, Duncan I, Boursnell MEG and Inglis SC (1994) Protective vaccination against primary and recurrent disease cause by herpes simplex virus type 2 using a genetically disabled herpes simplex type 1 virus. J Inf Dis 170: 1100-1109.
Meignier B (1978) Cell culture on beads used for the industrial production of foot and mouth disease virus. Develop Biol Standards 42: 141-145.
Montagnon BJ, Fanget B and Nicolas AJ (1981) The large-scale cultivation of Vero cells in microcarrier culture for virus vaccine production preliminary results for killed poliovirus vaccine. Develop Biol Standard 47: 55-64.
Reed LJ and Muench H (1938) A simple method of estimating 50% endpoints. American Journal of Hygiene 27(3): 493-497.
Sanford KK, Earle WR, Evans VJ et al. (1951) The measurement of proliferation in tissue cultures by enumeration of cell nuclei. J Nat Cancer Inst 11: 773-795.
Talbot PJ, Lapierre J, Daniel C, Dugre R and Trepanier P (1989) Growth of a murine coronavirus in a microcarrier system. J Virol Methods 25: 63-70.
Van Wezel AL (1972) New trends in the preparation of cell substrates for the production of virus vaccines. Progr Immunobiol Standard 5: 187-192.
Van Wezel AL (1973) Microcarrier cultures of animal cells in Tissue Culture: Methods and Applications, Kruse PF and Peterson MK (eds) (pp. 372-377) New York/London, Academic Press.
WHO Cell banks of continuous cell lines for the production of biologicals. Technical Report Series No. 786, 1989, WHO, Geneva.
Widell A, Hansson BG and Nordenfelt E (1984) A microcarrier cell culture system for large scale production of Hepatitis A virus. J Virol Methods 8: 63-71.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zecchini, T., Smith, R. Production of high titre disabled infectious single cycle (DISC) HSV from a microcarrier culture. Cytotechnology 30, 203–210 (1999). https://doi.org/10.1023/A:1008005200711
Issue Date:
DOI: https://doi.org/10.1023/A:1008005200711