Abstract
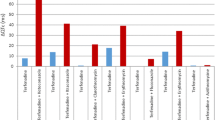
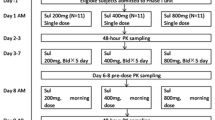
Procainamide is a class IA antiarrhythmic drug indicated for the treatment of life-threatening or symptomatic ventricular arrhythmias. The current sustained-release formulation requires 6-hour dosing (qid). To improve patient compliance, a new sustained-release formulation for twice-daily (bid) administration has been developed (Procanbid, Parke-Davis). This study assesses the pharmacologic equivalence of the bid and qid formulations in the suppression of symptomatic ventricular premature depolarizations (VPDs). Fourteen centers enrolled a total of 99 patients with frequent symptomatic VPDs (average ≥20 VPDs/hr) who previously responded to and tolerated the procainamide qid formulation. During the first week of the double-blind phase, patients were randomized to either placebo or procainamide dosages of 1000, 2000, or 4000 mg/d (bid or qid formulations). In the second week, the patients were crossed over to the alternate formulation. Seventy-seven patients qualified for the primary activity analysis. The bid and qid formulations showed comparable effectiveness in the suppression of mean VPDs with a linear dose-response relationship. The VPD suppression was not attenuated towards the end of the dosing interval for either formulation. Sixty-eight of these patients entered an optional 1-year extension to receive the bid formulation. Thirty-seven (54%) patients had adverse effects. Of those, 15 (22%) had side effects considered treatment related. Most of the adverse events occurred during the first 6 weeks of treatment. Only a few patients (8%) withdrew as a consequence of treatment with the bid formulation. The overall safety profile of the bid formulation was similar to other formulations, and the procainamide bid formulation has a low proarrhythmic rate (`3%). In conclusion, the effectiveness of the twice-daily formulation of procainamide in the suppression of VPDs is comparable to the currently available qid formulation.
Similar content being viewed by others
References
Hoffman BF, Rosen MR, Wit AL. Electrophysiology and pharmacology of cardiac arrhythmias. Cardiac effects of quinidine and procainamide. Am Heart J 1975;90:117–122.
Woske H, Belford J, Fastier FN, Brooks McC. The effect of procainamide on excitability, refractoriness and conduction in the mammalian heart. J Pharmacol Exp Ther 1953;107:134–140.
Hilleman DE. Bioavailability issues with procainamide. Arrhythmia Clinic 1992;5:3–8.
Lima JJ. Principles of therapeutic drug monitoring. In: Evans WE, Sclentag JJ, Jusko WJ, eds. Applied Pharmacokinetics. Vancouver, WA: 1992:22-1;22–33. Applied Therapeutics.
Smith TL, Kinkel A. Plasma levels of procainamide after administration of conventional and sustained-release preparations. Curr Therap Res 1980;27:217–228.
Baker BA, Reynolds JR, Gelckel L, A'Zary E, Bodenheimer MM. Comparative bioavailability of two oral sustained-release procainamide products. Clin Pharm 1988;7:135–138.
Yang BB, Abel RB, Uprichard ACG, Smithers JA, Forgue ST. Pharmacokinetic and pharmacodynamic comparisons of BID and QID formulations of procainamide in patients with frequent ventricular premature depolarizations. J Clin Pharmacol 1996;36:623–633.
Members of the Cardiac Arrhythmia Pilot Study (CAPS). Effects of encainide, flecainide, imipramine and moricizine on ventricular arrhythmias during the year after acute myocardial infarction: THE CAPS. Am J Cardiol 1988;61:501–509.
Caine ME, Josephson ME. Procainamide. In: Gould LA, ed. Drug Treatment of Cardiac Arrhythmias. New York: Futura, P 1983:73–108.
Michelson EL, Dreifus LS. Sustained-release and immediate-release procainamide: Comparative efficacy and equivalence in ambulatory patients with frequent premature ventricular contractions. Vasc Med 1984;12:155.
Anderson JL. Conventional and sustained release procainamide. Update on pharmacology and clinical use. Clin Ther 1985;7:618–640.
Giardina EGV, Fenster PE, Bigger JT Jr, Mayersohn M, Perrier D, Marcus FI. Efficacy, plasma concentrations and adverse effects of a new sustained release procainamide preparation. Am J Cardiol 1980;46:855–861.
Kerin NZ, Somberg J. Proarrhythmia: Definition, risk factors, causes, treatment and controversies. Am Heart J 1994;128:576–585.
Torres V, Flowers D, Somberg JC. The arrhythmogenicity of antiarrhythmic agents. Am Heart J 1985;109:1090–1097.
Velebit V, Podrid P, Lown B, Cohen BH, Graboys TB. Aggravation and provocation of ventricular arrhythmias by antiarrhytimic drugs. Circulation 1982;65:886–894.
Koch-Wesser J, Klein SM. Procainamide dosage schedules, plasma concentrations and clinical effects. JAMA 1971;215:1454–1460.
Kosowsky BD, Taylor J, Lown B, Ritchie RF. Long-term use of procainamide following acute myocardial infarction. Circulation 1973;42:1204–1210.
Karlsson E. Plasma levels of procainamide after administration of conventional and sustained-release tablets. Eur J Clin Pharmacol 1973;6:245–250.
Physicians Desk Reference. Procan SR prescribing information 1992;1760–1751.
Giardina EGV. Procainamide: Clinical pharmacology and efficacy against ventricular arrhythmias. In: Garfein OB, ed. Clinical Pharmacology of Cardiac Antiarrhythmic Agents: Classical and Current Concepts Reevaluated. New York: New York Academy of Sciences 1984:177–188.
Danielly J, DeJong R, Radke-Mitchell LC, Uprichard ACG. Procainamide-associated blood dyscrasias. Am J Cardiol 1994;74:1179–1180.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kerin, N.Z., Meengs, W.L., Timmis, G.C. et al. Activity of Procanbid Procainamide Twice-Daily Formulation, to Suppress Ventricular Premature Depolarizations. Cardiovasc Drugs Ther 11, 169–175 (1997). https://doi.org/10.1023/A:1007736931662
Issue Date:
DOI: https://doi.org/10.1023/A:1007736931662