Abstract
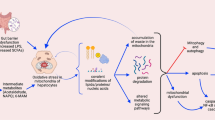
Mitochondria are involved in fatty acid β-oxidation, the tricarboxylic acid cycle, and oxidative phosphorylation, which provide most of the cell energy. Mitochondria are also the main source of reactive oxygen species in the cell and are involved in cell demise through opening of the mitochondrial permeability transition pore. It was therefore to be expected that mitochondrial dysfunction could be a major mechanism of drug-induced liver disease. Microvesicular steatosis (which may cause liver failure, coma, and death) is the consequence of severe impairment of mitochondrial β-oxidation. Endogenous compounds (such as cytokines or female sex hormones) or xenobiotics (including toxins such as ethanol and drugs such as aspirin, valproic acid, ibuprofen, or zidovudine) can inhibit β-oxidation directly or through a primary effect on the mitochondrial genome or the respiratory chain itself. In some patients, infections and cytokines, or inborn errors of β-oxidation enzymes or the mitochondrial genome, may favor the appearance of drug-induced microvesicular steatosis. Nonalcoholic steatohepatitis may develop under conditions causing prolonged, microvesicular, and/or macrovacuolar steatosis. In this condition, chronic impairment of mitochondrial β-oxidation (causing steatosis) and the respiratory chain (increasing the production of ROS) lead to lipid peroxidation, which, in turn, may cause the diverse lesions of steatohepatitis, namely, necrosis, inflammation, Mallory's bodies, and fibrosis. Finally, mitochondria are involved in several forms of drug-induced cytolytic hepatitis, through inhibition or uncoupling of respiration or through a drug-induced or reactive metabolite-induced mitochondrial permeability transition. The latter effect commits hepatocytes to either apoptosis or necrosis, depending on the number of organelles that have undergone the permeability transition.
Similar content being viewed by others
References
Berson A, Schmets L, Fau D et al. Inhibition by nilutamide of the mitochondrial respiratory chain and ATP formation. Possible contribution to the adverse effects of this antiandrogen. J Pharmacol Exp Ther. 1994; 270:167–76.
Berson A, Renault S, Lettéron P et al. Uncoupling of rat and human mitochondria: a possible explanation for tacrineinduced liver dysfunction. Gastroenterology 1996;110:1878–90.
Berson A, de Beco V, Lettéron P et al. Steatohepatitis-inducing drugs cause mitochondrial dysfunction and lipid peroxidation in rat hepatocytes. Gastroenterology 1998;114:764–74.
Bogenhagen DF. Repair of mtDNA in vertebrates. Am J Hum Genet. 1999;64:1276–81.
Botla R, Spivey JR, Bronk SF, Gores GJ. Ursodeoxycholic acid (UDCA) inhibits the mitochondrial membrane permeability transition induced by glycochenodeoxycholate: a mechanism of UDCA cytoprotection. J Pharmacol Exp Ther. 1995;272:930–8.
Chariot P, Drogou I, de Lacroix-Szmania I et al. Zidovudineinduced mitochondrial disorder with massive liver steatosis, myopathy, lactic acidosis, and mitochondrial depletion. J Hepatol. 1999;30:156–60.
Day CP, James OFW. Hepatic steatosis: innocent bystander or guilty party? Hepatology 1998;27:1463–6.
Deschamps D, Fisch C, Fromenty B, Berson A, Degott C, Pessayre D. Inhibition by salicylic acid of the activation and thus oxidation of long-chain fatty acids. Possible role in the development of Reye's syndrome. J Pharmacol Exp Ther. 1991;259:894–904.
Deschamps D, De Beco V, Fisch C, Fromenty B, Guillouzo A, Pessayre D. Inhibition by perhexiline of oxidative phosphorylation and the β-oxidation of fatty acids: possible role in pseudoalcoholic liver lesions. Hepatology. 1994;19:948–61.
Fau D, Eugène D, Berson A et al. Toxicity of the antiandrogen flutamide in isolated rat hepatocytes. J Pharmacol Exp Ther. 1994;269:954–62.
Fau D, Lekehal M, Farrell G et al. Diterpenoids from germander, a herbal medicine, induce apoptosis in isolated rat hepatocytes. Gastroenterology 1997;113:1334–46.
Fréneaux E, Labbe G, Lettéron P et al. Inhibition of the mitochondrial oxidation of fatty acids by tetracycline in mice and in man: possible role in microvesicular steatosis induced by this antibiotic. Hepatology 1988;8:1056–62.
Fréneaux E, Fromenty B, Berson A et al. Stereoselective and nonstereoselective effects of ibuprofen enantiomers on mitochondrial β-oxidation of fatty acids. J Pharmacol Exp Ther. 1990;255:529–35.
Fromenty B, Pessayre D. Inhibition of mitochondrial β-oxidation as a mechanism of hepatotoxicity. Pharmacol Ther. 1995;67:101–54.
Fromenty B, Fréneaux E, Labbe G et al. Tianeptine, a new tricyclic antidepressant metabolized by β-oxidation of its heptanoic side chain, inhibits the mitochondrial oxidation of medium and short chain fatty acids in mice. Biochem Pharmacol. 1989;38:3743–51.
Fromenty B, Fisch C, Labbe G et al. Amiodarone inhibits the mitochondrial β-oxidation of fatty acids and produces microvesicular steatosis of the liver in mice. J Pharmacol Exp Ther. 1990a;255:1371–6.
Fromenty B, Fisch C, Berson A, Lettéron P, Larrey D, Pessayre, D. Dual effect of amiodarone on mitochondrial respiration. Initial protonophoric uncoupling effect followed by inhibition of the respiratory chain at the levels of complex I and complex II. J Pharmacol Exp Ther. 1990b;255:1377–84.
Fromenty B, Grimbert S, Mansouri A et al. Hepatic mitochondrial DNA deletion in alcoholics: association with microvesicular steatosis. Gastroenterology 1995;108:193–200.
Fromenty B, Berson A, Pessayre D. Microvesicular steatosis and steatohepatitis: role of mitochondrial dysfunction and lipid peroxidation. J Hepatol. 1997;26(supplement1):13–22.
Genève J, Hayat-Bonan B, Labbe G et al. Inhibition of mitochondrial β-oxidation of fatty acids by pirprofen. Role in microvesicular steatosis due to this nonsteroidal anti-inflammatory drug. J Pharmacol Exp Ther. 1987;242:1133–7.
Grimbert S, Fromenty B, Fisch C et al. Decreased mitochondrial oxidation of fatty acids in pregnant mice: possible relevance to development of acute fatty liver of pregnancy. Hepatology 1993;17:628–37.
Grimbert S, Fisch C, Deschamps D et al. Effects of female sex hormones on liver mitochondria in non-pregnant female mice: possible role in acute fatty liver of pregnancy. Am J Physiol. 1995;268:G107–15.
Ibdah JA, Bennett MJ, Rinaldo P et al. A fetal fatty-acid oxidation disorder as a cause of liver disease in pregnant women. N Engl J Med. 1999;340:1723–31.
Kasahun K, Abbott F. In vivo formation of the thiol conjugates of reactive metabolites of 4-ene VPA and its analogue 4-pentenoic acid. Drug Metab Dispos. 1993;21:1098–106.
Kesterson JW, Granneman GR, Machinist JM. The hepatotoxicity of valproic acid and its metabolites in rats. I. Toxicologic, biochemical, and histopathologic studies. Hepatology 1984;4:1143–52.
Labbe G, Fromenty B, Fréneaux E et al. Effects of various tetracycline derivatives on in vitro and in vivo β-oxidation of fatty acids, egress of triglycerides from the liver, accumulation of hepatic triglycerides, and mortality in mice. Biochem Pharmacol. 1991;41:638–41.
Le Dinh T, Fréneaux E, Labbe G et al. Amineptine, a tricyclic antidepressant, inhibits the mitochondrial oxidation of fatty acids and produces microvesicular steatosis of the liver in mice. J Pharmacol Exp Ther. 1988;247:745–50.
Lemasters JJ, Nieminen AL, Qian T et al. The mitochondrial permeability transition in cell death: a common mechanism in necrosis, apoptosis and autophagy. Biochim Biophys Acta 1998;1366:177–96.
Lettéron P, Duchatelle V, Berson A et al. Increased ethane exhalation, an in vivo index of lipid peroxidation, in alcohol abusers. Gut 1993;34:409–14.
Lettéron P, Fromenty B, Terris B, Degott C, Pessayre D. Acute and chronic hepatic lipid steatosis lead to in vivo lipid peroxidation in mice. J Hepatol. 1995;24:200–8.
Lettéron P, Brahimi-Bourouina N, Robin MA, Moreau A, Feldmann G, Pessayre D. Glucocorticoids inhibit mitochondrial matrix acyl-coenzyme A dehydrogenases and fatty acid β-oxidation. Am J Physiol. 1997;272:G1141–50.
Lewis W, Dalakas MC. Mitochondrial toxicity of antiviral drugs. Nature Med. 1995;1:417–22.
Mahler H, Pasi A, Kramer JM et al. Fulminant liver failure in association with the emetic toxin of Bacillus cereus. N Engl J Med. 1997;336:1142–8.
Mansouri A, Fromenty B, Berson A et al. Multiple hepatic mitochondrial DNA deletions suggest premature oxidative aging in alcoholic patients. J Hepatol. 1997a;27:96–102.
Mansouri A, Gaou I, Fromenty B et al. Premature oxidative aging of hepatic mitochondrial DNA in Wilson's disease. Gastroenterology 1997b;113:599–605.
Mansouri A, Gaou I, de Kerguence C et al. An alcoholic binge causes massive degradation of hepatic mitochondrial DNA in mice. Gastroenterology 1999;117:181–90.
Pessayre D. Role of reactive metabolites in drug-induced hepatitis. J Hepatol. 1995;23:S16–24.
Pessayre D, Fromenty B, Mansouri A. Drug-induced steatosis and steatohepatitis. In: Lemasters JJ, Niemenen AL, eds. Mitochondria in pathogenesis. New York: Plenum Press; 2000a [In press].
Pessayre D, Feldmann G, Haouzi D, Fau D, Moreau A, Neuman M. Hepatocyte apoptosis triggered by natural substances (cytokines, other endogenous substances and foreign toxins). In: Cameron RG, Feuer G, eds. Apoptosis and its modulation by drugs. Heidelberg: Springer Verlag; Handbook of Experimental Pharmacology. 2000b;142:59–108.
Pessayre D, Haouzi D, Fau D, Robin MA, Mansouri A, Berson A. Withdrawal of life support, altruistic suicide, fratricidal killing and euthanasia by lymphocytes: different forms of drug-induced hepatic apoptosis. J Hepatol. 1999;31:760–70.
Robin MA, Maratrat M, Le Roy M et al. Antigenic targets in tienilic acid hepatitis. Both cytochrome P450 2C11 and 2C11-tienilic acid adducts are transported to the plasma membrane of rat hepatocytes and recognized by human sera. J Clin Invest. 1996;98:1471–80.
Saraste M. Oxidative phosphorylation at the fin de siècle. Science 1999;283:1488–93.
Schon EA, Bonilla E, DiMauro S. Mitochondrial DNA mutations and pathogenesis. J Bioenerg Biomembr. 1997;29:131–49.
Shan B, Vazquez E, Lewis JA. Interferon selectively inhibits the expression of mitochondrial genes: a novel pathway for interferon-mediated responses. EMBO J. 1990;9:4307–14.
Shigenaga MK, Hagen TM, Ames BN. Oxidative damage and mitochondrial decay in aging. Proc Natl Acad Sci USA 1994;91:10771–8.
Susin SA, Zamzami N, Kroemer G. Mitochondria as regulators of apoptosis: doubt no more. Biochim Biophys Acta 1998;1366:151–65.
Zamzami N, Susin SA, Marchetti P et al. Mitochondrial control of nuclear apoptosis. J Exp Med. 1996;183:1533–44.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pessayre, D., Mansouri, A., Haouzi, D. et al. Hepatotoxicity due to mitochondrial dysfunction. Cell Biol Toxicol 15, 367–373 (1999). https://doi.org/10.1023/A:1007649815992
Issue Date:
DOI: https://doi.org/10.1023/A:1007649815992