Abstract
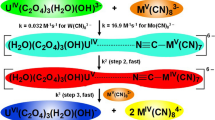
The kinetics of the oxidation of PhCHO by OsVIII has been studied in 0.01–0.05 M [OH−] range. Fe(CN)3− 6 was used to regenerate OsVIII. The very low solubility of PhCHO in H2O restricted the study to the 0.0024–0.0036 M [PhCHO] range. A mechanism involving the PhCHO hydrate has been proposed.
Similar content being viewed by others
References
S.P.S. Mehta, Transition Met. Chem., 25, 644 (2000).
I.L. Finar, Organic Chemistry, Vol. 1, 6th edit., ELBS, 1973, p. 737.
F. Aziz and G.A. Mirja, Talanta, 11, 889 (1964).
G.A. Bridgart, W.A. Waters and I.R. Wilson, J. Chem. Soc., Dalton Trans., 1582 (1973).
B.M. Chadwick and A.G. Sharpe, Adv. Inorg. Chem. Radiochem., 8, 83 (1966).
D. Mohan and Y.K. Gupta, J. Chem. Soc., Dalton Trans., 1085 (1977).
R.T. Morrison and R.N. Boyd, Organic Chemistry, Prentice Hall of India, New Delhi, 1995, p. 715.
K. Nakanishi, Infrared Absorption Spectroscopy, Holden Day, Inc., San Francisco, 1977, p. 40.
F. Krauss and D. Wilken, Z. All. Chem., 145, 166 (1925).
W.P. Griffith, (a) J. Chem. Soc., 245 (1964); (b) Quart. Rev., 19, 254 (1965).
M. Prasada Rao, B. Sethuram and T. Navneeth Rao, Indian J. Chem., 19A, 1980, p. 562.
N. Swarnlakshmi, V. Uma, B. Sethuram and T. Navneeth Rao, Indian J. Chem., 26A, 1987, p. 592.
S.P.S. Mehta and Raj N. Mehrotra, Transition Met. Chem., 17, 50 (1992).
V. Gold and D. Bethell, Advances in Physical Organic Chemistry, Vol. 18, Academic Press, New York, 1982, p. 162.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mehta, S.P.S., Tewary, G. Kinetics and mechanism of metal ion catalysis: Part II. Oxidation of PhCHO by osmium(VIII) continuously regenerated by hexacyanoferrate(III) ions. Transition Metal Chemistry 26, 290–294 (2001). https://doi.org/10.1023/A:1007184621336
Issue Date:
DOI: https://doi.org/10.1023/A:1007184621336