Abstract
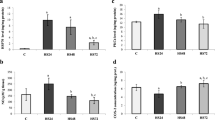
We examined by Western blots the effect of variations of the heating sessions, such as duration and intensity on the following aspects: 70-kDa heat shock protein (HSP70) and HSP72 induction. Protein ubiquitination PLCγ PKCε and PKCα levels in murine liver and brain were also studied. Results demonstrated that maximal induction of HSP72 was obtained after heat shock at 43.5°C in both organs. Preconditioning at lower temperatures (either acclimation to 39°C or induction of thermotolerance to 43.5°C with a single exposure to 39°C) attenuated the heat shock response. Hepatic HSP72 induction was elicited only as a consequence of hyperthermia since either fasting or restraint were unable to trigger its synthesis. On the contrary, a ubiquitination decrease of a 31 kDa protein was obtained both after hyperthermia and fasting This indicates that the latter is a more generic response of hepatic cells to noxious stimuli. Analysis of the above mentioned enzymes showed that in liver of naive mice PKCα is barely present while PKCε is quite abundant. All hyperthermic treatments caused a general decrease of the latter, except for the heat shock at 43.5°C that caused an increase. PLCγ decreased after all heating sessions. It is known that hyperthermia in the range of 41-45°C induces apoptotic death in many cell types. Therefore we analyzed the presence of the typical apoptotic DNA ladder. Our data strongly suggest that both hyperthermia and restraint induce necrosis in liver while apoptosis and necrosis become evident in brain. All these effects are still present 24 h from the last heating session: This indicates thatin vivo, hyperthermia produces long term modifications of the hepatic cell.
Similar content being viewed by others
References
Welch WJ: Mammalian stress response: cell physiology, structure/function of stress proteins and implications for medicine and disease. Physiol Rev 72: 1063-1081, 1992
Lindquist S, Craig EA: The heat shock proteins. Annu Rev Genet 22: 631-677, 1988
Carlson N, Rogers S, Rechsteiner M: Microinjection of ubiquitin: changes in protein degradation in HeLa cells subjected to heat shock. J Cell Biol 104: 547-555, 1987
Li GC, Werb Z: Correlation between synthesis of heat shock proteins and development of thermotolerance in Chinese hamster fibroblasts. Proc Natl Acad Sci USA 79: 3218-3222, 1982
Moseley PL: Mechanisms of heat adaptation: Thermotolerance and acclimatization. J Lab Clin Med 123: 48-52, 1994
Calderwood SK, Stevenson MA: Inducers of the heat shock response stimulate phospholipase C and Phospholipase A2 activity in mammalian cells. J Cell Physiol 155: 248-256, 1993
Dubois MF, Bensaude O: MAP kinase activation during heat shock in quiescent and exponentially growing mammalian cells. FEBS Lett 324: 191-195, 1993
Kim SH, Kim JH, Erdos G, Lee YJ: Effect of staurosporine on suppression of heat shock gene expression and thermotolerance development in HT-29 cells. Biochem Biophys Res Commun 193 (2): 759-763, 1993
Erdos G, Lee YJ: Effect of staurosporine on the transcription of HSP70 heat shock gene in HT-29 cells. Biochem Biophys Res Commun 202: 476-483, 1994
Bagi G, Hidvegi EJ: Protein phosphorylation and kinase activities in tumour cells after hyperthermia. Int J Radiat Biol 58: 633-650, 1990
Wooten MW: Alterations in protein kinase C type III-alpha during heat shock of rat embryo fibroblasts. Exp Cell Res 193: 274-278, 1991
Ritz MF, Masmoudi A, Matter N, Rogue P, Lang D, Freysz L, Malviya AN: Heat stressing stimulates nuclear protein kinase C raising diacylglycerol levels. Nuclear protein kinase C activation precedes HSP70 mRNA expression. Receptor 3: 311-324, 1993
Gabai VL, Meriin AB, Mosser DD, Caron AW, Rits S, Shifrin VI, Sherman MY: HSP70 prevents activation of stress kinases. J Biol Chem 272: 18033-18037, 1997
Flanagan SW, Ryan AJ, Gisolfi CV, Moseley: Tissue-specific HSP70 response in animals undergoing heat stress. Am J Physiol 268: 28-32, 1995
Strasser A, Anderson RL: Bcl-2 and thermotolerance cooperate in cell survival. Cell Growth Differ 5: 411-417, 1994
Oberhammer FA, Roberts RA: Apoptosis: a wide spread process involved in liver adaptation and carcinogenesis. In: I.M. Arias, J.L. Boyer, N. Fausto, W.B. Jakoby, D.A. Schachter, D.A. Shafritz (eds). The Liver: Biology and Pathobiology. Raven Press, Ltd, New York, 1994, pp 1547-1555
Beck SC, Paidas CN, Tan H, Yang J, De Maio A: Depressed expression of the inducible form of HSP70 (HSP72) in brain and heart after in vivo heat shock. Am J Physiol 269: 608-613, 1995
Tilly JL, Hsueh AJW: Microscale autoradiographic method for the qualitative and quantitative analysis of apoptotic DNA fragmentation. J Cell Physiol. 154:519-526, 1993
de Feo G, Lisciani R, de Joannon CA, Mazzanti G, Tolu L, Pace SA: Total body hyperthermia animal model for pharmacological studies. In Vivo 3:295-300, 1989
Fawcett TW, Sylvester SL, Sarge KD, Morimoto RI, Holbrook NJ: Effects of neurohormonal stress and aging on the activation of mammalian heat shock factor 1. J Biol Chem 269: 32272-32278, 1994
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Leoni, S., Brambilla, D., Risuleo, G. et al. Effect of different whole body hyperthermic sessions on the heat shock response in mice liver and brain. Mol Cell Biochem 204, 41–47 (2000). https://doi.org/10.1023/A:1007053504960
Issue Date:
DOI: https://doi.org/10.1023/A:1007053504960