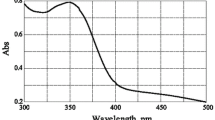
Abstract
Six transition metal(II) complexes with 8-acetyl-4-methyl umbelliferone (AMUH), ML2 (L = AMU−; M = Mn, Co, Ni, Cu, Zn and Cd), have been synthesized and characterized by elemental analysis, electrical conductance, i.r., 1H-n.m.r. and u.v. spectroscopic techniques. The e.s.r. spectra show that CuL2 is anisotropic, with g ⊥ ≈ 2.05 and g∥≈2.26. The MnL2 species is characterized by a very broad g≈2 centered resonance without manganese hyperfine structure. The antioxidative and other biological activities of selected complexes were also investigated, indicating that both the ligand and complexes exhibit a scavenging effect on the superoxide radical (O2 −·) and a suppressing effect on the hydroxyl radical (·OH). The suppressing effect on ·OH is greater than the scavenging effect on O2 −·. The Cu and Mn complexes exhibit very significant suppression ratios for both radicals. However, the inhibitory effect on lipid peroxides results show that, in the initial stage, the Cu and Mn complexes exhibit obvious inhibitory effects on lipid peroxides, but after one hour, they begin to accelerate the lipid peroxides. Lower catalytic activity and instability when the complexes react with active oxygen may be responsible for this dual nature. The ligand and four transition metal(II) complexes selectively, obviously, inhibit the growth of HCT-8 and HL-60 tumour cell lines. They also exhibit a minor influence on the proliferation of B cells in higher concentration (10−5 m), but only a weak effect on the proliferation of T cells of the BALB/C (nude rate) spleen cells.
Similar content being viewed by others
References
B. Halliwell and J.M. Gutteridge, Methods in Enzymology, 186, 1 (1990).
S.J. Stohs and D. Bagchi, Free Radical Biology and Medicine, 18, 321 (1995).
W.K. Pogozelski and T.D. Tullius, Chem. Rev., 98, 1089 (1998).
R.M. Burger, Chem. Rev., 98, 1153 (1998).
C.J. Burrows and J.G. Muller, Chem. Rev., 98, 1109 (1998).
D. Schmahl, Carcinogenicity of Anticancer Drugs and especially Alkylating Agents, in Carcinogenicity of Alkylating Cytostatic Drugs, D. Schmahl and J.M. Kdldor (Eds.), IARC Scientific Publications No. 78, International Agency for Research on Cancer, Lyon, 1986, pp. 29-36.
R.B. Arora and C.N. Mathur, Brit. J. Pharmacol., 20, 29 (1963).
C.R. Hancock, H.W.B. Barlow and H.J. Lacey, J. Exptl. Botany, 12, 401 (1961).
S. Lehnet, G. Fisher and G. Methot, Int. J. Radiat. Biol., 40, 63 (1981).
Z.S. Tong, M.X. Huang and C.S. Dai, Chinese J. Med. Chem., 4, 150 (1994).
V.J.T. Raju, V. Ranbaore, V. Atre and M.C. Ganorkar, J. Indian Chem. Soc., LIX, 199 (1982).
P. Singh, V. Singh, R.L. Goel and B.P. Singh, J. Indian Chem. Soc., LII, 958 (1975).
M.N. Hughes, The Inorganic Chemistry of Biological Processes, Wiley Interscience, London, 1972.
L.M. Mosmmann, J. Immuno. Meth., 65, 55 (1983).
V. Ponti, M.V. Dianzani, K.J. Cheeseman and T.F. Stater, Chem. Biol. Interact, 23, 281 (1978).
B. Halliwell and J.M.C. Gutteridge, Febs Letters, 128, 347 (1981).
E. Niki, E. Komuro, M. Takahashi, S. Urano, E. Ito and K. Terao, J. Biol. Chem., 263, 19809 (1988).
D.R. Davies and G.A. Webb, Coord. Chem. Rev., 6, 95 (1971).
K. Nakamoto, Infrared and Raman Spectra of Inorganic and Coordination Compounds, 3rd Edit., Wiley Interscience, New York, 1978.
Q. Zhang, L.F. Wang, X. Liu, S.B. Li and F.Y. He, Transition Met. Chem., 21, 23 (1996).
T. Minegishi, T. Hoshi and Y. Tanizaki, Nippon Kagaku Kaishi, 649 (1978).
B. Mabad, P. Cassoux, J.P. Tuchagues and D.N. Hendrickson, Inorg. Chem., 25, 1420 (1986).
R.D. Dowsing, J.F. Gibson, M. Goodgame and P.J. Hayward, J. Chem. Soc. A, 187 (1969).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhang, Q., Zhai, JJ., Zhang, YR. et al. Antioxidative and antitumour properties of metal(II) solid complexes with 8-acetyl-4-methyl umbelliferone. Transition Metal Chemistry 25, 93–98 (2000). https://doi.org/10.1023/A:1007036718734
Issue Date:
DOI: https://doi.org/10.1023/A:1007036718734