Abstract
In 1958 Professor Setsuro Ebashi found that serum creatine kinase activity is increased in patients suffering from various muscular dystrophies, especially Duchenne muscular dystrophy (DMD). He and others proposed that creatine kinase passes through the cell membrane as it is released from DMD muscle fibers.
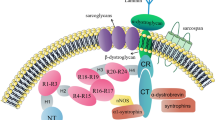
Since then, it has been found that dystrophin and dystrophin-associated proteins are connected to several other components, including the basal lamina and subsarcolemmal cytoskeletal networks on the cell membrane, while dystrophin anchors these dystrophin-associated proteins to the actin filaments inside the muscle cell. In DMD muscle, dystrophin has been found to be absent and dystroglycans and sarcoglycans decreased. However, how creatine kinase molecules can pass through the DMD muscle cell membrane still remains unanswered.
On the basis of recent findings on the structure of the protein layers which sandwich the lipid bilayer of muscle cell membranes, this essay stresses the importance of these lipid bilayers in protecting creatine kinase release from protoplasma in normal muscle. It further indicates the possibility that the absence of dystrophin in DMD muscle during muscle contraction may result in temporal damage to the lipid bilayer.
Similar content being viewed by others
References
Koenig M, Hoffman EP, Bertelson CJ, Monaco AP, Feener C, Kunkel LM: Complete cloning of the Duchenne muscular dystrophy (DMD) cDNA and preliminary genomic organization of the DMD gene in normal and affected individuals. Cell 50: 509–517, 1987
Hoffman EP, Brown RH Jr, Kunkel LM: Dystrophin: The protein product of the Duchenne muscular dystrophy locus. Cell 51: 919–928, 1987
Koenig M, Monaco AP, Kunkel LM: The complete sequence of dystrophin predicts a rod-shaped cytoskeletal protein. Cell 53: 219–228, 1988
Hoffman EP, Kunkel LM: Dystrophin abnormalities in Duchenne/Becker muscular dystrophy. Neuron 2: 1019–1029, 1989
Arahata K, Ishiura S, Ishiguro T, Tsukahara T, Suhara Y, Eguchi C, Ishihara T, Nonaka I, Ozawa E, Sugita H: Immunostaining of skeletal and cardiac muscle surface membrane with antibody against Duchenne muscular dystrophy peptide. Nature 333: 861–863, 1988
Watkins SC, Hoffman EP, Slayter HS, Kunkel LM: Immunoelectron microscopic localization of dystrophin in myofibres. Nature 333: 863–866, 1988
Hoffman EP, Fischbek KH, Brown RH, Johnson M, Medori R, Loike JD, Harris JB, Waterston R, Brooke M, Specht L, Kupsky W, Chamberlain J, Caskey T, Shapiro F, Kunkel LM: Characterization of dystrophin in muscle-biopsy specimens from patients with Duchenne's or Becker's muscular dystrophy. New Engl J Med 318: 1363–1368, 1988
Arahata K, Hoffman EP, Kunkel LM, Ishiura S, Tsukahara T, Ishihara T, Sunohara N, Nonaka I, Ozawa E, Sugita H: Dystrophin diagnosis: Comparison of dystrophin abnormalities by immunofluorescence and immunoblot analyses. Proc Natl Acad Sci USA 86: 7154–7158, 1989
Emery AEH, Emery MLH: The History of a Genetic Disease Duchenne Muscular Dystrophy or Meryon's Disease. The Royal Society of Medicine Press Limited, New York, 1995
Ebashi S, Toyokura Y, Momoi H, Sugita H: High creatine phosphokinase activity of sera of progressive muscular dystrophy. J Biochem 46: 103–104, 1959
Okinaka S, Sugita H, Momoi H, Toyokura Y, Kumagai H, Ebashi S, Fujie Y: Serum creatine phosphokinase and aldolase activity in neuromuscular disorders. Transact Amer Neurol Assoc 8462–64, 1959
Dreyfus J-C, Schapira G, Démos J: Etude de la créatine-kinase sérique chez les myopathes et leurs familles. Revue Française Etude Clinique et Biologique 5: 384–386, 1960
Engel AG, Yamamoto M, Fischbeck KH: Dystrophinopathies. In: A.G. Engel, C. Franzini-Armstrong (eds). Myology. McGraw-Hill Inc, New York, 1994, pp 1133–1187
Sugita H, Tyler FH: Pathogenesis of muscular dystrophy. Trans Assoc Amer Physicians 76: 231–243, 1963
Goto I, Nagamine M, Kathuki S: Creatine phosphokinase isozymes in-muscle. Arch Neurol 20: 422–429, 1969
Somer H, Donner M, Murros J, Konttinen A: A serum isozyme study in muscular dystrophy. Arch Neurol 29: 343–345, 1973
Takahashi K, Shutta K, Matsuo B, Takai T, Takao H, Imura H: Serum creatine kinase isoenzymes in Duchenne muscular dystrophy. Clinica Chimica Acta 75: 435–442, 1977
Rowland LP: The membrane theory of Duchenne dystrophy: Where is it? Ital J Neurol Sci 3(suppl): 13–28, 1984
Tinsley JM, Blake DJ, Zuellig RA, Davies KE: Increasing complexity of the dystrophin-associated protein complex. Proc Natl Acad Sci USA 91: 8307–8313, 1994
Ozawa E, Yoshida M, Suzuki A, Mizuno Y, Hagiwara Y, Noguchi S: Dystrophin-associated proteins in muscular dystrophy. Hum Mol Genet 4: 1711–1716, 1995
Campbell KP: Three muscular dystrophies: Loss of cytoskeletonextracellular matrix linkage. Cell 80: 675–679, 1995
Fawcett DW: A Textbook of Histology. Chapman & Hall, New York, 1994, pp 260–308
Zubrzycka-Gaarn EE, Bulman DE, Karpati G, Bughes AHM, Belfall B, Klamut HJ, Talbot J, Hodges RS, Ray PN, Worton RG: The Duchenne muscular dystrophy gene product is localized in sarcolemma of human skeletal muscle. Nature 333: 466–469, 1988
Bonilla E, Samitt CE, Miranda AF, Hays AP, Salviati G, DiMauro S, Kunkel LM, Hoffman EP, Rowland LP: Duchenne muscular dystrophy: Deficiency of dystrophin at the muscle cell surface. Cell 54: 447–452, 1988
Cullen MJ, Watkins SC: Ultrastructure of muscular dystrophy: New Aspects. Micron 24: 287–307, 1993
Ervasti JM, Campbell KP: Membrane organization of the dystrophinglycoprotein complex. Cell 66: 1121–1131, 1991
Ervasti JM, Ohlendieck, K, Kahl SD, Gaver MG, Campbell KP: Deficiency of a glycoprotein component of the dystrophin complex in dystrophic muscle. Nature 345: 315–319, 1990
Yoshida M, Ozawa E: Glycoprotein complex anchoring dystrophin to sarcolemma. J Biochem 108: 748–752, 1990
Nigro V, Piluso G, Belsito A, Politano J, Puca AA, Papparella S, Rossi E, Viglietto G, Esposito MG, Abbondanza C, Medici N, Molinari AM, Nigro G, Puca GA: Identification of a novel sarcoglycan gene at 5q33 encoding a sarcolemmal 35 kDa glycoprotein. Hum Mol Genet 5: 1179–1186, 1996
Jung D, Duclos F, Apostol B, Straub V, Lee JC, Allamand V, Venzke, DP, Sunada Y, Moomaw CR, Leveille CJ, Slaughter CA, Crawford TO, McPherson JD, Campbell KP: Characterization of δ-sarcoglycan, a novel component of the oligomeric sarcoglycan complex involved in limb-girdle muscular dystrophy. J Biol Chem 271: 32321–32329, 1996
Yoshida M, Noguchi S, Wakabayashi E, Piluso G, Belsito A, Nigro V, Ozawa E: The fourth component of the sarcoglycan complex. FEBS Lett 403: 143–148, 1997
Yamamoto H, Hagiwara Y, Mizuno Y, Yoshida M, Ozawa E: Heterogeneity of dystrophin-associated proteins. J Biochem 114: 132–139, 1993
Yoshida M, Suzuki A, Yamamoto H, Noguchi S, Mizurto Y, Ozawa E: Dissociation of the complex of dystrophin and its associated proteins into several unique groups by n-octyl β-D-glucoside. Eur J Biochem 222: 1055–1061, 1994
Ibraghimov-Beskrovnaya O, Ervasti JM, Leveille CJ, Slaughter CA, Sernett SW, Campbell KP: Primary structure of dystrophin-associated glycoproteins linking dystrophin to the extracellular matrix. Nature 355: 696–702, 1992
Roberds SL, Leturcq F, Allamand V, Piccolo F, Jeanpierr M, Anderson RD, Lim LE, Lee JC, Tomé FMS, Romero NB, Fardeau M, Beckmann JS, Kaplan J-Q Campbell KP: Missense mutations in the adhalin gene linked to autosomal recessive muscular dystrophy. Cell 78: 625–633, 1994
McNally EM, Yoshida M, Mizurto Y, Ozawa E, Kunkel LM: Human adhalin is alternatively spliced and the gene is located on chromosome 17q21. Proc Natl Acad Sci USA 91: 9690–9694, 1994
Lim LE, Duclos F, Broux O, Bourg N, Sunada Y, Allamand V, Meyer J, Richard I, Moomaw C, Slaughter C, Tomé FMS, Fardeau M, Jackson CE, Beckmann JS, Campbell KP: β-sarcoglycan: Characterization and role in limb-girdle muscular dystrophy linked to 4q12. Nature Genet 11: 257–265, 1995
Bönnemann CG, Modi R, Noguchi S, Mizuno Y, Yoshida M, Gussoni E, McNally EM, Duggan DJ, Angelini C, Hoffman EP, Ozawa E, Kunkel LM: β-sarcoglycan (A3b) mutations cause autosomal recessive muscular dystrophy with loss of the sarcoglycan complex. Nature Genet ll: 266–273, 1995
Noguchi S, McNally EM, Ben Othmane K, Hagiwara Y, Mizuno Y, Yoshida M, Yamamoto H, Bönnermann CG, Gussoni E, Denton PH, Kyriakides T, Middleton L, Hentati F, Ben Hamida M, Nonaka I, Vance JM, Kunkel LM, Ozawa E: Mutations in the dystrophin-associated protein γ-sarcoglycan in chromosome 13 muscular dystrophy. Science 270: 819–822, 1995
Ozawa E, Noguchi S, Mizuno Y, Hagiwara Y, Yoshida M: From dystrophinopathy to sarcoglycanopathy. Evolution of a concept of muscular dystrophy. Muscle Nerve 21: 421–438, 1998
Mizuno Y, Yoshida M, Yamamoto H, Hirai S, Ozawa E: Distribution of dystrophin isoforms and dystrophin-associated proteins 43DAG (A3a) and 50DAG (A2) in various monkey tissues. J Biochem 114: 936–941, 1993
Yamamoto H, Mizuno Y, Hayashi K, Nonaka I, Yoshida M, Ozawa E: Expression of dystrophin-associated protein 35DAG (A4) and 50DAG (A2) is confined to striated muscles. J Biochem 115: 162–167, 1994
Mizuno Y, Noguchi S, Yamamoto H, Yoshida M, Suzuki A, Hagiwara Y, Hayashi, YK, Arahata K, Nonaka I, Hirai S, Ozawa E: Selective defect of sarcoglycan complex in severe childhood autosomal recessive muscular dystrophy muscle. Biochem Biophys Res Comm 203: 979–983, 1994
Adams ME, Butler MH, Dwyer TM, Peters MF, Mumane AA, Froehner SC: Two forms of mouse syntrophin, a 58 kD dystrophin-associated protein, differ in primary structure and tissue distribution. Neuron 11: 531–540, 1993
Peters MF, Kramarcy, NR, Sealock R, Froehner SC: β2-syntrophin: Localization at the neuromuscular junction in skeletal muscle. NeuroReport 5: 1577–1580, 1994
Ahn AH, Yoshida M, Anderson MS, Feener CA, Selig S, Hagiwara Y, Ozawa E, Kunkel LM: Cloning of human basic Al, a distinct 59-kDa dystrophin-associated protein encoded on chromosome 8q23–24. Proc Natl Acad Sci USA 91: 4446–4450, 1994
Adams ME, Dwyer TM, Dowler LL, White RA, Froehner SC: Mouse α1-and β2-syntrophin gene structure, chromosome localization, and homology with a discs large domain. J Biol Chem 270: 25859–25865, 1995
Yang B, Ibraghimov-Beskrovnaya O, Moomaw CR, Slaughter CA, Campbell KP: Heterogeneity of the 59-kDa dystrophin-associated protein revealed by cDNA cloning and expression. J Biol Chem 269: 6040–6044, 1994
Ahn AH, Freener CA, Gussoni E, Yoshida M, Ozawa E, Kunkel LM: The three human syntrophin genes are expressed in diverse tissues, have distinct chromosomal locations, and each bind to dystrophin and its relatives. J Biol Chem 271: 2724–2730, 1996
Carr C, Fischbach GD, Cohen JB: A novel 87,000-Mr protein associated with acetylcholine receptors in Torpedo electric organ and vertebrate skeletal muscle. J Cell Biol 109: 1753–1764, 1989
Wagner KR, Cohen JB, Huganir RL: The 87K postsynaptic membrane protein from Torpedo is a protein-tyrosine kinase substrate homologous to dystrophin. Neuron 10: 511–522, 1993
Yoshida M, Yamamoto H, Noguchi S, Mizuno Y, Hagiwara Y, Ozawa E: Dystrophin-associated protein A0 is a homologue of the Torpedo 87K protein. FEBS Lett 367: 311–314, 1995
Blake DJ, Nawrotzki R, Peters MF, Froehner SC, Davies KE: Isoform diversity of dystrobrevin, the murine 87-kDa postsynaptic protein. J Biol Chem 271: 7802–7810, 1996
Sadoulet-Puccio HM, Khurana TS, Cohen JB, Kunkel LM: Cloning and characterization of the human homologue of a dystrophin related phosphoprotein found at the Torpedo electric organ post-synaptic membrane. Hum Mol Genet 5: 489–496, 1996
Suzuki A, Yoshida M, Ozawa E: Mammalian α1-and β1-syntrophin bind to the alternative splice-prone region of the dystrophin COOH terminus. J Cell Biol 128: 373–381, 1995
Ahn AH, Kunkel LM: Syntrophin binds to an alternatively spliced exon of dystrophin. J Cell Biol 128: 363–371, 1995
Yang B, Jung D, Rafael JA, Chamberlain JS, Campbell KP: Identification of α-syntrophin binding to syntrophin triplet, dystrophin, and utrophin. J Biol Chem 270: 4975–4978, 1995
Castelló A, Brochériou V, Chafey P, Kahn A, Gilgenkrantz H: Characterization of the dystrophin-syntrophin interaction using the two-hybrid system in yeast. FEBS Lett 383: 124–128, 1996
Ervasti JM, Kahl SD, Campbell KP: Purification of dystrophin from skeletal muscle. J Biol Chem 266: 9161–9165, 1991
Ohlendieck K, Ervasti JM, Snook JB, Campbell KP: Dystrophinglycoprotein complex is highly enriched in isolated skeletal muscle sarcolemma. J Cell Biol 112: 135–148, 1991
Suzuki A, Yoshida M, Yamamoto H, Ozawa E: Glycoprotein-binding site of dystrophin is confined to the cysteine-rich domain and the first half of the carboxy-terminal domain. FEBS Lett 308: 154–160, 1992
Beggs AH, Hoffman EP, Snyder JR, Arahata K, Specht L, Shapiro F, Angelini C, Sugita H, Kunkel LM: Exploring the molecular basis for variability among patients with Becker muscular dystrophy: dystrophin gene and protein studies. Am J Hum Genet 49: 54–67, 1991
Monaco AP, Bertelson CJ, Liechti-Gallati S, Moser H, Kunkel LM: An explanation for the phenotypic differences between patients bearing partial deletions of the DMD locus. Genomics 2: 90–95, 1988
Wapenaar, MC, Kievits T, Hart KA, Abbs S, Blonden LAJ, den Dunne JT, Grootscholten PM, Bakker E, Verellen-Dumoulin Ch, Bobrow M, van Ommen GJB, Pearson PL: A deletion hot spot in the Duchenne muscular dystrophy gene. Genomics 2: 101–108, 1988
Suzuki A, Yoshida M, Hayashi K, Mizuno Y, Hagiwara Y, Ozawa E: Molecular organization at the glycoprotein-complex-binding site of dystrophin. Three dystrophin-associated proteins bind directly to the carboxy-terminal portion of dystrophin. Eur J Biochem 220: 283–292, 1994
Jung D, Yang B, Meyer J, Chamberlain JS, Campbell KP: Identification and characterization of the dystrophin anchoring site on β-dystroglycan. J Biol Chem 270: 27305–27310, 1995
Leivo I, Engvall E: Merosin, a protein specific for basement membranes of Schwann cells, striated muscle, and trophoblast, is expressed late in nerve and muscle development. Proc Natl Acad Sci USA 85: 1544–1548, 1988
Vuolteenaho R, Nissinen M, Sainio K, Byers M, Eddy R, Hirvonen H, Show, T B, Sariola H, Engvall E, Tryggvason K: Human laminin M chain (Merosin): Complete primary structure, chromosomal assignment, and expression of the M and A chain in human fetal tissues. J Cell Biol 124: 381–394, 1994
Iwata Y, Nakamura H, Fujiwara K, Shigekawa M: Altered membranedystrophin association in the cardiomyopathic hamster heart muscle. Biochem Biophys Res Commun 190: 589–595, 1993
Mizuno Y, Noguchi S, Yamamoto H, Yoshida M, Nonaka I, Hirai S, Ozawa E: Sarcoglycan complex is selectively lost in dystrophic hamster muscle. Am J Pathol 146: 530–536, 1995
Osari S, Kobayashi O, Yamashita Y, Matsuish T, Goto M, Yanabe Y, Migita T, Nonaka I: Basement membrane abnormality in merosinnegative congenital muscular dystrophy. Acta Neuropathol 91: 332–336, 1996
Tomé FMS, Evangelista T, Leclerc A, Sunada Y, Manole E, Estornet B, Barois A, Campbell KP: Congenital muscular dystrophy with merosin deficiency. C R Acad Sci III 317: 351–357, 1994
Hillaire D, Leclerc A, Faure S, Topaloglu H, Chiannilulchai N, Guicheney P, Grinas L, Legos P, Phipot J, Evangelista T, Routon MC, Mayer M, Pellissier JF, Estournet B, Barois A, Hentati F, Feingold N, Beckmann JS, Dubowitz V, Tomé FMS, Fardeau M: Localization of merosin-negative congenital muscular dystrophy to chromosome 6q2 by homozygosity mapping. Hum Mol Genet 3: 1657–1661, 1994
Yoshida M, Mizuno Y, Nonaka I, Ozawa E: A dystrophin-associated glycoprotein, A3a (one of 43DAG doublets), is retained in Duchenne muscular dystrophy muscle. J Biochem 114: 634–639, 1993
Love DR, Hill DF, Dickson G, Spurr NK, Byth BC, Varsden RF, Walsh FS, Edwards YH, Davies KE: An autosomal transcript in skeletal muscle with homology to dystrophin. Nature 339: 55058, 1989
Tanaka H, Ishiguro T, Eguchi C, Saito K, Ozawa E: Expression of a dystrophin-related protein associated with the skeletal muscle cell membrane. Histochemistry 96: 1–5, 1991
Mizuno Y, Nonaka I, Hirai S, Ozawa E: Reciprocal expression of dystrophin and utrophin in muscles of Duchenne muscular dystrophy patients, female DMD-carriers and control subjects. J Neurol Sci 119: 43–52, 1993
Mizuno Y, Yoshida M, Nonaka I, Hirai S, Ozawa E: Expression of utrophin (dystrophin-related protein) and dystrophin-associated glycoproteins in muscles from patients with Duchenne muscular dystrophy. Muscle Nerve 17: 206–216, 1994
thi Man N, Ellis JM, Love DR, Davies KE, Gatter KC, Dickson G, Morris GE: Localization of the DMDL gene-encoded dystrophinrelated protein using a panel of nineteen monoclonal antibodies: Presence at neuromuscular junctions, in the sarcolemma of dystrophic skeletal muscle, in vascular and other smooth muscles, and in proliferating brain cell lines. J Cell Biol 115: 1695–1700, 1991
Matsumura K, Ervasti JM, Ohlendieck, K, Kahl SD, Cambpell KP: Association of dystrophin-related protein with dystrophin-associated proteins in mdx mouse muscle. Nature 360: 588–591, 1992
Tinsley, JM, Blake DJ, Roche A, Fairbrother U, Riss J, Byth BC, Knight AE, Kendrick-Jones J, Suthers GK, Love DR, Edwards YH, Davies KE: Primary structure of dystrophin-related protein. Nature 360: 591–593, 1992
Love DR, Morris GE, Ellis JM, Fairbrother U, Marsden RF, Bloomfield JF, Edwards YH, Slater CP, Parry DJ, Davies KE: Tissue distribution of the dystrophin-related gene product and expression in the mdx and dy mouse. Proc Natl Acad Sci USA 88: 3243–3247, 1991
Bulfield G, Siller WG, Wight PAL, Moore KJ: X chromosome-linked muscular dystrophy (mdx) in the mouse. Proc Natl Acad Sci USA 81: 1189–1192, 1984
Sicinski P, Geng Y, Ryder-Cook AS, Barnard EA, Darlison MG, Barnard PJ: The molecular basis of muscular dystrophy in the mdx mouse: A point mutation. Science 244: 1578–1580, 1989
Bar S, Barnea E, Levy Z, Neuman S, Yaffe D, Nudel U: A novel product of the Duchenne muscular dystrophy gene which greatly differs from the known isoforms in its structure and tissue distribution. Biochem J 272: 557–560, 1990
Cox GA, Sunada Y, Campbell KP, Chamberlain JS: Dp71 can restore the dystrophin-associated glycoprotein complex in muscle but fails to prevent dystrophy. Nature Genet 8: 333–337, 1994
Greenberg DS, Sunada Y, Campbell KP, Yaffe D, Nudel U: Exogenous Dp71 restores the levels of dystrophin-associated proteins but does not alleviate muscle damage in mdx mice. Nature Genet 8: 340–344, 1994
Cox GA, Cole NM, Matsumura K, Phelps SF, Hauschka SD, Campbell KP, Faulkner JA, Chamberlain JS: Overexpression of dystrophin in transgenic mdx mice eliminated dystrophic symptoms without toxicity. Nature 364: 725–729, 1993
Tinsley JM, Potter AC, Phelps SR, Fisher R, Tricket JI, Davies KE: Amelioration of the dystrophic phenotype of mdx mice using a truncated utrophin transgene. Nature 384: 349–353, 1996
Brenman JE, Chao DS, Xia H, Aldape K, Bredt DS: Nitric oxide synthase complexed with dystrophin and absent from skeletal muscle sarcolemma in Duchenne muscular dystrophy. Cell 82: 743–752, 1995
Belkin AM, Burridge K: Expression and localization of the phosphoglucomutase-related cytoskeletal protein, aciculin, in skeletal muscle. J Cell Sci 107: 1993–2003, 1994
Belkin AM, Burridge K: Localization of utrophin and aciculin at sites of cell-matrix and cell-cell adhesion in cultured cells. Esp Cell Res 221: 132–140, 1995
Moiseeva EP, Belkin AM, Spurr NK, Koteliansky VE, Critchley DR: A novel dystrophin/utrophin-associated protein is an enzymatically inactive member of the phosphoglucomutase superfamily. Eur J Biochem 235: 103–113, 1996
Yang B, Jung D, Motto D, Meyer J, Koretzky G, Campbell KP: SH3 domain-mediated interaction of dystroglycan and Grb2 J Biol Chem 270: 11711–11714, 1995
Song KS, Li S, Okainoto T, Quilliam LA, Sargiacomo M, Lisanti MP: Copurification and direct interaction of Ras with Caveolin, an integral membrane protein of caveolae microdomains. J Biol Chem 271: 9690–9697, 1996
Song KS, Scherer PE, Tang Z, Okamoto T, Li S, Chafel M, Chu C, Kohtz DS, Lisanti MP: Expression of caveolin-3 in skeletal, cardiac, and smooth muscle cells. J Biol Chem 271: 15160–15165, 1996
Ozawa E. Muscular Dystrophy (in Japanese). Japanese Muscular Dystrophy Association, 1997
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ozawa, E., Hagiwara, Y. & Yoshida, M. Creatine kinase, cell membrane and Duchenne muscular dystrophy. Mol Cell Biochem 190, 143–151 (1999). https://doi.org/10.1023/A:1006974613418
Issue Date:
DOI: https://doi.org/10.1023/A:1006974613418