Abstract
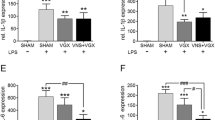
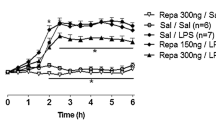
Immediately after bacterial endotoxin (LPS) enters the circulatory system there is increased production of free oxygen radicals by cells of the reticulo-endothelial system, followed by the release of cytokines considered as putative endogenous pyrogens. Fever originates by central nervous system activities, but neither exogenous nor endogenous pyrogens are able to cross the blood-brain barrier and the true signal which is transmitted to structures inside the blood-brain barrier is still unknown. To study the role of oxygen radicals in fever, we pretreated rats with methylene blue, an inhibitor of superoxide and hydroxyl radical production and investigated the febrile response to LPS in conscious rats by measuring malondialdehyde formation as an index of lipid peroxidation by oxygen radicals. Methylene blue lowered resting malondialdehyde levels to near detection level and significantly suppressed its rise which was regularly found following LPS in the untreated state. Pretreatment with methylene blue completely blocked the febrile response. Since fever is a central nervous system-mediated response these results indicate that the brain is able to sense oxidative stress and vicinal thiol groups of the redox-modulatory site of the N-methyl-D-aspartate (NMDA) subtype of glutamate receptor-channel complex could function as a possible receptive structure. To test this hypothesis we injected rabbits with the disulfide reducing agent dithiothreitol (DTT), known to penetrate the blood-brain barrier, and monitored its effect on normal and febrile body temperatures. DTT induced, independently of ambient temperature, within minutes and dose-dependently the full pattern of heat loss responses causing a fall of core temperature, indicative of a lowered thermoregulatory setpoint. Pretreatment with a bolus dose of 5 mg/kg DTT, followed by a continuous infusion of 5 mg/kg/h for 3 h completely prevented LPS-induced fever. A bolus dose of 20 mg/kg DTT, starting 30 min after LPS, immediately reversed the febrile cold defense pattern and lowered body temperature. We conclude that DTT reduces in the central nervous system oxidized vicinal thiol groups of NMDA receptors, thereby augmenting glutamate-induced nitric oxide synthase activation, and, thus, enhanced formation of NO, which, in turn, lowers the thermoregulatory setpoint. Reduction of other disulfide-containing molecules, especially oxidized glutathione and thiol-containing enzymes, by DTT by might additionally contribute to preventing fever.
Similar content being viewed by others
References
Sies H: Strategies of antioxidant defense. Eur J Biochem 215: 213–219, 1993
Moncada S, Herman AG, Vanhoutte PM: Endothelium-derived relaxing factor is identified as nitric oxide. Trends Pharmacol Sci 8: 365–368, 1987
Snyder SH: Nitric oxide: First in a new class of neurotransmitters? Science 257: 494–496, 1992
Lipton SA, Chol YB, Pan ZH, Lei SZ, Chen HSV, Sucher NJ, Loscalzo J, Singel DJ, Stamler JS: A redox-based mechanism for the neuroprotective and neurodestructive effects of nitric oxide and related nitroso-compounds. Nature 364: 626–6325,1993
Garthwaite J: Glutamate, nitric oxide and cell-cell signalling in the nervous system. TINS 14: 60–67, 1991
Radi R, Beckman JS, Bush KM, Freeman BA: Peroxynitrite oxidation of sulfhydryls. The cytotoxic potential of superoxide and nitric oxide. J Biol Chem 266: 4244–4250, 1991
Lafon-Cazal M, Pietri S, Culcasi M, Bockaert J: NMDA-dependent superoxide production and neurotoxicity. Nature 364: 535–537, 1993
Lei SZ, Pan ZH, Aggarwal SK, Chen HSV, Hartman J, Sucher NJ, Lipton SA: Effect of nitric oxide production on the redox modulatory site of the NMDA receptor-channel complex. Neuron 8: 1087–1099, 1992
McCall TB, Boughton-Smith N, Palmer RMJ, Whittle BJR, Moncada S: Synthesis of nitric oxide from L-arginine by neutrophils. Biochem J 261: 293–296, 1989
Rossi F: The O2-forming NADPH oxidase of the phagocytes: Nature, mechanisms of activation and function. Biochim Biophys Acta 853: 65–89, 1986
Segal AW: The electron transport chain of the microbicidal oxidase of phagocytic cells and its involvement in the molecular pathology of chronic granulomatous disease. J Clin Invest 83: 1785–1793, 1989
Blatteis CM: Cytokines as endogenous pyrogens. In: A Nowotny, JJ Spitzer, EJ Ziegler (eds). Cellular and Molecular Aspects of Endotoxin Reactions. Excerpta Medica, Amsterdam, 1990, pp 447–454
Blatteis CM, Quan N, Xin L, Ungar AL: Neuromodulation of acute phase response to interleukin 6 in guinea pigs. Brain Res Bull 25: 895–901, 1990
Salvemini D, Korbut R, Änggard E, Vane J: Immediate release of a nitric oxide-like factor from bovine aortic cells by Escherichia coli lipopolysaccharide. Proc Natl Acad Sci USA 87: 2593–2597, 1990
Bandaletova T, Brouet I, Bartsch H, Sugimura T, Esumi H, Ohsima H: Immunohistochemical localization of an inducible form of nitric oxide synthase in various organs of rats treated with Propionibacterium acnes and lipopolysaccharide. APMIS 101: 330–336, 1993
Stitt JT: Prostaglandin E1 fever induced in rabbits. J Physiol (Lond.) 232: 163–179, 1973
Coceani F: Prostaglandins and fever. In: P Mackowiak (ed). Facts and Controversies. Fever: Basic Mechanisms and Management. Raven Press, New York, 1991, pp 59–70
Sehic E, Székely M, Ungar AL, Oladehin A, Blatteis CM: Hypothalamic prostaglandin E2 during lipopolysaccharide-induced fever in guinea pigs. Brain Res Bull 39: 391–399, 1996
Riedel W: Antipyretic role of nitric oxide during endotoxin-induced fever in rabbits. Int J Tissue Reac 19: 171–178, 1997
Weihrauch D, Riedel W: Nitric oxide (NO) and oxygen radicals, but not prostaglandins, modulate fever. Ann NY Acad Sci 813: 373–3825, 1997
Mayer B, Brunner F, Schmidt K: Novel actions of methylene blue. Eur Heart J 14 (Suppl 1): 22–26, 1993
Oh S, McCaslin PP: Methylene blue induces a dose-dependent elevation in glutamate release but not cGMP levels in cultured neurons. Med Sci Res 24: 283–285, 1996
Cordis GA, Das DK, Riedel W: High-performance liquid chromatographic peak identification of 2,4–dinitrophenylhydrazine derivatives of lipid peroxidation aldehydes by photodiode array detection. J Chromatogr A 798: 117–123, 1998
Largillière C, Mélancon SB: Free malondialdehyde determination in human plasma by high-performance liquid chromatography. Anal Biochem 170: 123–126, 1988
Gupta RC: Nonrandom binding of the carcinogen N-hydroxy-2–acetylaminofluorene to repetitive sequences of rat liver DNA in vitro. Proc Natl Acad Sci USA 81: 6943–6947, 1984
Floyd RA, Watson JJ, Harris J, West M, Wong PK: Formation of 8–hydroxydeoxyguanosine, hydroxyl free radical adduct of DNA in granulocytes exposed to the tumor promoter, tetradeconylphorbolacetate. Biochem Biophys Res Comm 137: 841–846, 1986
Agarwal S, Draper HH: Isolation of a malonaldehyde-deoxyguanosine adduct from rat liver DNA. Free Rad Biol Med 13: 695–699, 1992
Cordis GA, Bagchi D, Riedel W, Stohs SJ, Das DK: Implication of DNA damage during reperfusion of ischemic myocardium. Ann NY Acad Sci 793: 427–430, 1997
Blatteis CM: Role of the OVLT in the febrile response to circulating pyrogens. In: A Ermisch, R Landgraf, HJ Rühle (eds). Circumventricular Organs and Brain Fluid Environment. Progr Brain Res, Vol. 91. Elsevier, Amsterdam, 1992, pp 409–412
Milton AS, Wendlandt S: Effects on body temperatures of prostaglandins of the A, E and F series on injection into the third ventricle of unanaesthetized cats and rabbits. J Physiol (London): 325–336, 1971
Milton AS: Prostaglandins in fever and the mode of action of antipyretic drugs. Springer-Verlag, Berlin, 1982, pp 257–303
Salvemini D, Misko TP, Masferrer JL, Seibert K, Currie MG, Needleman P: Nitric oxid activates cyclooxygenase enzymes. Proc Natl Acad Sci USA 90: 7240–7244, 1993
McCord JM, Fridovich I: The utility of superoxide dismutase in studying free radical reactions. J Biol Chem 242: 1374–1377, 1970
Kelner MJ, Bagnell R, Hale B, Alexander NM: Potential of methylene blue to block oxygen radical generation in reperfusion injury. Basic Life Sci 49: 895–898, 1988
Gruetter CA, Kadowitz PJ, Ignarro LJ: Methylene blue inhibits coronary arterial relaxation and guanylate cyclase activation by nitroglycerin, sodium nitrite and amyl nitrite. Can J Physiol Pharmacol 59: 150–156, 1981
Zeballos GA, Bernstein RD, Thompson CI, Forfia PR, Seyedi N, Shen W, Kaminski P, Wolin MS, Hintze TH: Pharmacodynamics of plasma nitrate/nitrite as an indication of nitric oxide formation in conscious dogs. Circulation 91: 2982–2988, 1995
Schmid HA, Riedel W, Simon E: Role of nitric oxide in temperature regulation. Progr Brain Res 115: 25–49, 1998
Sucher NJ, Lipton SA: Redox modulatory site of the NMDA receptorchannel complex: regulation by oxidized glutathione. J Neurosci Res 30: 582–591, 1991
Komori Y, Hyun J, Chiang K, Fukuto JM: The role of thiols in the apparent activation of rat brain nitric oxide synthase (NOS). J Biochem 117: 923–927, 1995
Meister A: Glutathione metabolism. Methods Enzymol 251: 3–7, 1995
Riedel W: Intravenous (iv) application of dithiothreitol (DTT) induces antipyresis or prevents bacterial endotoxin (LPS) fever in rabbits. Pflügers Arch Suppl to Vol 435: R162, 1998
Garthwaite J, Charles SL, Chess-Williams R: Endothelium-derived relaxing factor release on activation of NMDA receptors suggests role as intercellular messenger in the brain. Nature 336: 385–388, 1988
Stuehr DJ, Kwon NS, Nathan CF: FAD and GSH participate in macrophage synthesis of nitric oxide. Biochem Biophys Res Commun 168: 558–565, 1990
Miyamoto M, Murphy TH, Schnaar RL, Coyle JT: Antioxidants protect against glutamate-induced cytotoxicity in a neuronal cell line. J Pharmacol Exp Ther 250: 1132–1140, 1989
Atlante A, Gagliardi S, Minervini GM, Ciotti MT, Marra E, Calissano P: Glutamate neurotoxicity in rat cerebellar granular cells: a major role for xanthine oxidase in oxygen radical formation. J Neurochem 68: 2038–2045, 1997
Halliwell B, Gutteridge JMC: Role of free radicals and catalytic metal ions in human disease: An overview. Meth Enzymol 186: 1–85, 1990
Coyle JT, Puttfarcken P: Oxidative stress, glutamate, and neurodegenerative disorders. Science 262: 689–695, 1993
Stirpe F, Della Corte E: The regulation of rat liver xanthine oxidase. Conversion in vitro of the enzyme activity from dehydrogenase (type D) to oxidase (type O). J Biol Chem 244: 3855–3863, 1969
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Riedel, W., Maulik, G. Fever: An integrated response of the central nervous system to oxidative stress. Mol Cell Biochem 196, 125–132 (1999). https://doi.org/10.1023/A:1006936111474
Issue Date:
DOI: https://doi.org/10.1023/A:1006936111474