Abstract
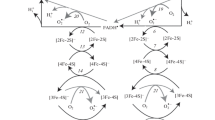
Oxidation of semiquinone by O2 in the Q cycle is known to be one of the sources of superoxide anion (O·2 -) in aerobic cells. In this paper, such a phenomenon was analyzed using the chemical kinetics model of electron transfer from succinate to cytochrome c, including coenzyme Q, the complex III non-heme iron protein FeSIII and cytochromes b1, bh and c1. Electron transfers from QH2 to FeSIII and cytochrome b1 were assumed to occur according to direct transfer mechanism (dynamic channelling) involving the formation of FeSred III -Q·- and Q·--cytochrome b1 complexes. For oxidation/reduction reactions involving cytochromes bh and b1, the dependence of the equilibrium and elementary rate constants on the membrane potential (ΔΨ) was taken into consideration. The rate of O·2 - generation was found to increase dramatically with increase in ΔΨ above the values found in State 3. On the other hand, the rate of cytochrome c reduction decreased sharply at the same values of the membrane potential. This explains experimental data that the O·2- generation at State 4 appears to be very much faster than at State 3. A mild uncoupling in State 4 can markedly decrease the superoxide generation due to a decrease in ΔΨ below the above mentioned critical level. ΔpH appears to be equally effective as ΔΨ in stimulation of superoxide production which depends, in fact, upon the Δμ- H + level.
Similar content being viewed by others
References
Kholodenko BN: Control of molecular transformations in multienzyme systems: quantitative theory of metabolic regulation. Mol Biologiya Russia 22: 1238–1256, 1988
Trumpower BL: The protonmotive Q cycle. J Biol Chem 265: 11409–11412, 1990
Skulachev VP: Membrane Bioenergetics. Springer, Berlin, 1988
Kholodenko BN, Westerhoff HV, Cascante M: Channelling and the concentration of bulk phase intermediates as cytosolic proteins get more concentrated. Biochem J 313: 921–926, 1996
Grivennikova VG, Vinogradov AD: Kinetics of ubiquinone reduction by the resolved succinate: Ubiquinone reductase. BBA 682: 491–495, 1982
Kotlyar AB, Vinogradov AD: Interaction of membrane-bound succinate dehydrogenase with substrate and competitive inhibitors. BBA 784: 24–34, 1984
Cramer WA, Knaff DB: Energy Transduction in Biological Membranes. Springer-Verlag, Berlin, 1991
Westerhoff HV, Van Dam K: Thermodynamics and Control of Biological Free-Energy Transduction. Elsevier, Amsterdam, 1987
Rich PR: Electron and proton transfers through quinones and cytochrome bc 1 complexes. BBA 768: 53–79, 1984
Hoffman P, Morgan TV, Der Vartanian DV: Respiratory chain characteristics mutants of Azotobacter vinelandii negative to tetramethyl-p-phenylenediamine oxidase. Eur J Biochem 100: 19–27, 1979
Boork J, Wennestrom H: The influence of membrane potentials on reaction rates. Control in free-energy transducing systems. BBA 767: 314–320, 1984
Reynolds IA, Johnson EA, Tanford C: Incorporation of membrane potential into theoretical analysis of electrogenic ion pumps. Proc Natl Acad Sci USA 82: 6869–6873, 1985
Kacser H, Burns JA: The control of the flux. In: DD Davies (ed). Rate Control of Biological Processes. Cambridge University Press, London, 1972, pp 65–104
Kacser H, Burns JA: Molecular democracy: Who shares the controls? Biochem Soc Trans 7: 1149–1160, 1979
Heinrich R, Rapoport TA: A linear steady-state treatment of enzymatic chains. General properties, control and effective strength. Eur J Biochem 42: 89–95, 1974
Heinrich R, Rapoport TA: Mathematical analysis of multienzyme systems: II. Steady state and transient control. BioSystems 7: 130–136, 1975
Kholodenko BN, Westerhoff HV: Control theory of one enzyme. Biochim Biophys Acta 1208: 294–305, 1994
Kholodenko BN, Westerhoff HV: Metabolic channelling and control of the flux. FEBS Lett 320: 71–74, 1993
Van Dam K, Van der Vlag J, Kholodenko BN, Westerhoff HV: The sum of the flux control coefficients of all enzymes on the flux through a group-transfer pathway can be as high as two. Eur J Biochem 212: 791–799, 1993
Kholodenko BN, Westerhoff HV: Control theory of group-transfer pathways. Biochim Biophys Acta 1229: 256–274, 1995
Kholodenko BN, Westerhoff HV: How to reveal various aspects of regulation in group-transfer pathways. Biochim Biophys Acta 1229: 275–289, 1995
Brand MD, Vallis BPS, Kesseler A: The sum of flux control coefficients in the electron-transport chain of mitochondria. Eur J Biochem 226: 819–829, 1994
Kholodenko BN, Westerhoff HV: The macro-versus microworld of biochemical regulation and control. Trends Biochem Sci 20: 52–54, 1995
Boveris A, Chance B: The mitochondrial generation and hydrogen peroxide. General properties and effect of hyperbaric oxygen. Biochem J 134: 707–716, 1973
Skulachev VP: Lowering of intracellular O2 concentration as a special function of respiratory systems of the cells. Biochemistry (Moscow) 59: 1910–1912, 1994 (Russian)
Skulachev VP: Non-phosphorylating respiration as a mechanism to minimize formation of reactive oxygen species in the cell. Mol Biologiya 29: 709–715, 1995 (Russian)
Skulachev VP: Role of uncoupled and non-coupled oxidations maintenance of safely low levels of oxygen and its one-electron reductants. Quart Rev Biophys 29: 169–202, 1996
Green DE, Wharton DC: Stoichiometry of the fixed oxidation-reduction components of the electron transfer chain of beef heart mitochondria. Biochem Z 336: 335–346, 1963
Srere PA: TIBS 6: 4–6, 1981
Jones DP: Intracellular diffusion gradients of O2 and ATP. Am J Physiol 250: C663–C675, 1986
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Demin, O., Kholodenko, B. & Skulachev, V. A model of O·2 - generation in the complex III of the electron transport chain. Mol Cell Biochem 184, 21–33 (1998). https://doi.org/10.1023/A:1006849920918
Issue Date:
DOI: https://doi.org/10.1023/A:1006849920918