Abstract
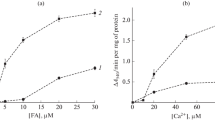
Hexadecylphosphocholine (HePC) is known as antitumor agent but the mechanism has not yet been understood. In rat liver mitochondria its effect on phospholipid transformation has been studied by quantitative HPTLC and phosphorus determination. From the results it can be concluded that HePC influences the activities of phospholipase A2, phospholipase C, phospholipase D, and lysophospholipase A. The phospholipid transformation as well as the influence of HePC are affected by exogenous calcium ions. In the presence of calcium HePC has been found to inhibit enzyme activities, whereas in the absence of exogenous calcium ions enzymatic phospholipid transformations are activated or inhibited depending on HePC concentrations.
Similar content being viewed by others
References
Hilgard P, Stekar J, Voegeli R, Engel J, Schumacher W, Eibl H, Unger C, Berger MR: Characterization of antitumor activity of hexadecylphosphocholine (D 18506). Eur J Cancer Clin Oncol 24: 1457–1461, 1988
Unger C, Damonz W, Fleer EA, Kim DJ, Breiser A, Hilgard P, Engel J, Nagel G, Eibl H: Hexadecylphosphocholine, a new ether lipid analogue. Studies on the antineoplastic activity in vitro and in vivo. Acta Oncol 28: 213–217, 1989
Hilgard P, Klenner T, Stekar J, Unger C: Alkylphosphocholines: A new class of membrane-active anticancer agents. Cancer Chemother Pharmacol 32: 90–95, 1993
Berkovic D, Grunwald U, Menzel W, Unger C, Hiddemann W, Fleer EAM: Effects of hexadecylphosphocholine on membrane phospholipid metabolism in human tumour cells. Eur J Cancer 31A: 2080–2085, 1995
Haase R, Wieder T, Geilen CC, Reutter W: The phospholipid analogue hexadecylphosphocholine inhibits phosphatidylcholine biosynthesis in Madin-Darby canine kidney cells. FEBS Lett 288: 129–132, 1991
Zheng B, Oishi K, Shoji M, Eibl H, Berdel WE, Hajdu J, Vogler WR, Kuo JF: Inhibition of protein kinase C, (sodium-potassium)-activated adenosine-triphosphatase, and sodium pump by synthetic phospholipid analogues. Cancer Res 50: 3025–3031, 1990
Maly K, Ñberall F, Schubert C, Kindler E, Stekar J, Brachwitz H, Grunicke HH: Interference of new alkylphospholipid analogues with mitogenic signal transduction. Anticancer Drug Design 10: 411–425, 1995
Geilen CC, Haase R, Buchner K, Wieder T, Hucho F, Reutter W: The phospholipid analogue, hexadecylphosphocholine, inhibits protein kinase C in vitro and antagonises phorbol ester-stimulated cell proliferation. Eur J Cancer 27: 1650–1653, 1991
Shoji M, Raynor RL, Fleer EAM, Eibl H, Vogler WR, Kuo JF: Effects of hexadecylphosphocholine on protein kinase C and TPA-induced differentiation of HL60 cells. Lipids 26: 145–149, 1991
Ñberall F, Oberhuber H, Maly K, Zaknun J, Demuth L, Grunicke HH: Hexadecylphosphocholine inhibits inositol phosphate formation and protein kinase C activity. Cancer Res 51: 807–812, 1991
Pawelczyk T, Lowenstein JM: Inhibition of phospholipase Cδ by hexadecylphosphorylcholine and lysophospholipids with antitumor activity. Biochem Pharmacol 45: 493–497, 1993
Wieder T, Zhang ZC, Geilen CC, Orfanos CE, Giuliano AE, Cabot MC: The antitumor phospholipid anolog, hexadecylphosphocholine, activates cellular phospholipase D. Cancer Lett 100: 71–79, 1996
Dittrich N, Nosser G, Kutscher B, Ulbrich-Hofmann R: Alkylphosphate esters as inhibitors of phospholipase D. J Enzyme Inhib 11: 67–75, 1996
Rakhimov MM, Gorbataya ON, Almatov KT: Properties of phos pholipase D of rat liver mitochondria. Biokhimiya 54: 1066–1074, 1989
Rakhimov MM, Almatov KT, Mirtalipov DT, Gorbataya ON, Kasimova GM: Change in the phospholipid composition of mitochondria during thermal incubation. Biokhimiya 54: 948–955, 1989
Steinbrecht I, Kunz W: Anwendung der ‘Cycling’-Technik zur stich-probenartigen quantitativen Bestimmung des Reduktionsgrades des NAD-und NADP-Systems von Rattenlebermitochondrien bei kontinuierlich registrierenden Messungen. Acta Biol Med Germ 25: 731–747, 1970
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ: Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275, 1959
Bligh EG, Dyer WJ: A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37: 911–917, 1959
Kates M: Techniques of Lipidology. Amsterdam, North-Holland, 1972
Vaskovsky VE, Kostetsky EG, Vasendin IM: A universal reagent for phospholipid analysis. J Chromatogr 114: 129–141, 1975
Halestrap AP, Armston AE, Quinlan PT, Whipps DE: Hormonal control of liver metabolism. Horm Cell Regul 7: 185–200, 1983
Wiswedel I, Barnstorf U, Augustin W, Holmuhamedov E, Medvedev B, Evtodienko Y: Involvement of periodic deacylation-acylation cycles of mitochondrial phospholipids during Sr2+-induced oscillatory ion transport in rat liver mitochondria. Biochim Biophys Acta 688: 597–604, 1982
Daum G: Lipids in mitochondria. Biochim Biophys Acta 822: 1–42, 1985
Gelb MH, Jain MK, Hanel AM, Berg OG: Interfacial enzymology of glycerolipid hydrolases: Lessons from secreted phospholipase A2. Ann Rev Biochem 64: 653–688, 1995
Vernon LP, Bell JD: Membrane structure, toxins and phospholipase A2 activity. Pharm Ther 54: 269–295, 1992
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vagina, O., Gellerich, F.N. & Ulbrich-Hofmann, R. The effect of hexadecylphosphocholine on the degradation of mitochondrial phospholipids. Mol Cell Biochem 183, 169–173 (1998). https://doi.org/10.1023/A:1006826122602
Issue Date:
DOI: https://doi.org/10.1023/A:1006826122602