Abstract
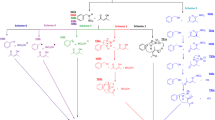
The mechanism of the reaction of the N-fluoro reagent F‐TEDA‐BF4 with dimethylenebicyclo[3.3.1]nonane was investigated by the semiempirical PM3 SCF MO method using the Cramer–Truhlar SM5.42R solvation model. It was shown that the mechanism of addition of the fluorine corresponds to classical S N 2 nucleophilic substitution involving single-electron transfer synchronous with the cleavage–formation of C—F and N—F bonds.
Similar content being viewed by others
REFERENCES
G. G. Frumkin and A. A. Fainzil'berg, Usp. Khim., 68, No.8, 752 (1999).
S. Stavber, T. Sotler-Pecan, and M. Zupan, Bull. Chem. Soc. Jpn, 69, 169-175 (1996).
J. J. P. Stewart, J. Comput. Chem., 10, 209 (1989).
K. Fukui, J. Phys. Chem., 74, 4161 (1970).
C. J. Cramer and D. J. Truhlar, Chem. Rev., 99, 2161-2200 (1999).
M. W. Shmidt, K. K. Baldridge, J. A. Boatz, et al., J. Comput. Chem., 14, 1347-1363 (1993).
G. D. Hawkins, D. J. Giesen, G. C. Lynch, et al., AMSOL-Version 6.5.2, University of Minnesota, Minneapolis (1997).
M. J. S. Dewar, E. G. Zoebisch, E. F. Healy, and J. J. P. Stewart, J. Am. Chem. Soc., 107, 3902 (1985).
S. Shaik, J. Am. Chem. Soc., 106, 1227-1232 (1984).
I. D. Gridnev and I. A. Gridneva, Usp. Khim., 64, No.11, 1091-1105 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tishchenko, O.V., Serguchev, Y.A., Lur'e, L.F. et al. Quantum-Chemical Investigation of the Mechanism of Transfer of Fluorine in the Reaction of F‐TEDA‐BF4 with Dimethylenebicyclo[3.3.1]nonane. Theoretical and Experimental Chemistry 36, 254–259 (2000). https://doi.org/10.1023/A:1005218311010
Issue Date:
DOI: https://doi.org/10.1023/A:1005218311010