Abstract
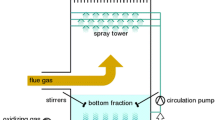
Selective removal of SO2, H2S and NOx from gas streams (air or towngas) was investigated using aqueous solutions of 12-molybdophosphoric acid and its reduced molybdenum blue species. The experimental results showed that H2S and SO2 in the gas streams can be removed quantitatively by yellow solution of 12-molybdophosphoric acid. The H2S was oxidized into recoverable elemental sulfur and SO2 into H2SO4, while the yellow solution was reduced into blue species (reduced molybdenum blue). The obtained reduced molybdenum blue solution was then used for the quantitative removal of NOx, which was reduced into N2 and the blue solution was then oxidized back into yellow species. It is possible that the regenration of scrubbing solution is not required and no chemical is consumed during absorption, when NOx, SO2 and/or H2S coexisted in wastegases and the molar ratios of NO2/(SO2 + H2S) is just equal to 1/2, although, in most cases, the ratio in real practical wastegases is not always equal to the value. The absorption mechanism was studied in detail. It was found that only one molybdenum atom in 12-molybdophosphoric acid was reduced from Mo(VI) to Mo(V) by H2S or H2SO3 and the Mo(V) was oxidized back into Mo(VI) in the case of Nox absorption. 12-molybdophsophoric acid used in this work is inexpensive, commercially available, easily regeneratable, relatively nontoxic, and gives no secondary pollutant in the process.
Similar content being viewed by others
References
Bacon, J. C.: 1967, Molybdenum Compounds, in: Kirk Othmer (eds.), Encyclopedia of Chemical Technology (2nd ed.), Interscience Publishers, J.Wiley and Sons, Inc., NewYork, 13, pp. 634–658.
Chang, S. G. and Lee, G. C.: 1992, Environmental Progress 11(1), 66.
Chen, S. L., Lyon, R. K. and Seeker, W. R.: 1991, Environmental Progress 10(3), 182.
Gullett, B. K., Bruce, K. R., Hansen, W. F. and Hofmann, J. E.: 1992, Environmental Progress 11(2), 155.
Habashi, F.: 1993, A Textbook of Hydrometallurgy, Metallurgie Extractive, Quebec, Canada, pp. 310–356.
Hedges, S. W. and Yeh, J. T.: 1992, Environmental Progress 11(2), 98.
Helfrritch, D. Rortz, S., Beittel, R., Bergman, P. and Toole-O'Nell, B.: 1992, Environmental Progress 11(1), 7–10.
Mizuta, S., Kondo, W. and Fujji, K.: 1991, Ind. Eng. Chem. Res. 30, 1601.
Neumann, D. W.: 1984, AIChE Journal 30(1), 62.
Perimutter, H. D., Hulhong, Ao, and Henry Shaw: 1992, Environmental Sciences and Technology 27(1), 128.
Pope, M.T.: 1983, Heteropoly and Isopoly Oxometalates, SpringerVerlag, pp. 1–150.
Shen, D., Liu, D. K. and Shih Ger Chang: 1992, China Environmental Science (in Chinese with English abstract), 12(3), 193.
Steverson, E. M.: 1991, Environmental Science and Technology 25(11), 1808.
Zhao, Y. C.: 1989, Removal of Phosphorus, Arsenic and Silica as Impurities from Tungstate or Molybdate Solutions by Synergistic Extraction with Primary Amine and Tributylphosphate as Solvents, Ph. D. Thesis, Institute of ChemicalMetallurgy, Chinese Academy of Sciences, Beijing, China (in Chinese with English summary).
Zhao, Y. C., Chen, S.W., Xu, D. M. and Lin, M.: 1995, Journal of Environmental Science 7(4), 468.
Zhao, Y. C., Zouboulis, A. I. and Matis, K. A.: 1996, Separation Science and Technology 31(6), 769.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhao, Y., Zhu, Q. & Gu, G. Combined Removal of SO2, H2S AND NOx from Gas Streams by Chemical Absorption with Aqueous Solution of 12-Molybdophosphoric Acid and its Reduced Species. Water, Air, & Soil Pollution 102, 157–176 (1998). https://doi.org/10.1023/A:1004956032764
Issue Date:
DOI: https://doi.org/10.1023/A:1004956032764