Abstract
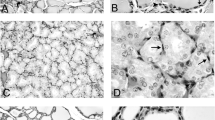
The effects of intracerebroventricularly administered somatostatin (SRIH-14 or -28) on growth and function of pituitary thyrotropes (TSH-cells) were examined in adult male Wistar rats. The animals were implanted with an intracerebroventricular cannula and after a recovery period, administered three 1 μg doses of SRIH-14 or -28 dissolved in 5 μl saline every second day. Controls were treated in the same way with the same volume of saline only. TSH-producing cells were studied using the peroxidase–antiperoxidase immunohistochemical procedure. Blood samples were collected for hormone (TSH) analyses 5 days after the last injection. Both SRIH-treatments significantly decreased (p < 0.05) all morphometric parameters obtained for TSH-cells in comparison with controls. The volume of TSH-cells decreased by 27%, nuclei by 44% and volume density by 33% in animals treated with SRIH-14. In animals treated with SRIH-28, these parameters were also significantly decreased (p < 0.05) (22%, 31%, and 25% respectively) compared to control rats. Serum concentrations of TSH were significantly decreased (p < 0.05) by 15% in SRIH-14- and by 12% in SRIH-28-treated animals in comparison with the controls. These observations suggest that centrally administered SRIH-14 or -28 is specifically involved in the control of growth and secretory activity of TSH cells.
Similar content being viewed by others
References
Ain KB, Taylor KD, Tofiq S, Venkataraman G (1997) Somatostatin receptor subtype expression in human thyroid and thyroid carcinoma cell lines. J Clin Endocrinol Metab 82: 1857–1862.
Brazeau P, Vale W, Burgus R, Ling N, Butcher M, Rivier J, Guillemin R (1973) Hypothalamic polypeptide that inhibits the secretion of immunoreactive pituitary growth hormones. Science 179: 77–79.
Critchlow V, Abe K, Urman S, Vale W (1981) Effects of lesions in the periventricular nucleus of the preoptic-anterior hypothalamus on growth hormone and thyrotropin secretion and brain somatostatin. Brain Res 222: 267–276.
De Rosa G, Corsello SM, Della Casa S, De Rosa E, Raimondo S (1983) Effect of somatostatin on the pituitary-thyroid axis. Ann Endocrinol (Paris) 44: 355–360.
Epelbaum J, Enjalbert A, Krantic S, Musset P, Bertrand R, Rasolonjanahary C, Shu C, Kordon C (1987) Somatostatin receptors on pituitary somatotrophs, thyrotrophs, and lactotrophs: Pharmacological evidence for loose coupling to adenylate cyclase. Endocrinology 121: 2177–2185.
Gillies G (1997) Somatostatin: the neuroendocrine story. Trends Pharmacol Sci 18: 87–95.
James RA, Sarapura VD, Bruns C, Raulf F, Dowding JM, Gordon DF, Wood WM, Ridgway CE (1997) Thyroid hormone-induced expression of specific somatostatin receptor subtypes correlates with involution of the TtT-97 murine thyrotrope tumor. Endocrinology 138: 719–724.
Krulich L, Dhariwal S, McCann S (1968) Stimulatory and inhibitory effects of purified hypothalamic extracts on growth hormone release from rat pituitary in vitro. Endocrinology 83: 783–790.
Lam LS, Wong RLC (1999) Thyroid hormones regulate the expression of somatostatin receptor subtypes in the rat pituitary. Neuroendocrinology 69: 460–464.
Milošević V, Brkić B, Velkovski S, Sekulić M, Lovren M, Starčević V, Severs W (1998) Morphometric and functional changes of the rat pituitary somatotropes and lactotropes after central administration of somatostatin. Pharmacology 57: 27–34.
Oliver C, Giraud P, Lissitzky JC, Cote J, Boudouresque F, Gillioz P, Conte-Devolx B (1982) Influence of endogenous somatostatin on growth hormone and thyrotropin secretion in neonatal rats. Endocrinology 110: 1018–1022.
Reichlin S (1983) Somatostatin. N Engl J Med 309: 1495–1501.
Reisine T, Bell IG (1995) Molecular biology of somatostatin receptors. Endocrine Rev 16: 427–442.
Reubi CJ (1997) Relevance of somatostatin receptors and other peptide receptors in pathology. Endocrine Path 8: 11–20.
Richardson I, Schonbrunn A (1981) Inhibition of adrenocorticotropin secretion by somatostatin in pituitary cells in culture. Endocrinology 108: 281–290.
Roussel JP, Grazzini E, Astier H (1997) Somatostatin blocks the potentiation of TRH-induced TSH secretion from perifused pituitary fragments and the change in intracellular calcium concentrations from dispersed pituitary cells elicited by prepro-TRH (PS4) or by tri-iodothyronine. J Mol Endocrinol 19: 87–97.
Sekulić M, Lovren M, Milošević V (2000) Somatostatin acts by inhibiting pituitary TSH cells in female rats. Acta Veterinaria (Beograd) 50: 3–12.
Senaris R, Lago F, Dieguez C (1996) Gonadal regulation of somatostain receptor 1, 2 and 3 mRNA levels in the rat anterior pituitary. Mol Brain Res 38: 171–175.
Srikant CB, Patel YC (1985) Somatostatin receptors. Adv Exp Med Biol 188: 291–304.
Starčević VP, Morrow BA, Farner LA, Keil LC, Severs WB (1988) Long-term recording of cerebrospinal fluid pressure in freely behaving rats. Brain Res 462: 112–117.
Sternberger LA, Hardy PHJ, Cuculius JJ, Meyer HG (1970) The unlabelled antibody enzyme method of immunohistochemistry. Preparation and properties of soluble antigen-antibody complex (horseradish peroxidase-antihorseradish peroxidase) and its use in identification of spirochetes. J Histochem Cytochem 18: 315–333.
Theodoropoulos TJ (1985) Somatostatin is a regulator of thyrotropin secretion in the perinatal rat. Endocrinology 117: 1683–1686.
Vale W, Rivier C, Brazeau P, Guillemin R (1974) Effects of somatostatin on the secretion of thyrotrophin and prolactin. Endocrinology 95: 968–977.
Weibel ER (1979) Stereological Methods. Practical Methods for Biological Morphometry. London: Academic Press: pp. 1–415.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Milošević, V., Sekulić, M., Brkić, B. et al. Effect of Centrally Administered Somatostatin on Pituitary Thyrotropes in Male Rats. Histochem J 32, 565–569 (2000). https://doi.org/10.1023/A:1004158412915
Issue Date:
DOI: https://doi.org/10.1023/A:1004158412915