Abstract
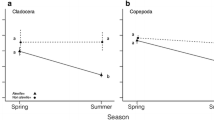
We determined clearance rates and ingestion rates of Daphnia galeata on bacteria and phytoplankton in order to test if bacteria are an important alternative food resource for daphnids during periods of low phytoplankton biomass in the biomanipulated Bautzen reservoir (Germany). D. galeata was able to feed on bacteria with the same efficiency as on algae during most of the time. In spite of similar clearance rates, bacteria ingestion was usually lower than phytoplankton ingestion due to lower bacterial biomass. Only at low biomass of algae in late fall and during the clear water phase, bacteria contributed up to 87% and 42%, respectively, to total carbon ingestion of D. galeata. However, even a short period of relatively high bacteria grazing by daphnids may be important for biomanipulation. Bacteria might bridge over periods of food limitation of daphnids thus promoting the maintenance of high Daphnia biomass. Therefore, ingestion of bacteria by daphnids is thought to stabilize biomanipulation and may hold a key position in the food web of biomanipulated lakes.
Similar content being viewed by others
References
Baines, S. B. & M. L. Pace, 1991. The production of dissolved organic matter by phytoplankton and its importance to bacteria: Pattern across marine and freshwater systems. Limnol. Oceanogr. 36: 1078–1090.
Benndorf, J., 1987. Food webmanipulation without nutrient control: a useful stategy in lake restoration? Schweiz. Z. Hydrol. 49: 234–248.
Benndorf, J., 1990. Conditions for effective biomanipulation: Conclusions derived from whole-lake experiments in Europe. Hydrobiologia 200/201 (Dev. Hydrobiol. 61): 187–203.
Benndorf, J., 1995. Possibilities and limits for controlling eutrophication by biomanipulation. Int. Rev. ges. Hydrobiol. 80: 519–534.
Benndorf, J., H. Schultz, A. Benndorf, R. Unger, E. Penz, H. Kneschke, K. Kossatz, R. Dumke, U. Hornig, R. Kruspe & S. Reichel, 1988. Food-web manipulation by enhancement of piscivorous fish stocks: long-term effects in the hypertrophic Bautzen Reservoir. Limnologica 19: 97–110.
Børsheim, K. Y. & S. Andersen, 1987. Grazing and food size selection by cructacean zooplankton compared to production of bacteria and phytoplankton in a shallow Norwegian mountain lake. J. Plankton Res. 9: 367–379.
Børsheim, K. Y. & Y. Olsen, 1984. Grazing activities by Daphnia pulex on natural populations of bacteria and algae. Verh. int. Ver. Limnol. 22: 644–648.
Bottrell, H. H., A. Duncan, Z. M. Gliwicz, E. Grygierek, A. Herzig, A. Hilbricht-Ilkowska, H. Kurasawa, P. Larsson& A. Weglenska, 1976. A review of some problems in zooplankton production studies. Norw. J. Zool. 24: 419–456.
Bratbak, G., 1985. Bacterial biovolume and biomass estimations. Appl. Environ. Microbiol. 49: 1488–1493.
Bratbak, G. & I. Dundas, 1984. Bacterial dry matter and biomass estimations. Appl. Environ. Microbiol. 48: 755–757.
Brendelberger, H., 1991. Filter mesh size of cladocerans predicts retention efficiency for bacteria. Limnol. Oceanogr. 36: 884–894.
Burns, C. W., 1968. The relationship between body size of filter feeding cladocera and the maximum size of particle ingested. Limnol. Oceanogr. 13: 675–678.
Christoffersen, K., B. Riemann, L. R. Hansen, A. Klysner & H. B. Sørensen, 1990. Qualitative importance of the microbiol loop and plankton community structure in a eutrophic lake during a bloom of Cyanobacteria. Microb. Ecol. 20: 253–272.
Cole, J. J., S. Findlay & M. L. Pace, 1988. Bacterial production in fresh and saltwater: A cross system overview. Mar. Ecol. Prog. Ser. 43: 1–10.
DeMelo, R., R. France & D. McQueen, 1992. Biomanipulation: Hit or myth? Limnol. Oceanogr. 37: 192–207.
Derenbach, J. B. & P. L. LeB. Williams, 1974. Autotrophic and bacterial production: fractionation of plankton populations by differential filtration of samples from the English Channel. Mar. Biol. 25: 263–269.
Gliwicz, Z. M., 1985. Predation or food limitation: an ultimate reason for extinction of planktonic cladoceran species. Arch. Hydrobiol. Beih. Ergebn. Limnol. 21: 419–430.
Güde, H., 1988. Direct and indirect influences of crustacean zooplankton on bacterioplankton in Lake Constance. Hydrobiologia 159: 63–73.
Haney, J. F. & D. J. Hall, 1973. Sugar-coated Daphnia: A preservation technique for cladocera. Limnol. Oceanogr. 18: 331–333.
Happey-Wood, C. M. & A. H. Lund, 1994. Production of new organic carbon and its distribution between autotrophic picoplankton, bacteria, extracellular organic carbon and phytoplankton in an upland lake. Freshwat. Biol. 31: 1–18.
Horn, W., 1991. The influence of biomass and structure of the crustacean plankton on the water transparency in the Saidenbach storage reservoir. Hydrobiologia 225: 115–120.
Jeppesen, E., J. P. Jensen, P. Kristensen, M. Søndergaard, E. Mortensen, O. Sortkaer & K. Olrik, 1990. Fish manipulation as a lake restoration tool in shallow, eutrophic, temperate lakes 2: threshold levels, long-term stability and conclusions. Hydrobiologia 200/201 (Dev. Hydrobiol. 61): 219–227.
Jeppesen, E., M. Søndergaard, J. P. Jensen, E. Mortensen & O. Sortkaer, 1996. Fish-induced changes in zooplankton grazing on phytoplankton and bacterioplankton: a long-term study in shallow hypertrophic Lake Søbygaard. J. Plankton Res. 18: 1605–1625.
Jørgensen, N. O. G. & S. Bosselmann, 1988. Concentration of free amino acids and their bacterial assimilation rates in vertical pro-files of two Danish lakes: Relation to diel changes in zooplankton grazing activity. Arch. Hydrobiol. Beih. Ergebn. Limnol. 31: 289–300.
Jürgens, K., 1994. Impact of Daphnia on planktonic microbial food webs – A review. Mar. Microb. Food Webs 8: 295–324.
Jürgens, K., H. Arndt & H. Zimmermann, 1997. Impact of metazoan and protozoan grazers on bacterial biomass distribution in mesocosm experiments. Aquat. Microb. Ecol. 12: 131–138.
Jürgens, K., J. M. Gasol, R. Massana & C. Pedros-Alio, 1994. Control of heterotrophic bacteria and protozoans by Daphnia pulex in the epilimnion of Lake Ciso. Arch. Hydrobiol. 131: 55–78.
Jürgens, K. & G. Stolpe, 1995. Seasonal dynamics of crustacean zooplankton, heterotrophic nanoflagellates and bacteria in a shallow, eutrophic lake. Freshwat. Biol. 33: 27–38.
Kamjunke, N., T. Deppe & J. Benndorf, 1998. Bacterial production under varying trophic conditions and its importance as food source for daphnids in a biomanipulated reservoir. Int. Rev. Hydrobiol. 83: 413–420.
Kamjunke, N., R. F. Herbst, A. Wagner & J. Benndorf, 1996. Size distribution of primary production in a whole-lake biomanipulation experiment under hypertrophic conditions. Arch. Hydrobiol. 138: 259–271.
Kankaala, P., 1988. The relative importance of algae and bacteria as food for Daphnia longispina (Cladocera) in a polyhumic lake. Freshwat. Biol. 19: 285–296.
Kirchman, D. L. & M. P. Hoch, 1988. Bacterial production in the Delaware Bay estuary estimated from thymidine and leucine incorporation rates. Mar. Ecol. Prog. Ser. 45: 169–178.
Köthe, A. & J. Benndorf, 1994. Top-down impact of Daphnia galeata on pelagic heterotrophic flagellates in a whole-lake biomanipulation experiment. Mar. Microb. Food Webs 8: 325–335.
Lampert, W., 1977. Studies on the carbon balance of Daphnia pulex de Geer as related to environmental conditions. II. The dependence of carbon assimilation on animal size, temperature, food concentration and diet species. Arch. Hydrobiol./Suppl. 48: 310–335.
Lampert, W., W. Fleckner, H. Rai & B. E. Taylor, 1986. Phytoplankton control by grazing zooplankton: A study on the spring clear-water phase. Limnol. Oceanogr. 31: 478–490.
Lyche, A., T. Andersen, K. Christoffersen, D. O. Hessen, P. H. B. Hansen & A. Klysner, 1996. Mesocosm tracer studies. 2. The fate of primary production and the role of consumers in the pelagic carbon cycle of a mesotrophic lake. Limnol. Oceanogr. 41: 475–487.
McCauley, E. & F. Briand, 1979. Zooplankton grazing and phytoplankton species richness, field tests of the predation hypothesis. Limnol. Oceanogr. 24: 243–252.
McQueen, D. J., 1998. Freshwater food web manipulation: A powerful tool for water quality improvement, but maintenance is required. Lakes and Reservoirs: Research and Management 3: 83–94.
Meffert, M.-E. & J. Overbeck, 1985. Dynamics of chlorophyll and photosynthesis in natural phytoplankton associations. II. Primary productivity, quantum yields and photosynthetic rates in small Northgerman lakes. Arch. Hydrobiol. 104: 363–385.
Mehner, T., S. Hülsmann, S. Worischka, M. Plewa & J. Benndorf, 1998. Is the midsummer decline of Daphnia really induced by age-0 fish predation? Comparison of fish consumption and Daphnia mortality and life history parameters in a biomanipulated reservoir. J. Plankton Res. 20: 1797–1811.
Pace, M. L. & J. J. Cole, 1994. Primary and bacterial production in lakes: are they coupled over depth? J. Plankton Res. 16: 661–672.
Pace, M. L., G. B. McManus & S. E. G. Findlay, 1990. Planktonic community structure determines the fate of bacterial production in a temperate lake. Limnol. Oceanogr. 35: 795–808.
Pedros-Alio, C. & T. D. Brock, 1983. The impact of zooplankton feeding on the epilimnetic bacteria of a eutrophic lake. Freshwat. Biol. 13: 227–239.
Rai, H., 1984. Magnitude of heterotrophic metabolism of photosynthetically fixed dissolved organic carbon (PDOC) in Schöhsee, West Germany. Arch. Hydrobiol. 102: 91–103.
Reche, I., P. Carrillo & L. Cruz-Pizarro, 1997. Influence of metazooplankton on interactions of bacteria and phytoplankton in an oligotrophic lake. J. Plankton Res. 19: 631–646.
Reynolds, C. S., 1994. The ecological basis for the successful biomanipulation of aquatic communities. Arch. Hydrobiol. 130: 1–33.
Riemann, B., 1985. Potential importance of fish predation and zooplankton grazing on natural populations of freshwater bacteria. Appl. envir. Microbiol. 50: 187–193.
Riemann, B. & S. Bosselmann, 1984. Daphnia grazing on natural populations of bacteria. Verh. int. Verein. Limnol. 22: 795–799.
Riemann, B., N. O. G. Jørgensen, W. Lampert & F. Azam, 1986. Zooplankton induced changes in dissolved free amino acids and in production rates of freshwater bacteria. Microb. Ecol. 12: 247–258.
Riemann, B. & M. Søndergaard, 1984. Measurement of diel rates of bacterial secondary production in aquatic environments. Appl. envir. Microbiol. 47: 632–638.
Sanders, R. W., C. E. Williamson, P. L. Stutzman, R. E. Moeller, C. E. Goulden & R. Aoki-Goldsmith, 1996. Reproductive success of 'herbivorous' zooplankton fed algal and nonalgal food recources. Limnol. Oceanogr. 41: 1295–1305.
Shapiro, J., V. Lamarra & M. Lynch., 1975. Biomanipulation: an ecosystem approach to lake restoration. In: Bresonik, P. L. & J. L. Fox (eds), Water Quality Management Through Biological Control. Report No. ENV–07–75–1. University of Florida, Gainesville: 85–96.
Simon, M. & F. Azam, 1989. Protein content and protein synthesis rates of planktonic bacteria. Mar. Ecol. Prog. Ser. 51: 201–213.
Sommaruga, R., 1995. Microbial and classical food webs: A visit to a hypertrophic lake. FEMS Microb. Ecol. 17: 257–270.
Sommer, U., Z. M. Gliwicz, W. Lampert & A. Duncan, 1986. The PEG-model of seasonal succession of planktonic events in fresh waters. Arch. Hydrobiol. 106: 433–471.
Urabe, J. & Y. Watanabe, 1991. Effect of food conditions on the bacterial feeding of Daphnia galeata. Hydrobiologia 225 (Dev. Hydrobiol. 71): 121–128.
Utermöhl, H., 1958. Zur Vervollkommnung der quantitativen Phytoplankton-Methodik. Mitt. int. Ver. Limnol. 9: 1–38.
Vollenweider, R. A. (ed.), 1969. A manual on methods for measuring primary production in aquatic environments. IBP Handbook No. 12, Blackwell Scientific Publications, Oxford: 213 pp.
Wylie, J. L. & D. J. Currie, 1991. The relative importance of bacteria and algae as food sources for crustacean zooplankton. Limnol. Oceanogr. 36: 708–728.
Zankai, N. P., 1983. Ingestion rates of some Daphnia species in a shallow Lake (Lake Balaton, Hungary). Int. Rev. ges. Hydrobiol. 68: 227–237.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kamjunke, N., Benndorf, A., Wilbert, C. et al. Bacteria ingestion by Daphnia galeata in a biomanipulated reservoir: a mechanism stabilizing biomanipulation?. Hydrobiologia 403, 109–121 (1999). https://doi.org/10.1023/A:1003722318598
Issue Date:
DOI: https://doi.org/10.1023/A:1003722318598