Abstract
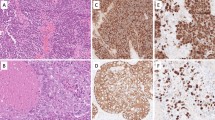
Our previous reports have demonstrated frequent and strong expression of glycogen phosphorylase (EC 2.4.1.1) activity mainly in the cytoplasm of gastric carcinoma. Although previous studies have suggested the phosphorylase glyco-syltransferase system to be in the nucleus from enzyme histochemical analyses, intranuclear localization of the phosphorylase has not been fully established. The aims of the present study are to investigate the nuclear localization of glycogen phosphorylase and to identify the isoform of phosphorylase in the nucleus of gastrointestinal carcinoma. The activity of glycogen phosphorylase in carcinoma cells corresponding to the nucleus was demonstrated using enzyme cytochemical analysis. The phosphorylase activity coincided with localization revealed by immunocytochemistry using affinity-purified specific anti-human brain-type glycogen phosphorylase antibody. The isoform expressed in the nuclei of carcinoma cells was identified as bei ng only the brain type according to a polymerase chain reaction-based assay using RNA obtained from gastric carcinoma cells and primers specific to muscle, liver and brain types of glycogen phosphorylase. The intranuclear localization of the brain-type isoform was confirmed by immunoelectron microscopical analyses. Further investigation to examine the nuclear localization in human carcinoma tissue (145 and 25 specimens with gastric and colonic carcinoma respectively) was carried out by immunohistochemistry using specific anti-brain-type antibody. Nuclear immunostaining was observed in seven cases out of 145 gastric carcinoma. The present study is the first to clarify the nuclear localization of glycogen phosphorylase with enzymatic activity in gastrointestinal carcinoma. The isoform of the enzyme expressed in the carcinoma was identified as the brain type. These results warrant further studies on the mechanisms for transporting the large molecule of brain-type glycogen phosphorylase to nuclei and its function in the nucleus of carcinoma cells.
Similar content being viewed by others
References
Chomczynski, P. & Sacchi, N. (1989) Single-step method of RNA isolation by acid guanidinium thiocyanate- phenol-chloroform extraction. Anal. Biochem. 162, 156-9.
Cori, C.F. & Cori, G.T. (1936) Mechanism and formation of hexosemonophosphate in muscle and isolation of a new phosphate ester. Proc. Soc. Exp. Biol. Med. 34, 702-12.
Davis, C.H., Schliselfeld, L.H., Wolf, D.P., Leavitt, C.A. & Krebs, E.G. (1967) Interrelationships among glycogen phosphorylase isozymes. J. Biol. Chem. 242, 4824-33.
Dingwall, C. & Laskey, R.A. (1986) Protein import into the cell nucleus. Annu. Rev. Cell Biol. 2, 367-90.
Gelinas, R.P., Froman, B.E., Mcelroy, F., Tait, R.C. & Gorin, F.A. (1989) Human brain glycogen phosphorylase: characterization of fetal cDNA and genomic sequence. Mol. Brain Res. 6, 177-85.
Goldfarb, D.S, Gariepy, J., Schoolnik, G. & Kornberg R.D. (1986) Synthetic peptides as nuclear localization signals. Nature 322, 641-4.
Gorin, F.A., Ignacio, P., Gelinas, R. & Carlsen, R. (1989) Abnormal expression of glycogen phosphorylase genes in regenerated muscle. Am. J. Physiol. 257, C495-503.
Hashimoto, K. Tamura, K. Otani, H. & Tanaka, O. (1995) Histochemical and immunohistochemical studies related to the role of glycogen in human developing digestive organs. Anat. Embryol. 192, 497-505.
Holm, L. & Sander, C. (1995) Evolutionary link between glycogen phosphorylase and a DNA modifying enzyme. EMBO J. 14, 1287-93.
Ignacio, P.C, Baldwin, B.A., Vijayan, V.K, Tait, R.C, & Gorin, F.A. (1990) Brain isozyme of glycogen phosphorylase: immunohistochemical localization within the central nervous system. Brain Res. 529, 42-9.
Kalderon, D., Roberts, B.L., Richardson, W.D. & Smith, A.E. (1984) A short amino acid sequence able to specify nuclear location. Cell 39, 499-509.
Karasaki, S. (1971) Cytoplasmic and nuclear glycogen synthesis in Novikoff ascites hepatoma. J. Ultrastruct. Res. 35, 181-96.
Lang, I., Scholz, M. & Peters, R. (1986) Molecular mobility and nucleocytoplasmic øux in hepatoma. J. Cell Biol. 102, 1183-90.
Matsuzaki, H., Shimada, S., Uno, K., Tsuruta, J. & Ogawa, M. (1998) Novel subtyping of intestinal metaplasia in the human stomach: brain-type glycogen phosphorylase expression in the proliferative zone and its relationship with carcinogenesis. Am. J. Clin. Pathol. 109, 181-9.
Nakano, K., Hwang, P. & Fletterick, R. J. (1986) Complete cDNA sequence for rabbit muscle glycogen phosphorylase. FEBS Lett. 204, 283-7.
Newgard, C.B., Littman, D.R., Genderen, C.V, Smith, M. & Fletterick, R.J. (1988) Human brain glycogen phosphorylase. J. Biol. Chem. 263, 3850-7.
Newgard, C.B, Hwang, P.K. & Fletterick, R.J. (1989) The family of glycogen phosphorylase: structure and function. Crit. Rev. Biochem. Mol. Biol. 24, 69-99.
Newmayer, D.D. & Forbes, D. J. (1988) Nuclear import can be separated into distinct steps in vitro: nuclear pore binding and translocation. Cell 52, 641-53.
Ohyumi, M. & Takano, S. (1977) Intranuclear synthesized and native glycogen particles in human gastric cancer: ultrastructure and histochemistry. Histochemistry 50, 239-50.
Robbins, J., Dilworth, S.M., Laskey, R.A. & Dingwall, C. (1991) Two interdependent basic domains in nucleoplasmin nuclear targeting sequence: identification of a class of bipartite nuclear targeting sequence. Cell 64, 615-23.
Roscoe, R.A, Gorin, F.A. & Tait, R.C. (1991) Human astrocytoma U251 RNA and genomic brain glycogen phosphorylase sequence. Mol. Brain Res. 10, 273-5.
Sato, K., Morris, H.P. & Weinhouse, S. (1972) Phos-phorylase: a new isozyme in rat hepatic tumors and fetal liver. Science 178: 879-81.
Shimada, S., Maeno, M., Misumi, A., & Akagi, M. (1984) Histochemical study of phosphorylase in proliferating cells of intestinal metaplasia and carcinoma of the human stomach. Scand. J. Gastroenterol. 19, 965-70.
Shimada, S., Maeno, M., Akagi, M., Hatayama, I., Sato, T. & Sato, K. (1986) Immunohistochemical detection of glycogen phosphorylase isoenzymes in rat and human tissues. Histochem J. 18, 334-8.
Shimada, S., Maeno, M., Misumi, A., Takano, S. & Akagi, M. (1987) Antigen reversion of glycogen phosphorylase isoenzyme in carcinoma and proliferative zone of intestinal metaplasia of the human stomach. Gastroenterology 93, 35-40.
Shimada, S., Honmyo, U., Yagi, Y., Ikeda, T., Yokota, T. & Ogawa M. (1992) Expression of glycogen phosphorylase activity in minute gastric carcinoma. Am. J. Gastroenterol. 87, 1230-1.
Takashi, M., Koshikawa, T., Kurobe, N. & Kato, K. (1989) Elevated concentrations of brain type glycogen phosphorylase in renal cell carcinoma. Jpn. J. Cancer Res. 80, 975-80.
Takeuchi, T. & Kuriaki, H. (1955) Histochemical detection of phosphorylase in animal tissues. J. Histochem. Cytochem. 3, 153-60.
Rights and permissions
About this article
Cite this article
Uno, K., Shimada, S., Tsuruta, J. et al. Nuclear Localization of Brain-type Glycogen Phosphorylase in Some Gastrointestinal Carcinoma. Histochem J 30, 553–559 (1998). https://doi.org/10.1023/A:1003239302471
Issue Date:
DOI: https://doi.org/10.1023/A:1003239302471