Abstract
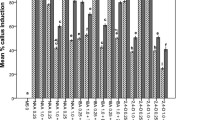
Shoot tips, cotyledonary nodes and hypocotyls of chickpea (Cicer arietinum L.) were grown on 3 media: plant induction medium (PIM), callus induction medium (CIM), and shoot induction medium (SIM). Maximum growth and differentiation was seen in PIM, whereas minimum was observed in CIM. Shoot tips which differentiated to multiple shoots evolved negligible amounts of ethylene. Maximum ethylene evolution was recorded by hypocotyls in PIM. Ethylene appears to have stimulatory effect on shoot bud differentiation in cotyledonary nodes. But in hypocotyls, increased ethylene inhibited growth and differentiation. Calli on media containing only auxin (PIM) evolved significantly more ethylene, whereas those on media with cytokinin (SIM) evolved more methane. Callus forming explants like cotyledonary nodes and hypocotyls evolve more ethylene than shoot tips.
Similar content being viewed by others
References
Adkins, S.W., Shiraishi, T.: Rice callus physiology-identification of volatile emissions and their effects on culture growth.-Physiol. Plant. 78: 526-531, 1990.
Chandra, R., Chatrath, A., Polisetty, R., Khetarpal, S.: Differentiation of in vitro grown explants of chickpea (Cicer arietinum L.).-Indian J. Plant Physiol. 36: 121-124, 1993.
Chi, G.L., Barfield, D.G., Sim, G.E., Chong, Pua, E.C.: Effect of silver nitrate and aminoethoxy-vinyl glycine on in vitro shoot and root organogenesis from seedling explants of recalcitrant Brassica genotypes.-Plant Cell Rep. 9: 195-198, 1990.
Chi, G.L., Pua, E.C., Goh, C.J.: Role of ethylene on de novo shoot regeneration from cotyledonary explants of Brassica campestris ssp. pekinensis (Lour) Olsson in vitro.-Plant Physiol. 96: 178-183, 1991.
Chraibi, K.M.B., Latche, A., Roustan, J.P., Fallot, J.: Stimulation of shoot regeneration from cotyledons of Helianthus annus by ethylene inhibitors, silver and cobalt.-Plant Cell Rep. 10: 204-207, 1991.
Chraibi, K.M.B., Castelle, J.C., Latche, A., Roustan, J.P., Fallot, J.: A genotype independent system of regeneration from cotyledons of sunflower Helianthus annus L. The role of ethylene.-Plant Sci. 86: 215-221, 1992.
Fontana, G.S., Santini, L., Caretto, S., Frugis, G., Mariotti, D.: Genetic transformation in grain legumes (Cicer arietinum L. (chickpea).-Plant Cell Rep. 12: 193-194, 1993.
Jackson, M.B., Abbott, A.J., Belcher, A.R., Hall, K.C., Butler, R., Comeron, J.: Ventilation in plant tissue culture and effects of poor aeration in ethylene and carbon dioxide accumulation, oxygen depletion and explant development.-Ann. Bot. 67: 229-237, 1991.
Keevers, C., Boyer, N., Courduroux, J.C., Gaspar, T.: Influence of ethylene on proliferation and growth of rose shoot culture.-Plant Cell Tissue Organ Cult. 28: 175-181, 1992.
Mustafa, M.D., Parthasarathy, M., Mallaian, B.: Methyl methane sulponate (MMS) induced multiple shoot from cotyledon cultures of cucumber (Cucumis sativa L. var. Poinsett).-Adv. Plant Sci. 4: 419-422, 1991.
Palmer, C.E.: Enhanced shoot regeneration from Brassica campestris by silver nitrate.-Plant Cell Rep. 11: 541-545. 1992.
Patil, P., Chandra, R., Khetarpal, S., Polisetty, R.: Role of polyamines and ethylene inhibitors on somatic embryo induction in chickpea (Cicer arietinum L.).-In: Abstract presented at 2nd International Crop Science Congress. P. 228. National Academy of Agricultural Sciences, New Delhi 1996.
Perez Bermudez, P., Cornejo, M.J., Segura, J.: A morphogenetic role for ethylene in hypocotyl cultures of Digitalis obscura L.-Plant Cell Rep. 4: 188-190, 1985.
Perrata, P., Alpi, A., Loschiavo, F.: Influence of ethanol on plant cells and tissues.-J. Plant Physiol. 126: 181-188, 1996.
Pua, E.C., Sim, G.E., Chi, C.L., Kong, L.F.: Synergestic effect of ethylene inhibitors and putrescine on shoot regeneration from hypocotyl explants of Chinese radish Raphanus sativum L. var. longipinnatus Bailey in vitro.-Plant Cell Rep. 15: 685-690, 1996.
Purnhauser, P., Medgyesy, P., Czeeko, M., Dix, P.J., Marton, L.: Stimulation of shoot regeneration in Triticum aestivum and Nicotiana plumbaginifolia Viv. tissue culture using the ethylene inhibitor AgNO3. Plant Cell Rep. 6: 1-4, 1987.
Righetti, E., Magnanini, E., Infante, R., Predieri, S.: Ethylene, ethanol, acctaldehyde and carbon dioxide released by Prunus airum shoot cultures.-Physiol. Plant. 78: 507-510, 1990.
Roustan, J.P., Latche, A., Fallot, J.: Control of carrot somatic embryogenesis by AgNO3, an inhibitor of ethylene action: Effect on arginine decarboxylase activity.-Plant Sci. 67: 89-95, 1990.
Sethi, U., Basu, A., Mukherjec, S.G.: Control of cell proliferation and differentiation by modulators of ethylene biosynthesis and action in Brassica hypocotyl explants.-Plant Sci. 69: 225-29, 1990.
Singh, R.P., Singh, B.D., Jaiswal, H.K., Singh, R.M., Singh, R.B.: Organogenesis in callus cultures of chickpea.-Indian J. agr. Sci. 52(2): 86-90, 1982.
Theriou, K.D., Economou, A.S., Sfakiotakis, M.: Promotion of petunia (Petunia hybrida L.) regeneration in vitro by ethylene.-Plant Cell Tissue Organ Cult. 32: 219-225, 1993.
Thomas, D.S., Murashige, T.: Volatile emission of plant tissue cultures. Effect of auxins 2,4-D on production of volatile callus cultures.-In vitro 15: 659-663, 1979.
Zobel, W.R., Roberts, H.W.: Effects of low concentrations of ethylene on cell division and cytodifferentiation in lettuce pith explants.-Can. J. Bot. 56: 987-990, 1978.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chandra, R., Khetarpal, S. & Polisetty, R. Effect of plant growth regulators on evolution of ethylene and methane by different explants of chickpea. Biologia Plantarum 40, 337–343 (1997). https://doi.org/10.1023/A:1001053627885
Issue Date:
DOI: https://doi.org/10.1023/A:1001053627885