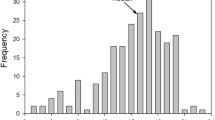
Abstract
Females of Dawson's burrowing bees have a well-defined brood cell cycle involving cell construction, waxing, provisioning, egg laying, and cell capping. In one study population, nesting bees built smaller brood cells for offspring of lower weight and larger ones for heavier offspring, demonstrating their ability to anticipate the desired size of an offspring at the outset of a brood cell cycle. Furthermore, individual females varied the number of provisioning trips made per brood cell cycle by a factor of two or more, apparently exercising control over the amount of brood provisions supplied to an offspring. The size distribution of emerging males at two widely separated locations in 1997 was nearly identical to that recorded in 1995. These findings suggest that the production of small males (minors) is the result of active female control rather than the product of food shortages that force females to undersupply some brood cells. Female foraging decisions resulted in a bimodal distribution of weights of mature dormant larvae at one site in 1997. However, the times required to complete brood cell cycles at this site were not distributed bimodally. This result stemmed in part from daily variation among females in the duration of their provisioning trips as well as from seasonal variation in provisioning trip times. When provisioning trips lasted longer, females tended to make fewer trips per brood cell cycle, and so were presumably more likely to produce minor sons. As a result, the weight of an offspring was not tightly linked to the time investment required to produce it, making it difficult to compare the relative costs of minors and majors in terms of maternal time investments.
Similar content being viewed by others
REFERENCES
Alcock, J. (1996a). Provisional rejection of three alternative hypotheses on the maintenance of a size dichotomy in Dawson's burrowing bees (Amegilla dawsoni) (Apidae, Anthophorini). Behav. Ecol. Sociobiol. 39: 181–188.
Alcock, J. (1996b). The relation between body size, fighting, and mating success in Dawson's burrowing bee, Amegilla dawsoni (Apidae, Apinae, Anthophorini). J. Zool. 239: 663–674.
Alcock, J. (1997a). Small males emerge earlier than large males in Dawson's burrowing bee (Amegilla dawsoni) (Hymenoptera: Anthophorini). J. Zool. 242: 453–462.
Alcock, J. (1997b). Competition from large males and the alternative mating tactics of small males of Dawson's burrowing bee (Amegilla dawsoni) (Apidae, Apinae, Anthophorini). J. Insect Behav. 10: 99–113.
Allen, T., Cameron, S., McGinley, R., and Heinrich, B. (1978). The role of workers and new queens in the ergonomics of a bumblebee colony (Hymenoptera: Apoidea). J. Kans. Entomol. Soc. 51: 329–342.
Boomsma, J. J. (1989). Sex-investment ratios in ants: Has female bias been systematically overestimated? Am. Nat. 133: 517–532.
Brockmann, H. J., and Grafen, A. (1992). Sex ratios and life-history patterns of a solitary wasp, Trypoxylon (Trypargilum) politum (Hymenoptera: Sphecidae). Behav. Ecol. Sociobiol. 30: 7–28.
Cardale, J. (1968). Observations on nests and nesting behaviour of Amegilla (Asarapoda) sp. (Hymenoptera: Apoidea: Anthophorinae). Aust. J. Zool. 16: 709–713.
Crozier, R. H., and Pamilo, P. (1993). Sex allocation in social insects: Problems in prediction and estimation. In Wrensch, D. I., and Ebbert, M. A. (eds.), Evolution and Diversity of Sex Ratios in Insects and Mites, Chapman and Hall, London, pp. 369–383.
Danforth, B. N. (1990). Provisioning behavior and the estimation of investment ratios in a solitary bee, Calliopsis (Hypomacrotera) persimilis (Cockerell) (Hymenoptera: Andrenidae). Behav. Ecol. Sociobiol. 27: 159–168.
Field, J. (1992). Patterns of nest provisioning and parental investment in the solitary digger wasp Ammophila sabulosa. Ecol. Entomol. 17: 43–51.
Frank, S. A. (1995). Sex allocation in solitary bees and wasps. Am. Nat. 146: 316–323.
Gerber, H. S., and Klostermeyer, E. C. (1970). Sex control by bees: A voluntary act during fertilization. Science 167: 82–84.
Helms, K. R. (1994). Sexual size dimorphism and sex ratios in bees and wasps. Am. Nat. 143: 418–434.
Houston, T. F. (1991). Ecology and behaviour of the bee Amegilla (Asaropoda) dawsoni (Rayment) with notes on a related species (Hymenoptera: Anthophoridae). Rec. W. Aust. Mus. 15: 591–609.
Kim, J.-Y. (1999). Influence of resource level on maternal investment in a leaf-cutter bee (Hymenoptera: Megachilidae). Behav. Ecol. (in press).
Klostermeyer, E. C., Mech, S. J., Jr., and Rasmussen, W. B. (1973) Sex and weight of Megachile rotundata (Hymenoptera: Megachilidae) progeny associated with provision weights. J. Kans. Entomol. Soc. 39: 524–535.
Maeta, Y., and Sugiura, N. (1990). Decision-making in a mason bee, Osmia cornifrons (Radoszkowski) (Hymenoptera, Megachilidae): Does the mother bee fertilize her eggs depending on their sizes? Japan. J. Entomol. 58: 197–203.
Minckley, R. L., Wcislo, W. T., Yanega, D., and Buchmann, S. L. (1994). Behavior and phenology of a specialist bee (Dieunomia) and sunflower (Helianthus) pollen availability. Ecology 75: 1406–1419.
Rosenheim, J. A., Nonacs, P., and Mangel, M. (1996). Sex ratios and multifaceted parental investment. Am. Nat. 148: 501–535.
Rust, R. W. (1993). Cell and nest construction costs in two cavity-nesting bees, Osmia lignaria propinqua and Osmia ribifloris biedermannii (Hymenoptera: Megachilidae). Ann. Entomol. Soc. Am. 86: 327–332.
Schmid-Hempel, P., Kacelnik, A., and Houston, A. I. (1985). Honeybees maximize efficiency by not filling their crop. Behav. Ecol. Sociobiol. 17: 61–66.
Strickler, K. (1982). Parental investment per offspring by a specialist bee: Does it change seasonally? Evolution 36: 1098–1100.
Torchio, P. F., and Tepedino, V. J. (1980) Sex ratio, body size and seasonality in a solitary bee, Osmia lignaria propinqua Cresson (Hymenoptera: Megachilidae). Evolution 34: 993–1003.
Torchio, P. F., and Tepedino, V. J. (1982). Phenotypic variability in nesting success among Osmia lignaria propinqua females in a glasshouse environment. Ecol. Entomol. 7: 453–462.
Trivers, R. L., and Hare, H. (1976). Haplodiploidy and the evolution of the social insects. Science 191: 249–263.
Visscher, P. K., and Danforth, B. N. (1993). Biology of Calliopsis pugionis (Hymenoptera: Andrenidae): Nesting, foraging and investment sex ratio. Ann. Entomol. Soc. Am. 86: 822–832.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alcock, J. The Nesting Behavior of Dawson's Burrowing Bee, Amegilla dawsoni (Hymenoptera: Anthophorini), and the Production of Offspring of Different Sizes. Journal of Insect Behavior 12, 363–384 (1999). https://doi.org/10.1023/A:1020843606530
Issue Date:
DOI: https://doi.org/10.1023/A:1020843606530