Abstract
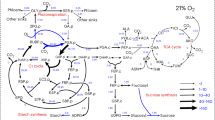
Carbon isotope effects were investigated for the reaction catalyzed by the glycine decarboxylase complex (GDC; EC 2.1.2.10). Mitochondria isolated from leaves of pea (Pisum sativum L.) and spinach (Spinacia oleracea L.) were incubated with glycine, and the CO2 evolved was analyzed for the carbon isotope ratio (δ13C). Within the range of parameters tested (temperature, pH, combination of cofactors NAD+, ADP, pyridoxal 5-phosphate), carbon isotope shifts of CO2 relative to the C1-carboxyl carbon of glycine varied from +14‰ to −7‰. The maximum effect of cofactors was observed for NAD+, the removal of which resulted in a strong 12C enrichment of the CO2 evolved. This indicates the possibility of isotope effects with both positive and negative signs in the GDC reaction. The measurement of δ13C in the leaves of the GDC-deficient barley (Hordeum vulgare L.) mutant (LaPr 87/30) plants indicated that photorespiratory carbon isotope fractionation, opposite in sign when compared to the carbon isotope effect during CO2 photoassimilation, takes place in vivo. Thus the key reaction of photorespiration catalyzed by GDC, together with the key reaction of CO2 fixation catalyzed by ribulose-1,5-bisphosphate carboxylase, both contribute to carbon isotope fractionation in photosynthesis.
Similar content being viewed by others
References
Arens NC, Jahren AH and Amundson R (2000) Can C3 plants faithfully record the carbon isotopic composition of atmospheric carbon dioxide? Paleobiology 26: 137–164
Bergman A and Ericson I (1983) Effects of NADH, succinate and malate on the oxidation of glycine in spinach leaf mitochondria. Physiol Plant 59: 421–427
Blackwell RD, Murray AJS and Lea PJ (1990) Photorespiratory mutants of the mitochondrial conversion of glycine to serine. Plant Physiol 94: 1316–1322
Christeller JT, Laing WA and Troughton JH (1976) Isotope discrimination by ribulose 1,5-bisphosphate carboxylase. No effect of temperature or HCO3 - concentration. Plant Physiol 57: 580–582
Craig H (1957) Isotopic standards for carbon and oxygen and correction factors for mass-spectrometric analysis of carbon dioxide. Geochim Cosmochim Acta 12: 133–149
Dalziel K and Londesborough JC (1968) The mechanisms of reductive carboxylation reactions. Carbon dioxide or bicarbonate as substrate of nicotinamide-adenine dinucleotide phosphate-linked isocitrate dehydrogenase and ‘malic’ enzyme. Biochem J 110: 223–230
Duranceau M, Ghashghaie J, Badeck F, Deleens E and Cornic G (1999) δ 13C of CO2 respired in the dark in relation to δ 13C of leaf carbohydrates in Phaseolus vulgaris L. under progressive drought. Plant Cell Environ 22: 515–523
Farquhar GD, Ehleringer JR and Hubick KT (1989) Carbon isotope discrimination and photosynthesis. Annu Rev Plant Physiol Plant Mol Biol 40: 503–537
Gardeström P and Edwards GE (1983) Isolation of mitochondria from leaves of C3, C4, and crassulacean acid metabolism plants. Methods Enzymol 148: 421–432
Gillon JS and Griffiths H (1997) The influence of (photo)respiration on carbon isotope discrimination in plants. Plant Cell Environ 20: 1217–1230
Gueguen V, Macherel D, Neuburger M, Saint Pierre C, Jaquinod M, Gans P, Douce R and Bourguignon J (1999) Structural and functional characterization of H protein mutants of the glycine decarboxylase complex. J Biol Chem 274: 26344–26352
Guy RD, Fogel ML and Berry JA (1993) Photosynthetic fractionation of the stable isotopes of oxygen and carbon. Plant Physiol 101: 37–47
Igamberdiev AU, Ivlev AA, Threlkeld CN and Bykova NV (1998) Mitochondrial oxidative decarboxylation of glycine is a point of carbon isotope fractionation in photosynthetic tissue. In: Møller IM, Gardeström P, Glimelius K and Glaser E (eds) Plant Mitochondria: From Gene to Function, pp 365–369. Backhuys Publishers, Leiden, The Netherlands
Igamberdiev AU, Bykova NV, Lea PJ and Gardeström P (2001) The role of photorespiration in redox and energy balance of photosynthetic plant cells: A study with a barley mutant deficient in glycine decarboxylase. Physiol Plant 111: 427–438
Ivlev AA (1993) On the flows of light and heavy carbon during photosynthesis and photorespiration coupling. Russian J Plant Physiol 40: 752–758
Ivlev AA, Bykova NV and Igamberdiev AU (1996) Fractionation of carbon (13C/12C) isotopes in glycine decarboxylase reaction. FEBS Lett 386: 174–176
Ivlev AA, Igamberdiev AU, Threlkeld C and Bykova NV (1999a) Carbon isotope effects in the glycine decarboxylase reaction in vitro on mitochondria from pea and spinach. Russian J Plant Physiol 46: 653–660
Ivlev AA, Pichuzkin VI and Knyazev DA (1999b) Developmental changes in the carbon isotope composition of wheat organs in relation to photorespiration. Russian J Plant Physiol 46: 443–451
Jordan F, Kuo DJ and Monse EU (1978) Carbon kinetic isotope effects on pyruvate decarboxylation catalyzed by yeast pyruvate dehydrogenase complex. J Am Chem Soc 100: 2872–2878
Kalinkina LG and Udelnova TM (1991) Mechanism of involvement of the glycolate pathway in accumulation of free proline in a marine Chlorella under conditions of salinity. Sov Plant Physiol 38: 687–696
Klein SM and Sagers RD (1966) Glycine metabolism. I. Properties of the system catalyzing the exchange of bicarbonate with the carboxyl group of glycine in Peptococcus glycinophilus. J Biol Chem 241: 197–205
Krömer S (1995) Respiration during photosynthesis. Annu Rev Plant Physiol Plant Mol Biol 46: 45–70
Lenne C, Neuburger M and Douce R (1993) Effect of high physiological temperatures on NAD+ content of green leaf mitochondria – apparent inhibition of glycine oxidation. Plant Physiol 102: 1157–1162
Lennon AM, Pratt J, Leach G and Moore A (1995) Developmental regulation of respiratory activity in pea leaves. Plant Physiol 107: 925–932
Lin GH and Ehleringer JR (1997) Carbon isotopic fractionation does not occur during dark respiration in C3 and C4. Plant Physiol 114: 391–394
Lowry OH, Rosebrough NJ, Farr AL and Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275
Melander L and Saunders J (1980) Reaction Rates of Isotopic Molecules. Wiley, New York
Neuburger M, Polidori AM, Pietre E, Faure M, Jourdain A, Bourguignon J, Pucci B and Douce R (2000) Interaction between the lipoamide-containing H-protein and the lipoamide dehydrogenase (L-protein) of the glycine decarboxylase multienzyme system. 1. Biochemical studies. Eur J Biochem 267: 2882–2889
O'Leary MH (1989) Multiple isotope effects on enzyme catalyzed reactions. Annu Rev Biochem 58: 377–401
Ohlsson KEA and Wallmark PH (1999) Novel calibration with correction for drift and non-linear response for continuous flow isotope ratio mass spectrometry applied to the determination of δ 15N, total nitrogen, δ 13C and total carbon in biological material. Analyst 124: 571–577
Oliver DJ (1994) The glycine decarboxylase complex from plant mitochondria. Annu Rev Plant Physiol Plant Mol Biol 45: 323–337
Oliver DJ and Sarojini G (1987) Regulation of glycine decarboxylase by serine. In: Biggins J (ed) Progress in Photosynthesis Research, Vol III, pp 573–576. Martinus Nijhoff Publishers, The Hague
Oliver DJ, Neuburger M, Bourguignon J and Douce R (1990) Glycine metabolism by plant mitochondria. Physiol Plant 80: 487–491
Park R and Epstein S (1960) Carbon isotope fractionation during photosynthesis. Geochim Cosmochim Acta 21: 110–119
Raven JA and Farquhar GD (1990) The influence of N metabolism and organic acid synthesis on the natural abundance of isotopes of carbon in plants. New Phytol 116: 505–529
Sarojini G and Oliver DJ (1987) Glyoxylate and pyridoxal 5-phosphate inhibition of isolated glycine decarboxylase. In: Biggins J (ed) Progress in Photosynthesis Research, Vol III, pp 569–572. Martinus Nijhoff Publishers, The Hague
Sarojini G and Oliver DJ (1993) Extraction and partial characterization of the glycine decarboxylase multienzyme complex from pea leaf mitochondria. Plant Physiol 72: 194–199
Smith BN and Epstein S (1971) Two categories of 13C/12C ratios for higher plants. Plant Physiol 47: 380–384
Tobin AK, Sumar N, Patel M, Moore AL and Stewart GR (1988) Development of photorespiration during chloroplast biogenesis in wheat leaves. J Exp Bot 39: 833–843
Tobin AK, Thorpe JR, Hylton CM and Rawsthorne S (1989) Spatial and temporal influences on the cell-specific distribution of glycine decarboxylase in leaves of wheat (Triticum aestivum L.) and pea (Pisum sativum L.). Plant Physiol 91: 1219–1225
Vauclare P, Diallo N, Bourguignon J, Macherel D and Douce R (1996) Regulation of the expression of the glycine decarboxylase complex during pea leaf development. Plant Physiol 112: 1523–1530
Wingler A, Lea PJ and Leegood RC (1997) Control of photosynthesis in barley plants with reduced activities of glycine decarboxylase. Planta 202: 171–178
Wingler A, Lea PJ and Leegood RC (1999a) Photorespiratory metabolism of glyoxylate and formate in glycine-accumulating mutants of barley and Amaranthus edulis. Planta 207: 518–526
Wingler A, Quick WP, Bungard RA, Bailey KJ, Lea PJ and Leegood RC (1999b) The role of photorespiration during drought stress: An analysis utilizing barley mutants with reduced activities of photorespiratory enzymes. Plant Cell Environ 22: 361–373
Yates KK and Robbins LL (1999) Radioisotope tracer studies of inorganic carbon and Ca in microbially derived CaCO3. Geochim Cosmochim Acta 63: 129–136
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Igamberdiev, A.U., Ivlev, A.A., Bykova, N.V. et al. Decarboxylation of glycine contributes to carbon isotope fractionation in photosynthetic organisms . Photosynthesis Research 67, 177–184 (2001). https://doi.org/10.1023/A:1010635308668
Issue Date:
DOI: https://doi.org/10.1023/A:1010635308668