Abstract
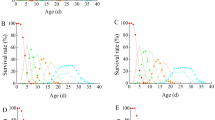
The chitin synthesis inhibitor lefenuron [N-(2,5-dichloro-4-(1, 1, 2, 3, 3, 3-hexafluoropropoxy)-phenylaminocarbonyl-2,6-difluorobenzamide] was found to have a negative effect on the encapsulation responses of Spodoptera littoralis larvae towards supernumeraries of Microplitis rufiventris parasitoid. Superparasitised hosts fed on compound-free diet showed strong encapsulation reaction (ca. 40.8%) to surplus parasitoid larvae having scars of physical combat. Treated hosts exhibited a significant reduction in either capsule thickness or encapsulation rate to the excess parasitoid larvae. Even when a capsule was formed, it had a thinner haemocy tic layer than tha t of control hosts. Studies on the differential haemocyte counts showed that failure of treated larvae to encapsulate injured surplus parasitoid larvae could be attributed to the following factors: (1) disturbance in plasmatocytes-granular cells (GR) ratio, e.g. abnormal increase in GR levels possibly causing faint haemocytic reaction to injured parasitoid larva(e); (2) the parasitoid surface becoming less attractive to the granulocytes and plasmatocytes; (3) the haemocytes involved in capsule formation (granulocytes, plasmatocytes) losing their ability to spread out over the surface of parasitoid larvae; and (4) the blood cells of the treated host failing to recognise wounded larvae as foreign bodies. Reduced encapsulation capacity might be due to a secondary effect(s) of IGR treatment inducing a suppression of adhesive activity of the plasmatocytes. These results may be useful for improving our understanding of insect immune reaction and/or manipulating parasitoids activity against nonhabitual Parasitoida.
Résumé
Il a été observée que l’inhibiteur de synthèse de la chitine—le lefenuron [N-(2,5 dichloro-4-(1, 1, 2, 3, 3, 3-hexafluoroprópoxy)-phenylaminocarbonyl)-2,6 difluorobénzamide], comportait un effet négatif sur les réponses des larves de S. littoralis, à encapsuler les larves surnuméraires issues du superparasitisme par le parasitoïde Microplitis rufiventris. Les larves hôtes superparasitées et nourries sur un milieu sans cet inhibiteur montraient une forte réaction d’encapsuler le surplus de larves (environ 48%) du parasitoïde, qui avaient des cicatrices résultant d’un combat physique. Chez les hôtes traités on a observé une réduction importante, soit de l’épaisseur de la capsule ou du taux d’encapsulation des larves excédentaires du parasitoïde. Même si une capsule était formée, celle-ci était caractérisée par une mince couche d’hémocytes en comparaison de celle des témoins. Les comptages différentiels d’hémocytes ont montré que l’échec des larves traitées à encapsuler les larves blessées excédentaires du parasitoïde pourrait être attribué aux facteurs suivants: (1) un déséquilibre du rapport entre plasmatocytes et granulocytes c-à-d une augmentation excessive du nombre de granulocytes, qui probablement débouche sur une faible réaction des hémocytes chez les larves blessées du parasitoïde; (2) la paroi corporelle du parasitoïde devient moins attrayante pour les granulocytes et les plasmatocytes; (3) les hémocytes (granulocytes, plasmatocytes) intervenant dans la formation de la capsule perdent leur pouvoir de couvrir la paroi corporelle du parasitoïde; et enfin, (4) les cellules sanguines de l’hôte traité sont incapables de reconnaître les larves blessées comme des corps étrangers. La capacité réduite d’encapsulation peut résulter des effets secondaires de régulation de la croissance de l’insecte due à l’inhibiteur qui annule l’activité adhesive des plasmatocytes. Ces résultats pourraient nous aider à mieux comprendre les mécanismes qui régissent la réaction immunitaire de l’insecte et/ou manipuler l’activité des parasitoïdes dans le sens d’éviter un parasitisme inhabituel au sein du groupe Parasitoida.
Similar content being viewed by others
References
Anonymous (1989) CGA 184’699—Insect growth inhibitor for cotton, soya, vegetables, potatoes, deciduous fruits, grapes and citrus. Ciba-Geigy Limited, Basle, Switzerland.
Davies, D. H. and Vinson, S. B. (1986) Passive evasion by eggs of the braconid parasitoid Cardiochiles nigrkeps of encapsulation in vitro by haemocytes of host Heliothis virescens. Possible role of fibrous layer in immunity. J. Insect Physiol. 32, 1003–1010.
Davies, D. H., Strand, M. R. and Vinson, S. B. (1987) Changes in differential haemocyte count and in vitro behaviour of plasma tocy tes from host Heliothis virescens caused by Campoletis sonorensis polydnavirus. J. Insect Physiol. 33, 143–153.
El-Minshawy A-M. (1963) Studies on the morphology and biology of Spodoptera exigua Hb. and Spodoptera latebrosa Led. in Alexandria. MSc Thesis, Faculty of Agriculture, University of Alexandria. 144 pp.
Götz, P. (1986) Encapsulation in arthropods, pp. 153–170. In Immunity in Invertebrates (Edited by M. Brehelin). Springer-Verlag, Berlin.
Gupta, A. P. (1985) Cellular elements in thehaemolymph, pp. 401–451. In Comprehensive Insect Physiology Biochemistry and Pharmacology, Vol. 3 (Edited by G. A. Kerkut and, L. T. Gilbert). Pergamon Press, New York.
Gupta, A. P. (1986) Haemocytic and Humoral Immunity in Insects. Wiley, New York. 535 pp.
Hammad, S. M., El-Minshawy, A. M. and Salama, A. (1965) Studies on Microplitis Rufiventris Kok. (Hym.; Braconidae). Bull. Soc. Entom. Egypte, XLIX, 215–219.
Hegazi, E. M. and El-Minshawy, A. M. (1979) Laboratory technique for mass rearing of Microplitis Rufiventris Kok. (Braconidae; Hymenoptera) an internal parasite of the cotton Ieafworm, Spodoptera Littoralis (Boisd.) (Noctuidae; Lepidoptera). Boll. Lab. Ent. Agr. "F. Silvestri" 36, 205–210.
Hegazi, E. M., El-Minshawy, A. M. and Hammad, S. M. (1977) Mass rearing of the Egyptian cotton Ieafworm, Spodoptera Littoralis (Bòisd.) on semi-artificial diet. Proceedings Second Arab Pesticide Confi, Tanta Univ., 61–70.
Hegazi, E. M., El-Minshawy, A. M. and Shaaban, M. A. (1984) The relation of parasite density of Microplitis Rufiventris Kok. (Hym.; Braconidae) to superparasitism and fate of its eggs. Z. ang. Ent. 97, 414–418.
Hung, S. H. and Boucias, D. G. (1992) Influence of Beauveria bassiana on the cellular defense response of thebeet army worm, Spodoptera exigua. J. Invertebr. Pathol. 60, 152–158.
Ibrahim, M. and Tawfik, K. (1975) A report on the survey of natural enemies attacking the American bollworm. Agricultural Research Center, Plant Protection Research Institute, Cairo (In Arabic).
Jones, J. C. (1962) Current concepts concerning insect haemocytes. Amer. Zool. 2(2), 209–246.
Kokujev, N. (1914) Hymenoptera parasitic nove fauna turanica, V.l. Platnikov Collecta, (parasitic Hymenoptera new to the fauna of Turkestan, collected by, V.l. Platnikov). Revue Russe d’Entomologie, St Petersburg, xiii (nos. 3 and 4), 513–514.
Lynn, D. C. and Vinson, S. B. (1977) Effects of temperature, host age, and hormones upon the encapsulation of Cardiochiles nigriceps eggs by Heliothis spp. J. Invertebr. Pathol. 29, 50–55.
Nappi, A. J. and Streams, F. A. (1969) Haemocytic reactions of Drosophila melanogaster to the parasites Pseudeucoila mellipes and P. bochei. J. Insect Physiol. 15, 1551–1566.
Neuwirth, M. (1974) Granular haemocytes, the main phagocytic blood cells in Calpodes ethlius. Can. J. Zool. 52, 783–784.
Ratcliffe, N. A. (1993) Cellular defense responses of insects: Unresolved problem, pp. 267–304. In Parasites and Pathogens of Insects 1 (Edited by N. E. Beckage, S. N. Thompson and, B. A. Federici). Academic Press, San Diego.
Ratcliffe, N. A. and Gagen, S. J. (1976) Cellular defense reactions of insect haemocytes in vivo: Nodule formation and development in G. mellonella and Pieris brassicae larvae. J. Invertebr. Pathol. 28, 373–382.
Ratcliffe, N. A. and Gagen, S. J. (1977) Studies on the in vivo cellular reactions of insects: An ultrastructural analysis of nodule formation in Galleria mellonella. Tissue Cell 9, 73–85.
Ratcliffe, N. A., Rowley, A. F., Fitzgerald, S. W. and Rhodes, C. P. (1985) Invertebrate immunity: Basic concepts and recent advances. Int. Rev. Cytol. 97, 186–350.
Rizki, R. M. and Rizki, T. M. (1984) Selective destruction of a host blood cell type by a parasitoid wasp. Proc. Natn. Acad. Sci., USA 81, 6154–6158.
Rosenberger, C. R. and Jones, J. C. (1960) Studies on total cell counts of the southern armyworm larva, Prodenia eridania (Lepidoptera). Ann. Entomol. Soc. Amer. 53, 351–355.
Rowley, A. F. and Ratcliffe, N. A. (1981) Insects. In Invertebrate Blood Cells Vol. 2 (Edited by N. A. Ratcliffe and, A. F. Rowley). Academic Press, London, New York.
Salt, G. (1968) The resistance of insect parasitoids to the defense reactions of their hosts. Biol. Rev. 43, 200–232.
Salt, G. (1970) The Cellular Defense Reactions of Insects. Cambridge University Press.
Schmit, A. R. and Ratcliffe, N. A. (1977) The encapsulation of foreign tissue implants in Galleria mellonella larvae. J. Insect Physiol. 23, 175–184.
Schmit, A. R., Rowley, A. F. and Ratcliffe, N. A. (1977) The role of Galleria mellonella haemocytes in melanin formation. J. Invertebr. Pathol. 29, 232–234.
Stoltz, D. B. and Guzo, D. (1986) Apparent haemocytic transformations associated with parasitoid induced inhibition of immunity In Malacosoma disstria larvae. J. Insect Physiol. 32, 377–388.
Strand, M. R. and Pech, L. L. (1995) Immunological basis for compatibility in parasitoid-host relationships. Annu. Rev. Entomol. 40, 31–56.
Takeda, N. (1977) Brain hormone carrier haemocytes in the moth Monemaflavescens. J. Insect Physiol. 23, 1245–1254.
Tanaka, T. (1987) Morphological changes in haemocytes of the host, Pseudaletia separata, parasitized by Microplitis mediator or Apenteles kariyai. Dev. Comp. Immunol. 11, 57–67.
Van den Bosch, R. and Messenger, P. S. (1973) Biological Control. Guildford and London, England. 180 pp.
Vilcinskas, A., Matha, V. and Götz, P. (1997) Inhibition of phagocytic activity of plasmatocytes isolated from Galleria mellonella by entomogenous fungi and their secondary metabolites. J. Insect Physiol. 43, 475–483.
Vinson, S. B. (1977) Microplitis croceipes: Inhibition of Heliothis zea defense’reaction to Cardiochiles nigriceps. Exp. Parasitol. 41, 112–117.
Vinson, S. B. and Abies, J. R. (1980) Interspecific competition among endoparasitoids of tobacco budworm larvae (Lep.; Noctuidae). Entomophaga 25, 357–362.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hegazi, E.M., Khafagi, W.E. & Abd El-Aziz, G.M. Effect of Lefenuron, a Chitin Synthesis Inhibitor, on Encapsulation Response of Spodoptera littoralis Larvae To Surplus Microplitis rufiventris Larvae. Int J Trop Insect Sci 18, 357–363 (1998). https://doi.org/10.1017/S1742758400018592
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1017/S1742758400018592
Key Words
- Spodoptera littoralis
- Microplitis rufiventris
- superparasitism
- IGR
- lefenuron
- encapsulation
- host haemocyte profile