Abstract
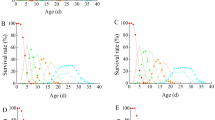
The immature development and reproduction of the predatory mites Amblyseius largoensis (Muma), Proprioseiopsis lenis (Corpuz and Rimando), and Amblyseius swirskii Athias-Henriot (Acari: Phytoseiidae) were investigated using both thrips eggs and first instars of the western flower thrips, Frankliniella occidentalis Pergande, as prey in a controlled laboratory environment at 25 °C and 60% relative humidity. When provided with thrips eggs as food, A. largoensis exhibited a notably shorter immature development period for both males (7.05 days) and females (6.51 days) as compared with A. swirskii (8.05 and 7.19 days, respectively) and P. lenis (8.10 days and 7.05 days, respectively). Amblyseius largoensis also displayed a higher oviposition rate (2.19 eggs/female/day) than A. swirskii and P. lenis (1.79 and 1.78 eggs/female/day, respectively). Moreover, it exhibited the highest fecundity (25.34 eggs/female), followed by P. lenis (24.23 eggs/female) and A. swirskii (22.86 eggs/female). These variations led to A. largoensis having the highest intrinsic rate of increase (rm) at 0.209, followed by A. swirskii at 0.188, and P. lenis at 0.165. However, when the predatory mites were provided with first instars of F. occidentalis, A. swirskii demonstrated a faster immature development period for both males (7.67 days) and females (7.59 days) as compared with P. lenis (9.00 days and 7.86 days, respectively) and A. largoensis (8.47 days and 8.61 days, respectively). While the oviposition rates of P. lenis (1.92 eggs/female/day) and A. swirskii (1.90 eggs/female/day) were similar when feeding on this prey, A. largoensis produced fewer eggs (1.83 eggs/female/day). Further, A. swirskii exhibited the highest fecundity (31.93 eggs/female), followed by A. largoensis (25.71 eggs/female) and P. lenis (23 eggs/female). Consequently, the intrinsic rate of increase (rm) on thrips first instars was highest in A. swirskii (0.190), followed by A. largoensis (0.186), and P. lenis (0.176). In summary, our findings indicate that in terms of life history parameters A. largoensis performs optimally when feeding on thrips eggs, whereas A. swirskii performs best when preying on the mobile first instars of the thrips. These insights into the dietary preferences and reproductive capabilities of the studied predatory mite species have important implications for their potential use as biological control agents against F. occidentalis in agricultural settings.
Similar content being viewed by others
Data availability
The data and materials generated or analyzed during this study are available upon reasonable request. Researchers interested in accessing the dataset or materials can contact the corresponding author, Patrick De Clercq, through his ORCID profile (https://orcid.org/0000-0003-0664-1602) at patrick.declercq@ugent.be. Access to the data and materials will be provided in accordance with ethical and legal considerations, ensuring participant confidentiality and privacy. This availability aligns with our commitment to fostering transparency and facilitating future scientific inquiries.
References
Ahmed N, Lou M (2018) Efficacy of two predatory phytoseiid mites in controlling the western flower thrips, Frankliniella occidentalis (Pergande) (Thysanoptera: Thripidae) on cherry tomato grown in a hydroponic system. Egypt J Biol Pest Co 28:1–6
Alipour Z, Fathipour Y, Farazmand A (2016) Age-stage predation capacity of Phytoseiulus persimilis and Amblyseius Swirskii (Acari: Phytoseiidae) on susceptible and resistant rose cultivars. Int J Acarol 42:224–228. https://doi.org/10.1080/01647954.2016.1171797
Bakker FM, Sabelis MW (1989) How larvae of Thrips tabaci reduce the attack success of Phytoseiid predators. Entomol Exp Appl 50:47–51. https://doi.org/10.1111/j.1570-7458.1989.tb02313.x
Birch LC (1948) The intrinsic rate of natural increase of an insect population. J Anim Ecol 17:15–26. https://doi.org/10.2307/1605
Calvo FJ, Knapp M, Van Houten YM, Hoogerbrugge H, Belda JE (2015) Amblyseius Swirskii: what made this predatory mite such a successful biocontrol agent? Exp Appl Acarol 65:419–433. https://doi.org/10.1007/s10493-014-9873-0
Carrillo D, Peña JE, Hoy MA, Frank JH (2010) Development and reproduction of Amblyseius largoensis (Acari: Phytoseiidae) feeding on pollen, Raoiella indica (Acari: Tenuipalpidae), and other microarthropods inhabiting coconuts in Florida, USA. Exp Appl Acarol 52:119–129. https://doi.org/10.1007/s10493-010-9360-1
Choh Y, Ignacio M, Sabiles MW, Janssen A (2012) Predator-prey role reversals, juvenile experience and adult antipredator behaviour. Sci Rep 2:1–6
Chow A, Chau A, Heinz KH (2010) Compatibility of Amblyseius (Typhlodromips) swirskii (Athias-Henriot) (Acari: Phytoseiidae) and Orius insidiosus (Hemiptera: Anthocoridae) for biological control of Frankliniella occidentalis (Thysanoptera: Thripidae) on roses. Biol Control 53:188–196. https://doi.org/10.1016/j.biocontrol.2009.12.008
Corpuz LA, Rimando L (1966) Some Philippine Amblyseiinae (Phytoseiidae: Acarina). Philipp Agric Sci 50:114–136. https://www.researchgate.net/publication/307598333
Da Silva RR, Teodoro AV, De Sousa Silva MDJ, Reis PR, Silva SS (2015) Compatibility of pesticides with the generalist predatory mite Amblyseius largoensis (Acari: Phytoseiidae). Rev Colomb Entomol 41:76–80
De Alfaia JP, Barros MEN, Melo LL, Lima DB, Dias-Pini NDS, Melo JWS (2018) Biological performance of the predatory mites Amblyseius largoensis and Euseius concordis fed on eggs of Aleurodicus cocois. Syst Appl Acarol 23:2099–2103. https://doi.org/10.11158/saa.23.11.2
De Almeida AA, Janssen A (2013) Juvenile prey induce antipredator behaviour in adult predators. Exp Appl Acarol 59:275–282. https://doi.org/10.1007/s10493-012-9601-6
De Bruijn PJA, Egas M, Janssen A, Sabelis MW (2006) Pheromone-induced priming of a defensive response in western flower thrips. J Chem Ecol 32:1599–1603
Demiroz D, Kumral NA (2022) Research of resistance of western flower thrips, Frankliniella occidentalis (Pergande) (Thysanoptera: Thripidae) populations to different insecticide groups. Balkan Agric Congr 4:824–830
Demite PR, McMurtry JA, De Moraes GJ (2014) Phytoseiidae Database: a website for taxonomic and distributional information on phytoseiid mites (Acari). Zootaxa 3795:571–577. https://doi.org/10.11646/zootaxa.3795.5.6
Demite PR, De Moraes GJ, McMurtry JA, Denmark HA, Castilho RC (2020) Phytoseiidae Database www.lea.esalq.usp.br/phytoseiidae (accessed 02/06/2022)
Faraji F, Janssen A, Sabelis MW (2001) Predatory mites avoid ovipositing near counterattacking prey. Exp Appl Acaro 25:613–623
Gallego CE, Aterrado ED, Batomalaque CG (2003) Biology of the false spider mite, Rarosiella Cocosae Rimando, infesting coconut palms in Camiguin, northern Mindanao (Philippines). Philippine Entomol 17:187–192
Galvão A, Gondim MG, De Moraes GJ, De Oliveira JV (2007) Biology of Amblyseius largoensis (Muma) (Acari: Phytoseiidae), a potential predator of Aceria Guerreronis Keifer (Acari: Eriophyidae) on coconut trees. Neotrop Entomol 36:465–470
Gao Y, Lei Z, Reitz SR (2012) Western flower thrips resistance to insecticides: detection, mechanisms and management strategies. Pest Manag Sci 68:1111–1121
Ghasemzadeh S, Leman A, Messelink GJ (2017) Biological control of Echinothrips americanus by phytoseiid predatory mites and the effect of pollen as supplemental food. Exp Appl Acarol 73:209–221. https://doi.org/10.1007/s10493-017-0191-1
Goleva I, Zebitz CPW (2013) Suitability of different pollen as alternative food for the predatory mite Amblyseius Swirskii (Acari, Phytoseiidae). Exp Appl Acarol 61:259–283. https://doi.org/10.1007/s10493-013-9700-z
Hosseininia A, Khanjani M, Asadi M, Soltani J (2020) Life-history of the predatory mite Amblyseius Swirskii (Athias-Henriot) (Acari: Phytoseiidae) on Tetranychus Urticae Koch (Acari: Tetranychidae), Carpoglyphus lactis Linnaeus (Acari: Carpoglyphidae) and Trialeurodes vaporariorum (Westwood) (Hemiptera: Aleyrodidae). J Ornam Plants 10:155–166
Hulting FL, Orr DB, Obrycki JJ (1990) A computer program for calculation and statistical comparison of intrinsic rate of increase and associated life table parameters. Fla Entomol 73:601–612. https://doi.org/10.2307/3495274
Jacobson RJ, Croft P, Fenlon J (2001) Suppressing establishment of Frankliniella occidentalis Pergande (Thysanoptera: Thripidae) in cucumber crops by prophylactic release of Amblyseius Cucumeris Oudemans (Acarina: Phytoseiidae). Biocontrol Sci Technol 11:27–34. https://doi.org/10.1080/09583150020029718
Kamburov SS (1971) Feeding, development, and reproduction of Amblyseius largoensis on various food substances. J Econ Entomol 64:643–648
Khaliq A, Afzal M, Raza AM, Kamran M, Khan AA, Aqeel MA, Ullah MI, Khan BS, Kanwal H (2018) Suitability of Thrips tabaci L. (Thysonaptera: Thripidae) as prey for the phytoseiid mite, Neoseiulus barkeri Hughes (Acari: Phytoseiidae). Afr Entomol 16:131–135. https://doi.org/10.4001/003.026.0131
Kolokytha PD, Fantinou AA, Papadoulis G Th (2011) Temperature and diet effects on immature development of predatory mite Typhlodromus Athenas Swirski and Ragusa (Acari: Phyotseiidae). Environ Entomol 40:1577–1584. https://doi.org/10.1603/EN11098
Leman A, Messelink GJ (2015) Supplemental food that supports both predator and pest: a risk for biological control? Exp Appl Acarol 65:511–524
Liao JR, Ho CC, Ko CC (2013) Checklist of Phytoseiidae (Acari: Mesostigmata) from Taiwan. Formos Entomol 33:67–90
Liao JR, Ho CC, Ko CC (2021) Predatory mites (Acari: Mesostigmata: Phytoseiidae) intercepted from samples imported to Taiwan, with description of a new species. Zootaxa 4927:301–330. https://doi.org/10.11646/zootaxa.4927.3.1
Lotka AJ (1907) Relation between birth rates and death rates. Science 26:21–22
Magalhães S, Janssen A, Montserrat M, Sabelis MW (2005) Prey attack and predators defend: counterattacking prey trigger parental care in predators. Proc R Soc Lond Ser B-Biol Sci 272:1929–1933. https://doi.org/10.1098/rspb.2005.3127
Mai HT, Shamim S, Pham DKH (2019) Consumer concern about food safety in Hanoi. Vietnam Food Control 98:238–244. https://doi.org/10.1016/j.foodcont.2018.11.031
Maia ADN, Luiz AJB, Campanhola C (2000) Statistical inference on associated fertility life table parameters using jackknife technique. Computational aspects. J Econ Entomol 93:511–518. https://doi.org/10.1603/0022-0493-93.2.511
Marisa C, Sauro S (1990) Biological observations and life table parameters of Amblyseius Cucumeris (Oud.) (Acarina: Phytoseiidae) reared on different diets. Redia 73:569–583
McMurtry JA, Croft BA (1997) Lifestyles of phytoseiid mites and their roles in biological control. Annu Rev Entomol 42:291–321. https://doi.org/10.1146/annurev.ento.42.1.291
McMurtry JA, De Moraes GJ, Sourassou NF (2013) Revision of the lifestyles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Syst Appl Acarol 18:297–320. https://doi.org/10.11158/saa.18.4.1
Messelink GJ, Van Steenpaal EF, Ramakers PMJ (2006) Evaluation of phytoseiid predators for control of western flower thrips on greenhouse cucumber. Biocontrol 51:753–768. https://doi.org/10.1007/s10526-006-9013-9
Messelink GJ, Van Maanen R, Van Steenpaal EF, Janssen A (2008) Biological control of thrips and whiteflies by a shared predator: two pests are better than one. Biocontrol 44:372–379. https://doi.org/10.1016/j.biocontrol.2007.10.017
Meyer JS, Ingersoll CG, McDonald LL, Boyce MS (1986) Estimating uncertainty in population growth rates: jackknife vs. bootstrap techniques. Ecology 67:1156–1166. https://doi.org/10.2307/1938671
Momen FM (2009) Potential of three species of predatory phytoseiid mites as biological control agents of the peach silver mite, Aculus fockeui (Acari: Phytoseiidae and Eriophyidae). Acta Phytopathol Hun 44:151–158
Momen FM, Nasr A, El-Rady K, Metwally AM, Mahmoud YA (2016) Performance of five species of Phytoseiid mites (Acari: Phytoseiidae) on Bactrocera zonata eggs (Diptera: Tephritidae) as a factitious food. Acta Phytopathol Hun 51:123–132
Nasr A, Momen FM, Metwally AM, Gesraha MA, Abdallah A, Saleh KM (2015) Suitability of Corcyra Cephalonica eggs (Lepidoptera: Pyralidae) for the development, reproduction and survival of four predatory mites of the family Phytoseiidae (Acari: Phytoseiidae). Gesunde Pflanzen 67:175–181. https://doi.org/10.1007/s10343-015-0350-4
Navasero M, Corpuz-Raros L (2005) Functional response of three predatory phytoseiid mites (Phytoseiidae: Acari) to two-spotted mite, Tetranychus Urticae Koch (Tetranychidae: Acari). Philippine Entomol 19:168–181
Nguyen DT, Than TA, Jonckheere W, Nguyen VH, Van Leeuwen T, De Clercq P (2019a) Life tables and feeding habits of Proprioseiopsis Lenis (Acari: Phytoseiidae) and implications for its biological control potential in Southeast Asia. Syst Appl Acarol 24:857–865. https://doi.org/10.11158/saa.24.5.9
Nguyen VH, Jonckheere W, Nguyen DT, De Moraes GJ, Van Leeuwen T, De Clercq P (2019b) Phytoseiid mites prey effectively on thrips eggs: evidence from predation trials and molecular analyses. Biol Control 137:104012
Nguyen VS, Nguyen TMP, Ho NC, Nguyen XD, Do TD, Vu NH, Vuong TKH, Nguyen CT, Tran TTT (2020) Vietnamese agriculture before and after opening economy. Mod Econ 11:894–907. https://doi.org/10.4236/me.2020.114067
Nomikou M, Janssen A, Schraag R, Sabelis MW (2001) Phytoseiid predators as potential biological control agents for Bemisia tabaci. Exp Appl Acarol 25:271–291. https://doi.org/10.1023/A:1017976725685
Nomikou M, Janssen A, Schraag R, Sabelis MW (2004) Vulnerability of B. Tabaci immatures to Phytoseiid predators: consequences for oviposition and influence of alternative food. Entomol Exp Appl 110:95–102. https://doi.org/10.1111/j.0013-8703.2004.00114.x
Oliveira DC, Charanasri V, Kongchuensin M, Konvipasruang P, Chandrapatya A, De Moraes GJ (2012) Phytoseiidae of Thailand (Acari: Mesostigmata), with a key for their identification. Zootaxa 3453:1–24. https://doi.org/10.11646/Zootaxa.3453.1.1
Peña JE, Rodrigues JCV, Roda A, Carrillo D, Osborne LS (2009) Predator-prey dynamics and strategies for control of the red palm mite (Raoiella indica) (Acari: Tenuipalpidae) in areas of invasion in the neotropics. Proceedings of the 2nd meeting of IOBC/WPRS Bull 50:69–79
Pham VH, Mol A, Oosterveer P (2013) State governance of pesticide use and trade in Vietnam. NJAS-Wagen J Life Sc 67:19–26. https://doi.org/10.1016/j.njas.2013.09.001
Pham VH, Mol A, Oosterveer P, Van den Brink PJ, Pham TMH (2016) Pesticide use in Vietnamese vegetable production: a 10-year study. Int J Agr Sustain 14:325–338. https://doi.org/10.1080/14735903.2015.1134395
Pijnakker J, Arijs Y, de Souza A, Cellier M, Wäckers F (2016) The use of Typha angustifolia (cattail) pollen to establish the predatory mites Amblyseius Swirskii, Iphiseius degenerans, Euseius Ovalis and Euseius Gallicus in glasshouse crops. Integr Control Plant-Feeding Mites IOBC/WPRS Bull 120:47–52
Poushkova SV, Kasatkin DG (2020) Materials to the knowledge of the fauna of thrips (Thysanoptera) in Vietnam as a result of the expedition of FGBU VNIIKR. Plant Quarantine Nr 2(2):55–68. https://www.researchgate.net/publication/343474393
Prins M, Goldbach R (1998) The emerging problem of tospovirus infection and non-conventional methods of control. Trends Microbiol 6:31–35
Riahi E, Fathipour Y, Talebi AA, Mehrabadi M (2017) Linking life table and consumption rate of Amblyseius Swirskii (Acari: Phytoseiidae) in presence and absence of different pollens. Ann Entomol Soc Am 110:244–253. https://doi.org/10.1093/aesa/saw091
Saito Y (1986) Prey kills predator: counter-attack success of a spider mite against its specific phytoseiid predator. Exp Appl Acarol 2:47–62
Sarkar SC, Wang E, Zhang Z, Wu S, Lei Z (2019) Laboratory and glasshouse evaluation of the green lacewing, Chrysopa pallens (Neuroptera: Chrysopidae) against the western flower thrips, Frankliniella occidentalis (Thysanoptera: Thripidae). Appl Entomol Zool 54:115–121. https://doi.org/10.1007/s13355-018-0601-9
Schicha E, Elshafie M (1980) Four new species of phytoseiid mites from Australia, and three species from America redescribed (Acari: Phytoseiidae). Aust J Entomol 19:27–36. https://doi.org/10.1111/j.1440-6055.1980.tb00957.x
Seiedy M, Soleymani S, Hakimitabar M (2017) Development and reproduction of the predatory mite Amblyseius Swirskii Athias-Henriot (Acari: Phytoseiidae) on Tetranychus Urticae Koch (Acari: Tetranychidae) and Bemisia tabaci Gennadius (Heteroptera: Aleyrodidae). Int J Acarol 43:160–164. https://doi.org/10.1080/01647954.2016.1248486
Stavrinides MC, Mills NJ (2011) Influence of temperature on the reproductive and demographic parameters of two spider mite pests of vineyards and their natural predator. Biocontrol 56:315–325. https://doi.org/10.1007/s10526-010-9334-6
Teerling CR, Gillespie DR, Borden JH (1993) Utilization of western flower thrips alarm pheromone as a prey-finding kairomone by predators. Can Entomol 125:431–437
Tixier M-S, Douin M, Lopes I, Migeon A, Fossoud A, Navajas M (2022) Genetic diversity of the predatory mite Amblyseius Swirskii Athias-Henriot (Acari: Phytoseiidae) with an overview of its distribution and implications for biological control. Biol Control 168:104841. https://doi.org/10.1016/j.biocontrol.2022.104841
Vangansbeke D, Pijnakker J, Arijs Y, Wäckers F (2018) Thrips egg predation by phytoseiids: an overlooked pest control mechanism. IOBC/WPRS Bull 124:184–189
Vantornhout I, Minnaert HL, Tirry L, De Clercq P (2004) Effect of pollen, natural prey and factitious prey on the development of Iphiseius degenerans. Biocontrol 49:627–644. https://doi.org/10.1007/s10526-004-5280-5
Walzer A, Paulus W, Schausberger P (2004) Ontogenetic shifts in intraguild predation on thrips by Phytoseiid mites: the relevance of body size and diet specialization. Bull Entomol Res 94:577–584. https://doi.org/10.1079/BER2004329
Wimmer D, Hoffmann D, Schausberger P (2008) Prey suitability of western flower thrips, Frankliniella occidentalis, and onion thrips, Thrips tabaci, for the predatory mite Amblyseius Swirskii. Biocontrol Sci Technol 18:533–542
Yuan J, Zheng X, Wang J, Qian K, Feng J, Zhang Y, Zhang K, Liang P, Wu Q (2023) Insecticide resistance of western flower thrips, Frankliniella occidentalis (Pergande) in China. Crop Prot 172:106339. https://doi.org/10.1016/j.cropro.2023.106339
Acknowledgements
This research was supported in part by a scholarship of the Vietnamese government, and in part by a project funded by the Vietnam National Foundation for Science and Technology Development (NAFOSTED), and the Research Foundation– Flanders (FWO) (grant number FWO.106.2020.01 and G0E1221N).
Funding
This research was supported in part by a scholarship of the Vietnamese government, and in part by a project funded by the Vietnam National Foundation for Science and Technology Development (NAFOSTED), and the Research Foundation– Flanders (FWO) (grant number FWO.106.2020.01 and G0E1221N).
Author information
Authors and Affiliations
Contributions
Viet Ha Nguyen were responsible for conceptualization, methodology, experimental activities, statistical analysis, and writing of the manuscript. Duc Tung Nguyen contributed by providing advice on methodology, statistical analysis and assisting in revising the manuscript. Patrick De Clercq and Thomas Van Leeuwen were responsible for supervision, funding acquisition, project administration, resources and revision of the manuscript.
Corresponding author
Ethics declarations
We confirm that all the listed authors have read and approved the manuscript, and there are no other authors who haven’t been mentioned. We assure that the order of authors listed in the manuscript has been approved by all of us.
We confirm that all of the research mentioned in this manuscript that has involved experimental animals subject has been conducted with the approval of all relevant authorities and that such approvals are recognized in the manuscript.
We understand that the Corresponding Author is the primary contact for the Editorial process. The Corresponding Author is responsible for communicating with the other authors about progress, submitting revisions and final approval of proofs. We confirm that we have provided a current, accurate email address that the Corresponding Author can access.
Ethical approval
There are no human subjects in this article; hence, the research project does not require ethical approval, as all experimental work was conducted with known invertebrate species.
Consent to participate
I understand the general purposes, risks and methods of the research project titled “ Life table parameters of Amblyseius largoensis, Amblyseius swirskii and Proprioseiopsis lenis (Acari: phytoseiidae) fed on eggs and larvae of Frankliniella occidentalis”. I have obtained the informed consent from all individual participants enlisted in the study.
Consent to publish
I hereby grant permission for the researchers involved in the project titled “ Life table parameters of Amblyseius largoensis, Amblyseius swirskii and Proprioseiopsis lenis (Acari: Phytoseiidae) fed on eggs and larvae of Frankliniella occidentalis” to use, publish, and disseminate the results of this research.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nguyen, V.H., Nguyen, D.T., Van Leeuwen, T. et al. Life table parameters of Amblyseius largoensis, Amblyseius swirskii and Proprioseiopsis lenis (Acari: Phytoseiidae) fed on eggs and larvae of Frankliniella occidentalis. Exp Appl Acarol (2024). https://doi.org/10.1007/s10493-024-00908-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10493-024-00908-5