Abstract
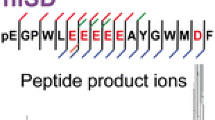
The use of 5-aminosalicylic acid (5-ASA) as a new matrix for in-source decay (ISD) of peptides including mono- and di-phosphorylated peptides in matrix-assisted laser desorption/ionization (MALDI) mass spectrometry (MS) is described. The use of 5-ASA in MALDI-ISD has been evaluated from several standpoints: hydrogen-donating ability, the outstanding sharpness of molecular and fragment ion peaks, and the presence of interference peaks such as metastable peaks and multiply charged ions. The hydrogen-donating ability of several matrices such as α-cyano-4-hydroxycinnamic acid (CHCA), 2,5-dihydroxybenzoic acid (2,5-DHB), 1,5-diaminonaphthalene (1,5-DAN), sinapinic acid (SA), and 5-ASA was evaluated by using the peak abundance of a reduction product [M + 2H + H]+ to that of non-reduced protonated molecule [M + H]+ of the cyclic peptide vasopressin which contains a disulfide bond (S-S). The order of hydrogendonating ability was 1,5-DAN > 5-ASA > 2,5-DHB > SA = CHCA. The chemicals 1,5-DAN and 5-ASA in particular can be classified as reductive matrices. 5-ASA gave peaks with higher sharpness for protonated molecules and fragment ions than other matrices and did not give any interference peaks such as multiply-protonated ions and metastable ions in the ISD mass spectra of the peptides used. Particularly, 1,5-DAN and 5-ASA gave very little metastable peaks. This indicates that 1,5-DAN and 5-ASA are more “cool” than other matrices. The 1,5-DAN and 5-ASA can therefore be termed “reductive cool” matrix. Further, it was confirmed that ISD phenomena such as N-Cα bond cleavage and reduction of S-S bond is a single event in the ion source. The characteristic fragmentations, which form a− and (a + 2)-series ions, [M + H − 15]+, [M + H − 28]+, and [M + H − 44]+ ions in the MALDI-ISD are described.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Karas, M.; Bachmann, D.; Bahr, U.; Hillenkamp, F. Matrix-Assisted Ultraviolet Laser Desorption of Non-Volatile Compounds. Int. J. Mass Spectrom. Ion Processes 1987, 78, 53–68.
Tanaka, K.; Waki, H.; Ido, Y.; Akita, S.; Yoshida, T. Protein and Polymer Analyses up to m/z 100,000 by Laser Ionization Time-of-Flight Mass Spectrometry. Rapid Commun. Mass Spectrom. 1988, 2, 151–153.
Karas, M.; Hillenkamp, F. Laser Desorption Ionization of Proteins with Molecular Masses Exceeding 10,000 Daltons. Anal. Chem. 1988, 60, 2299–2301.
Whitehouse, C. M.; Dreyer, R. N.; Yamashita, M.; Fenn, J. B. Electrospray Interface for Liquid Chromatographs and Mass Spectrometers. Anal. Chem. 1985, 57, 675–679.
Fenn, J. B.; Mann, M.; Meng, C. K.; Wong, S. F.; Whitehouse, C. M. Electrospray Ionization for Mass Spectrometry of Large Biomolecules. Science 1989, 246, 64–71.
Derrick, P.; Patterson, S. Mass Spectrometry of Proteomics. Proteomics 2001, 1, 919–1058.
Abu-Farha, M.; Elisma, F.; Zhou, H.; Tian, R.; Zhou, H.; Asmer, M. S.; Figeys, D. Proteomics: From Technology Developments to Biological Applications. Anal. Chem. 2009, 81, 4585–4599.
Zubarev, R. A.; Kelleher, N. L.; McLafferty, F. W. Electron Capture Dissociation of Multiply Charged Protein Cations. A Nonergodic Process. J. Am. Chem. Soc. 1998, 120, 3265–3266.
Syka, J. E.; Coon, J. J.; Schroeder, M. L.; Shabanowitz, J.; Hunt, D. F. Peptide and Protein Sequence Analysis by Electron Transfer Dissociation Mass Spectrometry. Proc. Natl. Acad. Sci. U.S.A. 2004, 101, 9528–9533.
Brown, R. S.; Lennon, J. Sequence-Specific Fragmentation of Matrix-Assisted Laser-Desorbed Protein/Peptide Ions. J. Anal. Chem. 1995, 67, 3990–3999.
Coon, J. J. Collisions or Electrons? Protein Sequence Analysis in the 21st Century. Anal. Chem. 2009, 81, 3208–3215.
Biemann, K. Contributions of Mass Spectrometry to Peptide and Protein Structure. Biomed. Environ. Mass Spectrom. 1988, 16, 99–111.
Takayama, M. N-Cα Bond Cleavage of the Peptide Backbone Via Hydrogen Abstraction. J. Am. Soc. Mass Spectrom. 2001, 12, 1044–1049.
Koecher, T.; Engstroem, A.; Zubarev, R. A. Fragmentation of Peptides in MALDI In-Source Decay Mediated by Hydrogen Radicals. Anal. Chem. 2005, 77, 172–177.
Hardouin, J. Protein Sequence Information by Matrix-Assisted Laser Desorption/Ionization In-Source Decay Mass Spectrometry. Mass Spectrom. Rev. 2007, 26, 672–682.
Takayama, M. In-source Decay Characteristics of Peptides in Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2001, 12, 420–427.
Fukuyama, Y.; Iwamoto, S.; Tanaka, K. Rapid Sequencing and Disulfide Mapping of Peptides Containing Disulfide Bonds by Using 1,5-Diaminonaphthalene as a Reductive Matrix. J. Mass Spectrom. 2006, 41, 191–201.
Demeure, K.; Quinton, L.; Gabelica, V.; De Pauw, E. Rational Selection of the Optimum MALDI Matrix for Top-Down Proteomics by In-Source Decay. Anal. Chem. 2007, 79, 8678–8685.
See the pollution information site, http://www.scorecard.org/cehmicalprofiles/.
Soltwisch, J.; Souady, J.; Berkenkamp, S.; Dreisewerd, K. Effect of Gas Pressure and Gas Type on the Fragmentation of Peptide and Oligosaccharide Ions Generated in an Elevated Pressure UV/IR-MALDI Ion Source Coupled to an Orthogonal Time-of-Flight Mass Spectrometer. Anal. Chem. 2009, 81, 2921–2934.
Dreisewerd, K. The Desorption Process in MALDI. Chem. Rev. 2003, 103, 395–425.
O’Connor, P. B.; Hillenkamp, F. MALDI MS—A Practical Guide to Instrumentation, Methods, and Applications; Wiley-VCH Verlag GmbH and KGaA: Weinheim, 2007; p. 35–38.
Takayama, M.; Tsugita, A. Does In-Source Decay Occur Independent of the Ionization Process in Matrix-Assisted Laser Desorption? Int. J. Mass Spectrom. Ion Processes 1998, 181, L1-L6.
Kinumi, T.; Niwa, H.; Matsumoto, H. Phosphopeptide Sequencing by In-Source Decay Spectrum in Delayed Extraction Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry. Anal. Biochem. 2000, 277, 177–186.
Takayama, M.; Tsugita, A. Sequence Information of Peptides and Proteins with In-Source Decay in Matrix Assisted Laser Desorption/ Ionization-Time of Flight-Mass Spectrometry. Electrophoresis 2000, 21, 1670–1677.
Katta, V.; Chow, D. T.; Rohde, M. F. Applications of In-Source Fragmentation of Protein Ions for Direct Sequence Analysis by Delayed Extraction MALDI-TOF Mass Spectrometry. Anal. Chem. 1998, 70, 4410–4416.
Salazopyrin, S. N. A New Sulfanilamide Preparation: A. Therapeutic Results in Rheumatic Polyarthritis. B. Therapeutic Results in Ulcerative Colitis. C. Toxic Manifestations in Treatment with Sulfanilamide Preparation (2nd ed.). Acta Med. Scand. 1942, 110, 557–590.
Detlev, C. Y.; Sauerland, V.; Ronk, M.; Ma, M. Toward Top-Down Determination of PEGylation Site Using MALDI In-Source Decay MS Analysis. J. Am. Soc. Mass Spectrom. 2009, 20, 326–333.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakakura, M., Takayama, M. In-source decay and fragmentation characteristics of peptides using 5-aminosalicylic acid as a matrix in matrix-assisted laser desorption/ionization mass spectrometry. J Am Soc Mass Spectrom 21, 979–988 (2010). https://doi.org/10.1016/j.jasms.2010.01.018
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2010.01.018