Abstract
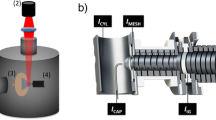
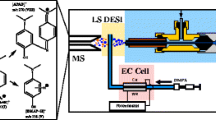
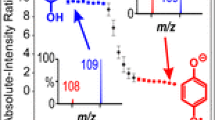
We report here the development of a corona discharge (CD) initiated electrochemical (EC) electrospray ionization (ESI) technique using a standard electrospray ion source. This is a new ionization technique distinct from ESI, electrochemistry inherent to ESI, APCI, and techniques using hydroxyl radicals produced under atmospheric pressure conditions. By maximizing the observable CD at the tip of a stainless steel ESI capillary, efficient electrochemical oxidation of electrochemically active compounds is observed. For electrochemical oxidation to be observed, the ionization potential of the analyte must be lower than Fe. Ferrocene labeled compounds were chosen as the electrochemically active moiety. The electrochemical cell in the ESI source was robust, and generated ions with selectivity according to the ionization potential of the analytes and up to zeptomolar sensitivity. Our results indicate that CD initiated electrochemical ionization has the potential to become a powerful technique to increase the dynamic range, sensitivity, and selectivity of ESI experiments.
Article PDF
Similar content being viewed by others
References
Chang, J. S.; Lawless, P. A.; Yamamoto, T. Corona Discharge Processes. IEEE Trans. Plasma Sci. 1991, 19, 1152–1166.
Druyvesteyn, M. J.; Penning, F. M. The Mechanism of Electrical Discharges in Gases of Low Pressure. Rev. Mod. Phys. 1940, 12, 87–174.
Carroll, D. I.; Dzidic, I.; Stillwell, R. N.; Haegle, K. D.; Horning, E. C. Atmospheric Pressure Ionization Mass Spectrometry: Corona Discharge Ion Sources for Use in Liquid Chromatograph-Mass Spectrometer Computer Analytical System. Anal. Chem. 1975, 47, 2369–2373.
Van Berkel, G. J. The Electrolytic Nature of Electrospray. In Electrospray Ionization Mass Spectrometry, Cole, R. B., Ed.; Wiley: New York, 1997; pp 65–105.
Bruins, A. P. ESI Source Design and Dynamic Range Considerations. In Electrospray Ionization Mass Spectrometry, Cole, R. B., Ed.; Wiley: New York, 1997; pp 114–115.
Hail, M.; Mylchreest, I. U.S. Patent 1995, 5393975.
Uhm, H. S.; Lee, W. M. An Analytical Theory of Discharge Plasmas. Phys. Plasmas. 1997, 4, 3117–3128.
Gao, L.; Song, Q.; Noll, R. J.; Duncan, J.; Cooks, R. G.; Ouyang, Z. Glow Discharge Electron Impact Ionization Source for Miniature Mass Spectrometers. J. Mass Spectrom. 2007, 42, 675–680.
Higashi, T.; Takayama, N.; Nishio, T.; Taniguchi, E.; Shimada, K. Procedure for Increasing the Detection Responses of Estrogens in LC-MS Based on Introduction of a Nitrobenzene Moiety Followed by Electron Capture Atmospheric Pressure Chemical Ionization. Anal. Bioanal. Chem. 2006, 386, 658–665.
Van Berkel, G. J.; Kertesz, V. Using the Electrochemistry of the Electrospray Ion Source. Anal. Chem. 2007, 79, 5510–5520.
De la Mora, J. F.; Van Berkel, G. J.; Enke, C. G.; Cole, R. B.; Martinez-Sanchez, M.; Fenn, J. B. Electrochemical Processes in Electrospray Ionization Mass Spectrometry. J. Mass Spectrom. 2000, 35, 939–952.
Van Berkel, G. J.; Quirke, J. M. E.; Adams, C. L. Derivatization for Electrospray Ionization-Mass Spectrometry: 4. Alkenes and Alkynes. Rapid Commun. Mass Spectrom. 2000, 14, 849–858.
Kertesz, V.; Van Berkel, G. J. Expanded use of a Battery-Powered Two-Electrode Emitter Cell for Electrospray Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2006, 17, 953–961.
Van Berkel, G. J.; Kertesz, V.; Ford, M. J.; Granger, M. C. Efficient Analyte Oxidation in an Electrospray Ion Source Using a Porous Flow-Through Electrode emitter. J. Am. Soc. Mass Spectrom. 2004, 15(12), 1755–1766.
Van Berkel, G. J.; Kertesz, V. Expanded Electrochemical Capabilities of the Electrospray Ion Source Using Porous Flow-Through Electrodes as the Upstream Ground and Emitter High-Voltage Contact. Anal. Chem. 2005, 77, 8041–8049.
Van Berkel, G. J. Insights into Analyte Electrolysis in an Electrospray Emitter from Chronopotentiometry Experiments and Mass Transport Calculations. J. Am. Soc. Mass Spectrom. 2000, 11, 951–960.
Williams, D.; Young, M. K. Analysis of Neutral Isomeric Low Molecular Weight Carbohydrates Using Ferrocenyl Boronate Derivatization and Tandem Electrospray Mass Spectrometry. Rapid Commun. Mass Spectrom. 2000, 14, 2083–2091.
Williams, D.; Chen, S.; Young, M. K. Radiometric Analysis of the Ferrocene Boronate Esters of 2- and 4-Hydroxyestradiol by Tandem Electrospray Mass Spectrometry. Rapid Commun. Mass Spectrom. 2000, 15, 182–186.
Diehl, G.; Karst, U. On-Line Electrochemistry-MS and Related Techniques. Anal. Bioanal. Chem. 2002, 373(6), 390–398.
Meyer, J.; Liesener, A.; Götz, S.; Hayen, H.; Karst, U. Liquid Chromatography with On-Line Electrochemical Derivatization and Fluorescence Detection for the Determination of Phenols. Anal. Chem. 2003, 75(4), 922–926.
Seiwert, B.; Henneken, H.; Karst, U. Ferrocenoyl Piperazide as Derivatizing Agent for the Analysis of Isocyanates and Related Compounds Using Liquid Chromatography/Electrochemistry/Mass Spectrometry (LC/EC/MS). J. Am. Soc. Mass Spectrom. 2004, 15(12), 1727–1736.
Seiwert, B.; Karst, U. Analysis of Cysteine-Containing Proteins Using Precolumn Derivatization with N-(2-Ferroceneethyl)Maleimide and Liquid Chromatography/Electrochemistry/Mass Spectrometry. Anal. Bioanal. Chem. 2007, 388(8), 1633–1642.
Seiwert, B.; Karst, U. Simultaneous LC/MS/MS Determination of Thiols and Disulfides in Urine Samples Based on Differential Labeling with Ferrocene-Based Maleimides. Anal. Chem. 2007, 79(18), 7131–7138.
Karst, U. Electrochemistry/Mass Spectrometry (EC/MS)—a New Tool to Study Drug Metabolism and Reaction Mechanisms. Angew. Chem. Int. Ed. 2004, 43, 2476–2478.
Seiwert, B.; Karst, U. Analysis of Cysteine-Containing Proteins Using Precolumn Derivatization with N-(2-Ferroceneethyl)Maleimide and Liquid Chromatography/Electrochemistry/Mass Spectrometry. Anal. Bioanal. Chem. 2007, 388, 1633–1642.
Seiwert, B.; Karst, U. Ferrocene-Based Derivatization in Analytical Chemistry. Anal. Bioanal. Chem. 2008, 390, 181–200.
Lo, K. K.-W.; Lau, J. S.-Y.; Ng, D. C.-M.; Zhu, N. Specific Labeling of Sulfhydryl-Containing Biomolecules with Redox-Active N-(Ferrocenyl)Iodoacetamide. J. Chem. Soc. Dalton Trans. 2002, 8, 1753–1756.
Bajic, S. U.S. Patent 1996, 5756994.
Kebarle, P.; Tang, L. From Ions in Solution to Ions in the Gas Phase—the Mechanism of Electrospray Mass Spectrometry. Anal. Chem. 1993, 65, 972A-986A.
Kebarle, P.; Ho, Y. On the Mechanism of Electrospray Mass Spectrometry. In Electrospray Ionization Mass Spectrometry, Cole, R. B., Ed.; Wiley: New York, 1997; pp 3–63.
Kebarle, P.; Peschke, M. On the Mechanisms by which the Charged Droplets Produced by Electrospray Lead to Gas Phase Ions. Anal. Chim. Acta. 2000, 406, 11–35.
Kebarle, P. A brief overview of the present status of the mechanisms involved in electrospray mass spectrometry. J. Mass Spectrom. 2000, 35, 804–817.
Fenn, J. Electrospray Wings for Molecular Elephants (Nobel Lecture). Angew. Chem. Int. Ed. Engl. 2003, 42, 3871–3894.
Chen, J.; Davidson, J. H. Model of the Negative DC Corona Plasma: Comparison to the Positive DC Corona Plasma. Plasma Chem. Plasma Processing. 2002, 22, 199–224.
Trichel, G. W. The Mechanism of the Positive Point-to Plain Corona in Air at Atmospheric Pressure. Phys. Rev. 1939, 55, 382–390.
Morrow, R. The Theory of Positive Glow Corona. J. Phys. D Appl. Phys. 1997, 30, 3099–3114.
Maleknia, S. D.; Downard, K. Radical Approaches to Probe Protein Structure, Folding, and Interactions by Mass Spectrometry. Mass Spectrom. Rev. 2001, 20, 388–401.
Maleknia, S. D.; Chance, M. R.; Downard, K. M. Electrospray-Assisted Modification of Proteins: A Radical Probe of Protein Structure. Rapid Commun. Mass Spectrom. 1999, 13, 2352–2358.
Maleknia, S. D.; Chance, M. R.; Downard, K. M. Electrospray Generated Oxygenated Radicals to Probe Protein Structure. In Advances in Mass Spectrometry, Gelpi, E., Ed.; Wiley: 2001; pp 543–544.
Asakawa, Y.; Tokida, N.; Ozawa, C.; Ishiba, M.; Tagaya, O.; Asakawa, N. Suppression Effects of Carbonate on the Interaction Between Stainless Steel and Phosphate Groups of Phosphate Compounds in High-Performance Liquid Chromatography and Electrospray Ionization Mass Spectrometry. J. Chromatogr. A. 2008, 1198/1199, 80–86.
Tang, K.; Gomez, A. Generation of Monodisperse Water Droplets from Electrospray in a Corona-Assisted Cone-Jet Mode. J. Colloid Interface Sci. 1995, 175, 326–332.
Kertesz, V.; Van Berkel, G. J.; Granger, M. C. Efficient Analyte Oxidation in an Electrospray Ion Source Using a Porous Flow-Through Electrode Emitter. Anal. Chem. 2005, 77, 4366–4373.
Van Berkel, G. J.; Asano, K. G.; Granger, M. C. Controlling Analyte Electrochemistry in Electrospray Using a Three-Electrode Emitter Cell. Anal. Chem. 2004, 76, 1493–1499.
Seiwert, B.; Hayen, H.; Karst, U. Differential Labeling of Free and Disulfide-Bound Thiol Functions in Proteins. J. Am. Soc. Mass Spectrom. 2008, 19(1), 1–7.
Gygi, S. P.; Rist, B.; Gerber, S. A.; Turecek, F.; Gelb, M. H.; Aebersold, R. Quantitative Analysis of Complex Protein Mixtures Using Isotope-Coded Affinity Tags. Nat. Biotechnol. 1999, 17(10), 994–999.
Kraatz, H. B.; Galka, M. Ferrocenoyl Amino Acids and Peptides: Probing Peptide. Metal Ions Biol. Syst. 2001, 38, 385–409.
Van Staveren, D. R.; Metzler-Nolte, N. Bioorganometallic Chemistry of Ferrocene. Chem. Rev. 2004, 104, 5931–5985.
Parsons, R. Handbook of Electrochemical Constants; Academic Press: London, 1959; pp 1–110.
Levin, R. D.; Lias, S. G. Ionization Potential and Appearance Potential Measurements; 1971–1981, NSRDS—NBS 71 U.S. Department of Commerce, National Bureau of Standards: Washington, DC, 1982.
Grishina, A. D.; Vannikov, A. V.; Khazova, G. O.; Teodoradze, M. G.; Koltsov, Y. I. Photochemical Processes in Photoresists Containing Electron Donor Molecules. Photochem. Photobiol. 1998, 114, 159–162.
Hsieh, Y. APPI: A New Ionization Source for LC and MS/MS Assays. In Using Mass Spectrometry for Drug Metabolism Studies, Korfmacher W. A., Ed.; CRC Press: 2004; pp 253*2-276.
Takáts, Z.; Wiseman, J. M.; Gologan, B.; Cooks, R. G. Mass Spectrometry Sampling Under Ambient Conditions with Desorption Electrospray Ionization. Science. 2004, 306(5695), 471–473.
Venter, A.; Nefliu, M.; Cooks, R. G. Ambient Desorption Ionization Mass Spectrometry. Trends Anal. Chem. 2008, 27(4), 284–290.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online August 12, 2009
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Lloyd, J.R., Hess, S. A corona discharge initiated electrochemical electrospray ionization technique. J Am Soc Mass Spectrom 20, 1988–1996 (2009). https://doi.org/10.1016/j.jasms.2009.07.021
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2009.07.021