Abstract
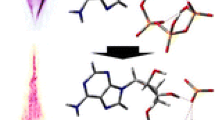
Aqueous solutions of dichloro(ethylenediamine)palladium(II) were investigated using electrospray mass spectrometry (ESMS). The most abundant peak (m/z 436.8) was attributed to the dimeric Pd(en)Cl2·Pd(en)Cl+ ion. We conjecture that the structures of the observed ions arise from the clustering of the hydrolysis products of the parent compound. This hypothesis was tested experimentally by carrying out a series of collision-induced dissociation (CID) experiments and deuterium exchange reactions. It was also assessed by performing density functional theory (DFT) calculations, from which optimized structures and reaction energetics were obtained. These results were compared with our earlier ESMS study of an aqueous Pd(en)Br2 solution. Calculations were also carried out on the Pd(en)Br2 system to facilitate the comparisons. Conclusions are drawn regarding the species present in the two aqueous solutions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fuertes, M. A.; Alonso, C.; Perez, J. M. Biochemical Modulation of Cisplatin Mechanisms of Action: Enhancement of Antitumor Activity and Circumvention of Drug Resistance. Chem. Rev. 2003, 103, 645–662.
Kasparkova, J.; Zehnulova, J.; Farrell, N.; Brabec, V. DNA Interstrand Cross-links of the Novel Antitumor Trinuclear Platinum Complex BBR3464: Conformation, Recognition by High Mobility Group Domain Proteins, and Nucleotide Excision Repair. J. Biol. Chem. 2002, 277, 48076–48086.
Rosenberg, B.; Van Camp, L.; Trosko, J. R.; Mansour, V. A. Platinum Compounds: A New Class of Potent Antitumor Agents. Nature. 1969, 222, 385.
Milovic, M. N.; Kostic, N. M. Palladium(II) Complexes as a Sequence-Specific Peptidase: Hydrolytic Cleavage Under Mild Conditions of X-Pro Peptide Bonds in X-Pro-Met and X-Pro-Met His Segments. J. Am. Chem. Soc. 2003, 125, 781–788.
Zhu, L.; Kostic, N. M. Toward Artificial Metallopeptidases: Mechanisms by which Platinum(II) and Palladium(II) Complexes Promote Selective, Fast Hydrolysis of Unactivated Amide Bonds in Peptides. Inorg. Chem. 1992, 31, 3944–4001.
Pettit, L. D.; Bezer, M. Complex Formation Between Palladium(II) and Amino Acids, Peptides, and Related Ligands. Coord. Chem. Rev. 1985, 61, 97–114.
Appleton, T. G.; Bailey, A. J.; Bedgood, J.; Danny, R.; Hall, J. R. Amino Acid Complexes of Palladium(II). 1: NMR Study of Reactions of the Diaqua(Ethylenediamine) Palladium(II) Cation with Ammonia, Betaine, and the Amino Acids +H3N(CH2)nCO −2 (n = 1–3). Inorg. Chem. 1994, 33, 217–226.
Wyatt, K. S.; Harrison, K. N.; Jensen, C. M. Release of Platinum from Cysteine Residues Induced by N,S-Donor Chelation. Inorg. Chem. 1992, 31, 3867–3868.
Martin, R. B. Platinum Complexes: Hydrolysis and Binding to N(7) and N(1) of Purines; Wiley VCH: New York, 1999, pp 183–205.
Djuran, M. I.; Lempers, E. L. M.; Reedijk, J. Reactivity of Chloro- and Aqua(Diethylenetriamine) Platinum(II) Ions with Glutathione, S-Methylglutathione, and Guanosine 5′-Monophosphate in Relation to the Antitumor Activity and Toxicity of Platinum Complexes. Inorg. Chem. 1991, 30, 2648–2652.
Rochon, F. D.; Buculei, V. Multinuclear Magnetic Resonance Spectroscopy and Crystal Structures of Iodo-Bridged Dinuclear Pt(II) Complexes with Amines. Inorg. Chim. Acta. 2005, 358, 3919–3926.
Over, D.; Bertho, D. G. Fixing the Conformations of Diamineplatinum(II)-GpG Chelates: NMR and CD Signatures of Individual Rotamers. J. Biol. Inorg. Chem. 2006, 11, 139–152.
Tsierkezos, N. G.; Schröder, D.; Schwarz, H. Complexation of Nickel(II) by Ethylenediamine Investigated by Means of Electrospray Ionization Mass Spectrometry. Int. J. Mass Spectrom. 2004, 235, 33–42.
Tercero-Moreno, J. M.; Matilla-Hernández, A.; Gonzáles-Garcia, S.; Niclós-Gutiérrez, J. Hydrolytic Species of the Ion Cis-Diaqua(Ethylenediamine) Palladium(II) Complex and of Cis-Dichloro(Ethylenediamine) Palladium (II): Fitting Its Equilibrium Models in Aqueous Media with or without Chloride Ion. Inorg. Chim. Acta. 1996, 253, 23–29.
Giacomelli, A.; Malatesta, F.; Spinetti, M. C. Solution Reactions at Palladium(II) Complexes Producing Dimeric Species: Stability Constants Determination for Simultaneous Equilibria. Inorg. Chim. Acta. 1981, 51, 55–60.
Anderegg, G. The Stability of the Palladium(II) Complexes with Ethylenediamine, Diethylenetriamine, and Tris(β-Aminoethyl)-Amine. Inorg. Chim. Acta. 1986, 111, 25–30.
Tercero, J. M.; Matilla, A.; Sanjuán, M. A.; Moreno, C. F.; Martin, J. D.; Walmsley, J. A. Synthesis, Characterization, Solution Equilibria, and DNA Binding of Some Mixed-Ligand Palladium(II) Complexes: Thermodynamic Models for Carboplatin Drug and Analogues Compounds. Inorg. Chim. Acta. 2003, 342, 77–87.
Henderson, W.; Sabat, M. Platinum(II)- and Palladium(II)-Amide Complexes [M{NC(O)CH2CH2CH2}2L2] Derived from 2-Azetidinone (β-Propiolactam); a Synthetic, Electrospray Mass Spectrometric and X-Ray Crystallographic Study. Polyhedron. 1997, 16, 1663–1677.
Vrkic, A. K.; O’Hair, R. A. J. Gas Phase Ion Chemistry of Para Substituted Benzene Diazonium Ions, Their Salt Clusters, and Their Related Phenyl Cations. Int. J. Mass Spectrom. 2002, 218, 131–160.
Bach, S. B. H.; Sepeda, T. G.; Merrill, G. N.; Walmsley, J. Complexes of Dibromo (Ethylenediamine) Palladium (II) Observed from Aqueous Solutions by Electrospray Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2005, 16, 1461–1469.
Henderson, W.; Nicholson, B. K.; McCaffrey, L. J. Applications of Electrospray Mass Spectrometry in Organometallic Chemistry. Polyhedron. 1998, 17, 4291–4313.
Stypinski-Mis, B.; Anderegg, G. The Stability of Palladium(II) Complexes with Sulfur-Containing Ligands. Anal. Chim. Acta. 2000, 406, 325–332.
Pankratov, A. N.; Borodulin, V. B.; Chaplygina, O. A. A Quantum Chemical Consideration of Ligand Exchange in Palladium(II) Aqueous and Chloride Complexes. J. Coord. Chem. 2004, 57, 833–842.
Becke, A. D. Density-Functional Exchange-Energy Approximation with Correct Asymptotic Behavior. Phys. Rev. 1988, A38, 3098–3100.
Lee, C.; Yang, W.; Parr, R. G. Development of the Collesalvetti Correlation-Energy Formula into a Functional of the Electron Density. Phy. Rev. 1988, B37, 785–789.
Becke, A. D. A New Mixing of Hartree-Fock and Local-Density-Functional Theories. J. Chem. Phys. 1993, 98, 1372–1377.
Stevens, W. J.; Basch, H.; Krauss, M. J. Compact Effective Potentials and Efficient Shared-Exponent Basis Sets for the First- and Second-Row Atoms. J. Chem. Phys. 1984, 81, 6026–6033.
Stevens, W. J.; Krauss, M. J.; Basch, H.; Jasien, P. G. Relativistic Compact Effective Potentials and Efficient, Shared-Exponent Basis Sets for the Third-, Fourth-, and Fifth-Row Atoms. Can. J. Chem. 1992, 70, 612–630.
Cundari, T. R.; Stevens, W. J. Effective Core Potential Methods for the Lanthanides. J. Chem. Phys. 1993, 98, 5555–5565.
Hariharan, P. C.; Pople, J. A. The Influence of Polarization Functions on Molecular Orbital Hydrogenation Energies. Theor. Chim. Acta. 1973, 28, 213–222.
Schmidt, M. W.; Baldridge, K. K.; Boatz, J. A.; Elbert, S. T.; Gordon, M. S.; Jensen, J. H.; Koseki, S.; Matsunaga, N.; Nguyen, K. A.; Su, S.; Windus, T. L.; Montgomery, J.; Dupuis, M. General Atomic and Molecular Electronic Structure System. J. Comput. Chem. 1993, 14, 1347–1363.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online February 20, 2007
Rights and permissions
About this article
Cite this article
Bach, S.B.H., Green, C.E., Nagore, L.I. et al. Complexes of dichloro(ethylenediamine)palladium(II) observed from aqueous solutions by electrospray mass spectrometry. J Am Soc Mass Spectrom 18, 769–777 (2007). https://doi.org/10.1016/j.jasms.2006.12.013
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2006.12.013