Abstract
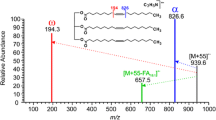
The application of multiple-stage ion-trap (IT) mass spectrometric methods for the structural characterization of cardiolipin (CL), a 1,3-bisphosphatidyl-sn-glycerol that consists of four fatty acyl chains and three glycerol backbones (designated as A, B, and central glycerol, respectively), as the sodiated adduct ions in the positive-ion mode was evaluated. Following collisionally activated dissociation (CAD), the [M−2H+3Na]+ ions of CL yield two prominent fragment ion pairs that consist of the phosphatidyl moieties attached to the 1′- and 3′-position of the central glycerol, respectively, resulting from the differential losses of the diacylglycerol moieties containing A and B glycerol, respectively. The results are consistent with those previously described for the [M−H]− and [M−2H+Na]− ions in the negative-ion mode, thus permitting assignment of the two phosphatidyl moieties attached to the 1′-or 3′-position of the central glycerol. The identities of the fatty acyl substituents and their positions on the glycerol backbones (glycerol A and B) are deduced from further degradation of the above ion pairs that give the fragment ions reflecting the fatty acid substituents at the sn-1 (or sn-1′) and sn-2 (or sn-2′) positions. The ions that arise from losses of the fatty acid substituents at sn-1 and sn-1′, respectively, are prominent, but the analogous ions from losses of the fatty acid substituents at sn-2 and sn-2′, respectively, are of low abundance in the MS2 product-ion spectra. This feature further confirms the assignment of the positions of the fatty acid substituents. The similar IT multiple-stage mass spectrometric approaches including MS2 and MS3 for structural characterization of CL using its [M+Na]+ and the [M−H+2Na]+ ions are also readily applicable. However, their uses for structural characterization are less desirable because formation of the [M+Na]+ and the [M−H+2Na]+ ions for CL is not predictable.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
LeCocq, J.; Ballou, C. E. On the Structure of Cardiolipin. Biochemistry 1964, 155, 976–980.
Powell, G. L.; Jacobus, J. The Nonequivalence of the Phosphorus Atoms in Cardiolipin. Biochemistry 1974, 13, 4024–4026.
Schlame, M.; Brody, S.; Hostetler, K. Y. Mitochondrial Cardiolipin in Diverse Eukaryotes. Comparison of Biosynthetic Reactions and Molecular Acyl Species. Eur. J. Biochem 1993, 212, 727–735.
Schlame, M.; Otten, D. Analysis of Cardiolipin Molecular Species by High-Performance Liquid Chromatography of Its Derivative 1,3-Bisphosphatidyl-2-Benzoyl-sn-Glycerol Dimethyl Ester. Anal. Biochem 1991, 195, 290–295.
Schlame, M.; Ren, M.; Xu, Y.; Greenberg, M. L.; Haller, I. Molecular Symmetry in Mitochondrial Cardiolipins. Chem. Phys. Lipids 2005, 138, 38–49.
Valianpour, F.; Wanders, R. J. A.; Barth, P. G.; Overmars, H.; van Gennip, A. H. Quantitative and Compositional Study of Cardiolipin in Platelets by Electrospray Ionization Mass Spectrometry: Application for the Identification of Barth Syndrome Patients. Clin. Chem 2002, 48, 1390–1397.
Beckedorf, A. I.; Schaffer, C.; Messner, P.; Peter-Katalinic, J. Mapping and Sequencing of Cardiolipins from Geobacillus stearothermophilus NRS 2004/3a by Positive and Negative Ion Nano-ESI-QTOF-MS and MS/MS. J. Mass Spectrom 2002, 37, 1086–1094.
Lesnefsky, E. S.; Stoll, M. S. K.; Minkler, P. E.; Hoppel, C. L. Separation and Quantitation of Phospholipids and Lysophospholipids by High-Performance Liquid Chromatography. Anal. Biochem 2000, 285, 246–254.
Peter-Katalinic, J.; Fischer, W. α-D-Glucopyranosyl-, D-Alanyl-, and L-Lysylcardiolipin from Gram-Positive Bacteria: Analysis by Fast Bombardment Mass Spectrometry. J. Lipid Res 1998, 39, 2286–2292.
Hsu, F.-F.; Turk, J.; Rhoades, E. R.; Russell, D. G.; Shi, Y.; Groisman, E. A. Structural Characterization of Cardiolipin by Tandem Quadrupole and Multiple-Stage Quadrupole Ion-Trap Mass Spectrometry with Electrospray Ionization. J. Am. Soc. Mass Spectrom 2005, 16, 491–504.
Hsu, F.-F.; Turk, J. Structural Characterization of Cardiolipin from Escherichia coli by Electrospray Ionization with Multiple Stage Quadrupole Ion-Trap Mass Spectrometric Analysis of [M−2H+Na]− Ions. J. Am. Soc. Mass Spectrom 2006, 17, 420–429.
Hsu, F.-F.; Turk, J. Electrospray Ionization/Tandem Quadrupole Mass Spectrometric Studies on Phosphatidylcholines: The Fragmentation Processes. J. Am. Soc. Mass Spectrom 2003, 14, 352–363.
Hsu, F.-F.; Turk, J. Structural Studies on Phosphatidylserine by Tandem Quadrupole and Multiple Stage Quadrupole Ion-Trap Mass Spectrometry with Electrospray Ionization. J. Am. Soc. Mass Spectrom 2005, 16, 1510–1522.
Hsu, F.-F.; Turk, J. Structural Characterization of Triacylglycerols as Lithiated Adducts by Electrospray Ionization Mass Spectrometry Using Low-Energy Collisionally Activated Dissociation on a Triple Stage Quadrupole Instrument. J. Am. Soc. Mass Spectrom 1999, 10, 587–599.
Hsu, F.-F.; Turk, J. Electrospray Ionization with Low-Energy Collisionally Activated Dissociation Tandem Mass Spectrometry of Complex Lipids: Structural Characterization and Mechanisms of Fragmentation In Modern Methods for Lipid Analysis by Liquid Chromatography/Mass Spectrometry; Byrdwell, W.C., Ed.; AOCS Publication: Champaign, IL, 2005; pp 61–178.
Henderson, T. O.; Glonek, T.; Myers, T. C. Phosphorus-31 Nuclear Magnetic Resonance Spectroscopy of Phospholipids. Biochemistry 1974, 13, 623–628.
Kates, M.; Syz, J. Y.; Gosser, D.; Haines, T. H. pH-Dissociation Characteristics of Cardiolipin and Its 2′-Deoxy Analog. Lipids 1993, 28, 877–882.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online June 5, 2006
Rights and permissions
About this article
Cite this article
Hsu, F.F., Turk, J. Characterization of cardiolipin as the sodiated ions by positive-ion electrospray ionization with multiple stage quadrupole ion-trap mass spectrometry. The official journal of The American Society for Mass Spectrometry 17, 1146–1157 (2006). https://doi.org/10.1016/j.jasms.2006.04.024
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2006.04.024