Abstract
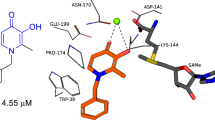
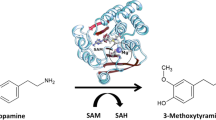
Z-Vallesiachotamine is a monoterpene indole alkaloid that has a β-N-acrylate group in its structure. This class of compounds has already been described in different Psychotria species. Our research group observed that E/Z-vallesiachotamine exhibits a multifunctional feature, being able to inhibit targets related to neurodegeneration, such as monoamine oxidase A, sirtuins 1 and 2, and butyrylcholinesterase enzymes. Aiming at better characterizing the multifunctional profile of this compound, its effect on cathecol-O-methyltransferase activity was investigated. The cathecol-O-methyltransferase activity was evaluated in vitro by a fluorescence-based method, using S-(5′-adenosyl)-l-methionine as methyl donor and aesculetin as substrate. The assay optimization was performed varying the concentrations of methyl donor (S-(5′-adenosyl)-l-methionine) and enzyme. It was observed that the highest concentrations of both factors (2.25 U of the enzyme and 100 ′M of S-(5′-adenosyl)-l-methionine) afforded the more reproducible results. The in vitro assay demonstrated that Z-vallesiachotamine was able to inhibit the cathecol-O-methyltransferase activity with an IC50 close to 200 ′M. Molecular docking studies indicated that Z-vallesiachotamine can bind the catechol pocket of catechol-O-methyltransferase enzyme. The present work demonstrated for the first time the inhibitory properties of Z-vallesiachotamine on cathecol-O-methyltransferase enzyme, affording additional evidence regarding its multifunctional effects in targets related to neurodegenerative diseases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Berger, A., Fasshuber, H., Schinnerl, J., Brecker, L., Greger, H., 2012. Various types of tryptamine-iridoid alkaloids from Palicourea acuminata (=Psychotria acuminata, Rubiaceae). Phytochem. Lett. 5. 558–562.
Bonifácio, M.J., Sutcliffe, J.S., Torrão, L., Wright, L.C., Soares-da-Silva, P., 2014. Brain and peripheral pharmacokinetics of levodopa in the cynomolgus monkey following administration of opicapone, a third generation nitrocatechol COMT inhibitor. Neuropharmacology 77. 334–341.
Brisch, R., Bernstein, H.G., Krell, D., Dobrowonly, H., Bielau, H., Steiner, J., Gos, T., Funke, S., Stauch, R., Knüppel, S., Bogerts, B., 2009. Dopamine–glutamate abnormalities in the frontal cortex associated with the catechol-O-methyltransferase (COMT) in schizophrenia. Brain Res. 1269. 166–175.
Cheng, G.-G., Zhao, Y.-L., Zhang, Y., Lunga, P.-K., Hu, D.-B., Li, Y., Gu, J., Song, C.-W., Sun, W.-B., Liu, Y.-B., Luo, X.-D., 2014. Indole alkaloids from cultivated Vinca major. Tetrahedron 70. 8723–8729.
Dickinson, D., Elvevág, B., 2009. Genes, cognition and brain through a COMT lens. Neuroscience 164. 72–87.
Dillin, A., Kelly, J.W., 2007. The yin-yang of sirtuins. Science 317. 461–462.
Djerassi, C., Monteiro, H.J., Walser, A., Durham, L.J., 1966. Alkaloids studies. LVI. The constitution of vallesiachotamine. J. Am. Chem. Soc. 88. 1792–1798.
Donmez, G., Outeiro, T.F., 2013. SIRT1 and SIRT2: emerging targets in neurodegeneration. EMBO Mol. Med. 5. 344–352.
Evans, D.A., Joule, J.A., Smith, G.F., 1968. The alkaloids of Rhazya orientalis. Phytochemistry 7. 1429–1431.
Green, K.N., Steffan, J.S., Martinez-Coria, H., Sun, X., Schreiber, S.S., Thompson, L.M., Laferla, F.M., 2008. Nicotinamide restores cognition in Alzheimer’s disease transgenic mice via a mechanism involving sirtuin inhibition and selective reduction of Thr231-Phosphotau. J. Neurosci. 28. 11500–11510.
Hamaue, N., Ogata, A., Terado, M., Tsuchida, S., Yabe, I., Sasaki, H., Hirafuji, M., Togashi, H., Aoki, T., 2010. Entacapone, a catechol-O-methyltransferase inhibitor, improves the motor activity and dopamine content of basal ganglia in a rat model of Parkinson’s disease induced by Japanese encephalitis virus. Brain Res. 1309. 110–115.
Heitzman, M.E., Neto, C.C., Winiarz, E., Vaisberg, A.J., Hammond, G.B., 2005. Ethnobotany, phytochemistry and pharmacology of Uncaria (Rubiaceae). Phytochemistry 66. 5–29.
Jatana, N., Sharma, A., Latha, N., 2013. Pharmacophore modeling and virtual screening studies to design potential COMT inhibitors as new leads. J. Mol. Graph. Model. 39. 145–164.
Kastner, R.S., Van Os, J., Steinbusch, H., Schimtz, C., 2006. Gene regulation by hypoxia and the neurodevelopmental schizophrenia. Schizophr. Res. 84. 253–271.
Klein-Júnior, L.C., Passos, C.S., Moraes, A.P., Wakui, V.G., Konrath, E.L., Nurisso, A., Carrupt, P.-A., Oliveira, C.M.A., Kato, L., Henriques, A.T., 2014. Indole alkaloids and semisynthetic indole derivatives as multifunctional scaffolds aiming the inhibition of enzymes related to neurodegenerative diseases–a focus on Psychotria L. genus. Curr. Top. Med. Chem. 14. 1056–1075.
Kurkela, M., Siiskonen, A., Finel, M., Tammela, P., Taskinen, J., Vuorela, P., 2004. Microplate screening assay to identify inhibitors of human catechol-Omethyltransferase. Anal. Biochem. 331. 198–200.
Mannisto, P.T., Kaakkola, S., 1999. Catechol-O-methyltransferase (COMT): biochemistry, molecular biology, pharmacology, and clinical efficacy of the new selective COMT inhibitors. Pharmacol. Rev. 51. 593–628.
Matsumoto, M., Weickert, C.S., Beltaifa, S., Kolachana, B., Chen, J., Hyde, T.M., Herman, M.M., Weinberger, D.R., Kleinman, J.E., 2003. Catechol-O-methyltransferase (COMT) mRNA expression in the dorsolateral prefrontal cortex of patients with schizophrenia. Neuropsychopharmacology 28. 1521–1530.
Maxwell, M.M., Tomkinson, E.M., Nobles, J., Wizeman, J.W., Amore, A.M., Quinti, L., Chopra, V., Hersch, S.M., Kazantsev, A.G., 2011. The sirtuin 2 microtubule deacetylase is an abundant neuronal protein that accumulates in the aging CNS. Hum. Mol. Genet. 20. 3986–3996.
Passos, C.S., Soldi, T.C., Abib, R.T., Apel, M.A., Simões-Pires, C., Marcourt, L., Gottfried, C., Henriques, A.T., 2013a. Monoamine oxidase inhibition by monoterpene indole alkaloids and fractions from Psychotria suterella and Psychotria laciniata. J. Enzyme Inhib. Med. Chem. 28. 611–618.
Passos, C.S., Simões-Pires, C., Nurisso, A., Soldi, T.C., Kato, L., Oliveira, C.M.A., Faria, E.O., Marcourt, L., Gottfried, C., Carrupt, P.-A., Henriques, A.T., 2013b. Indole alkaloids of Psychotria as multifunctional cholinesterases and monoamine oxidases inhibitors. Phytochemistry 86. 8–20.
Paul, J.H.A., Maxwell, A.R., Reynolds, W.F., 2003. Novel Bis(monoterpenoid) indole alkaloids from Psychotria bahiensis. J. Nat. Prod. 66. 752–754.
Rutherford, K., Le Trong, I., Stenkamp, R.E., Parson, W.W., 2008. Crystal structures of human 108 V and 108 M catechol-O-methyltransferase. J. Mol. Biol. 380. 120–130.
Rybakowski, J.K., Borkowska, A., Czerski, P.M., Dmitrzak-Weglarz, M., Skibinska, M., Kapelski, P., Hauser, P., 2006. Performance on the Wisconsin Card Sorting Test in schizophrenia and genes of dopaminergic inactivation (COMT, DAT, NET). Psychiat. Res. 143. 13–19.
Sacconnay, L., Ryckewaert, L., Passos, C.S., Guerra, M.C., Kato, L., Oliveira, C.M.A., Henriques, A.T., Carrupt, P.-A., Simões-Pires, C., Nurisso, A., 2015. Alkaloids from Psychotria target sirtuins: in silico and in vitro interaction studies. Planta Med. 81. 517–524.
Shang, J.-H., Cai, X.-H., Feng, T., Zhao, Y.-L., Wang, J.-K., Zhang, L.-Y., Yan, M., Luo, X.-D., 2010. Pharmacological evaluation of Alstonia scholaris: anti-inflammatory and analgesic effects. J. Ethnopharmacol. 129. 174–181.
Shirakawa, T., Abe, M., Oshima, S., Mitome, M., Oguchi, H., 2004. Neuronal expression of the catechol-O-methyltransferase nRNA in neonatal rat suprachiasmatic nucleus. Neuroreport 15. 239–1243.
Soares, P.R.O., Oliveira, P.L., Oliveira, C.M.A., Kato, L., Guillo, L.A., 2012. In vitro antiproliferative effects of the indole alkaloid vallesiachotamine on human melanoma cells. Arch. Pharm. Res. 35. 565–571.
Solis, P.N., Wright, C.W., Gupta, M.P., Phillipson, J.D., 1993. Alkaloids from Cephaelis dichroa. Phytochemistry 33. 1117–1119.
Tiwari, G., Tiwari, R., 2010. Bioanalytical method validation: an update review. Pharm. Methods 1. 25–38.
Yalcin, D., Bayraktar, O., 2010. Inhibition of catechol-O-methyltransferase (COMT) by some plant-derived alkaloids and phenolics. J. Mol. Catal. B: Enzym. 64. 162–166.
Zhong, X.-H., Xiao, L., Wang, Q., Zhang, B.-J., Bao, M.-F., Cai, X.-H., Peng, L., 2014. Cytotoxic 7S-oxindole alkaloids from Gardneria multiflora. Phytochem. Lett. 10. 55–59.
Acknowledgements
This work was supported by CNPq (grant #478496/2011-7) and by FAPERGS (grant #2281-2551/14-2SIAFEM). LCKJ and ATH thank CNPq for the fellowships. JMMA thanks to CAPES for the PhD scholarship.
Author information
Authors and Affiliations
Contributions
CSP (Postdoctoral Researcher) performed the in silico experiments and part of the in vitro method optimization. LCKJ (PhD Student) performed the in vitro experiments and wrote the paper. JMMA (PhD Student) performed part of the in vitro experiments. Both CSP and JMMA contributed to write the manuscript. CM and ATH supervised the experimental work and contributed to the manuscript. All the authors have read the final manuscript and approved the submission.
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
dos Santos Passos, C., Klein-Júnior, L.C., de Mello Andrade, J.M. et al. The catechol-O-methyltransferase inhibitory potential of Z-vallesiachotamine by in silico and in vitro approaches. Rev. Bras. Farmacogn. 25, 382–386 (2015). https://doi.org/10.1016/j.bjp.2015.07.002
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2015.07.002