Abstract
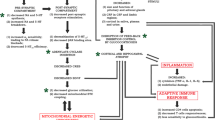
Although depression is a common disorder that is often resistant to pharmacotherapy, its pathophysiology has remained elusive. Since the early 1950s, when the first antidepressants were introduced, i.e., the non-selective MAO inhibitors and tricyclic drugs, a number of hypotheses describing ethiopathogenesis of depression and antidepressant drug action have been formulated. The Institute of Pharmacology, the Polish Academy of Sciences has performed experimental and clinical research focused on the pathophysiology of depression and the mechanisms of action of antidepressant drugs for over 40 years. Our results from this period have significantly contributed to understanding the complex mechanisms of antidepressant drug actions and new pathways that underpin the pathophysiology of depression. Most of these theories are based on the finding that the chronic administration of antidepressants leads to adaptive changes in pre- and post-synaptic monoaminergic and glutamatergic neurotransmission as well as to alterations in gene transcription and immune-inflammatory and neurotrophic factors, resulting in neuroplastic changes in the brain. Taking into account the functional interdependence of the neuronal, hormonal and immunologic systems, we propose neurodevelopmental and neuroimmune theories for affective disorders. Moreover, commonalities have been documented for the pathomechanisms of depression and neurodegenerative and metabolic disorders as well as drug dependence. The aim of this special issue is to briefly present the major research contributions and the new research directions of the Institute of Pharmacology, the Polish Academy of Sciences with respect to the neurobiology of affective disorders and the mechanisms of action of marketed and new putative antidepressant drugs.
Similar content being viewed by others
References
Amsterdam JD, Hornig-Rohan M: Treatment algorithms in treatment-resistant depression. Psychiatry Clin North Am, 1996, 19, 371–386.
Anderson G, Kubera M, Duda W, Lasoñ W, Berk M, Maes M: Increased IL-6 trans-signaling in depression: focus on the tryptophan catabolite pathway, melatonin and neuroprogression. Pharmacol Rep, 2013, 65, 1647–1654.
Carlezon WA Jr, Mague SD, Andersen SL: Enduring behavioral effects of early exposure to methylphenidate in rats. Biol Psychiatry, 2003, 54, 1330–1337.
Chocyk A, Majcher-Maślanka I, Dudys D, Przyborowska A, Wędzony K: Impact of early-life stress on the medial prefrontal cortex functions – a search for the pathomechanisms of anxiety and mood disorders. Pharmacol Rep, 2013, 65, 1462–1470.
Curzytek K, Kubera M, Szczepanik M, Basta-Kaim A, Leśkiewicz M, Budziszewska B, Lasoń W, Maes M: Crosstalk between contact hypersensitivity reaction and antidepressant drugs. Pharmacol Rep, 2013, 65, 1673–1680.
Darras VM: Endocrine disrupting polyhalogenated organic pollutants interfere with thyroid hormone signaling in the developing brain. Cerebellum, 2008, 26–37.
Detka J, Kurek A, Basta-Kaim A, Kubera M, Lasoń W, Budziszewska B: Neuroendocrine link between stress, depression and diabetes. Pharmacol Rep, 2013, 65, 1591–1600.
Doze VA, Handel EM, Jensen KA, Darsie B, Luger EJ, Haselton JR, Talbot JN, Rorabaugh BR: α1A- and α1B-adrenergic receptors differentially modulate antidepres-sant-like behavior in the mouse. Brain Res, 2009, 1285, 148–157.
Faron-Górecka A, Kuśmider M, Solich J, Kolasa M, Szafran K, Żurawek D, Pabian P, Dziedzicka-Wasylewska M: Involvement of prolactin and somatostatin in depression and the mechanism of action of antidepressant drugs. Pharmacol Rep, 2013, 65, 1640–1646.
Filip M, Frankowska M, Jastrzębska J, Wydra K, Przegaliński E: Preclinical studies on comorbidity between depression and psychostimulant addiction. Pharmacol Rep, 2013, 65, 1529–1534.
Gądek-Michalska A, Tadeusz J, Rachwalska P, Bugajski J: Cytokines, prostaglandins and nitric oxide in the regulation of stress-response systems. Pharmacol Rep, 2013, 65, 1655–1662.
Haduch A, Bromek E, Daniel WA: Role of brain cytochrome P450 (CYP2D) in the metabolism of monoaminergic neurotransmitters. Pharmacol Rep, 2013, 65, 1519–1528.
Hoyer S, Lannert H: Long-term effects of corticosterone on behavior, oxidative and energy metabolism of parietotemporal cerebral cortex and hippocampus of rats: comparison to intracerebroventricular streptozotocin. J Neural Transm, 2008, 115, 1241–1249.
Kajta M, Wójtowicz A: Impact of endocrine-disrupting chemicals on neural development and the onset of neurological disorders. Pharmacol Rep, 2013, 65, 1632–1639.
Kreiner G, Chmielarz P, Roman A, Nalepa I: Gender differences in genetic moue models evaluated for depressive-like and antidepressant behavior. Pharmacol Rep, 2013, 65, 1580–1590.
Magariños AM, McEwen BS: Experimental diabetes in rats causes hippocampal dendritic and synaptic reorganization and increased glucocorticoid reactivity to stress. Proc Natl Acad Sci USA, 2000, 97, 11056–11061.
Mika J, Osikowicz M, Rojewska E, Korostynski M, Wawrzczak-Bargiela A, Przewlocki R, Przewlocka B: Differential activation of spinal microglial and astroglial cells in a mouse model of peripheral neuropathic pain. Eur J Pharmacol, 2009, 623, 65–72.
Mika J, Zychowska M, Makuch W, Rojewska E, Przewlocka B: Neuronal and immunological basis of action of antidepressants in chronic pain – clinical and experimental studies. Pharmacol Rep, 2013, 65, 1611–1621.
Nalepa I, Kreiner G, Bielawski A, Rafa-Zabłocka K, Roman A: α-Adrenergic receptor subtypes in the central nervous system: insights from genetically engineered moue models. Pharmacol Rep, 2013, 65, 1489–1497.
Nikiforuk A, Popik P: Neurochemical modulation of stress-induced cognitive inflexibility in a rat model of an attentional set-shifting task. Pharmacol Rep, 2013, 65, 1479–1488.
Ossowska K, Lorenc-Koci E: Depression in Parkinson’s disease. Pharmacol Rep, 2013, 65, 1545–1557.
Pałucha-Poniewiera A, Wierońska JM, Brański P, Burnat G, Chruścicka B, Pilc A: Is the mGlu5 receptor a possible target for new antidepressant drugs? Pharmacol Rep, 2013, 65, 1506–1511.
Pan Y, Hong Y, Zhang QY, Kong LD: Impaired hypothalamic insulin signaling in CUMS rats: restored by icariin and fluoxetine through inhibiting CRF system. Psycho-neuroendocrinology, 2013, 38, 122–134.
Politis M, Wu K, Loane C, Turkheimer FE, Molloy S, Brooks DJ, Piccini P: Depressive symptoms in PD correlate with higher 5-HTT binding in raphe and limbic structures. Neurology, 2010, 75, 1920–1927.
Remy P, Doder M, Lees A, Turjanski N, Brooks D: Depression in Parkinson’s disease: loss of dopamine and noradrenaline innervation in the limbic system. Brain, 2005, 128, 1314–1322.
Rogóż Z: Combined treatment with atypical antipsychotics and antidepressants in treatment-resistant depression: preclinical and clinical efficacy. Pharmacol Rep, 2013, 65, 1535–1544.
Roman A, Kreiner G, Nalepa I: Macrophages and depression – A misalliance or well-arranged marriage? Pharmacol Rep, 2013, 65, 1663–1672.
Siwek M, Sowa-Kućma M, Dudek D, Styczeń K, Szewczyk B, Kotarska K, Misztak P et al.: Oxidative stress markers in affective disorders. Pharmacol Rep, 2013, 65, 1558–1571.
Siwek M, Szewczyk B, Dudek D, Styczeń K, Sowa-Kućma M, Młyniec K, Siwek K et al.: Zinc as a marker of affective disorders. Pharmacol Rep, 2013, 65, 1512–1518.
Skuza G: Ethanol withdrawal-induced depressive symptoms in animals and therapeutic potential of sigma1 receptor ligands. Pharmacol Rep, 2013, 65, 1681–1687.
Szafran K, Faron-Górecka A, Kolasa M, Kuśmider M, Solich J, Żurawek D, Dziedzicka-Wasylewska M: Potential role of G protein-coupled receptor (GPCR) heterodi-merization in neuropsychiatric disorders: Focus on depression. Pharmacol Rep, 2013, 65, 1498–1505.
Szczęsny E, Ślusarczyk J, Głombik K, Budziszewska B, Kubera M, Lasoń W, Basta-Kaim A: Possible contribution of IGF-1 to depressive disorder. Pharmacol Rep, 2013, 65, 1622–1631.
Śmiałowska M, Szewczyk B, Woźniak M, Wawrzak-Wleciał A, Domin H: Glial degeneration as a model of depression. Pharmacol Rep, 2013, 65, 1572–1579.
Varea E, Blasco-Ibáñez JM, Gómez-Climent MA, Castillo-Gómez E, Crespo C, Martínez-Guijarro FJ, Nácher J: Chronic fluoxetine treatment increases the expression of PSA-NCAM in the medial prefrontal cortex. Neuropsychopharmacology, 2007, 32, 803–812.
Varea E, Guirado R, Gilabert-Juan J, Martí U, Castillo-Gomez E, Blasco-Ibáñez JM, Crespo C, Nacher J: Expression of PSA-NCAM and synaptic proteins in the amygdala of psychiatric disorder patients. Psychiatr Res, 2012, 46: 189–197.
Vetulani J: Early maternal separation: a rodent model of depression and a prevailing human condition. Pharmacol Rep, 2013, 65, 1451–1461.
VolkowND: The reality of comorbidity: depression and drug abuse. Review Biol Psychiatry, 2004, 56, 714–717.
Vriend C, Raijmakers P, Veltman DJK, van Dijk KD, van der Werf YD, Foncke EM, Smit JH et al.: Depressive symptoms in Parkinson’s disease are related to reduced [123I]FP-CIT binding in the caudate nucleus. J Neurol Neurosurg Psychiatry, 2013 [Epub ahead of print].
Watkins LR, Hutchinson MR, Johnston IN, Maier SF: Glia: novel counter-regulators of opioid analgesia. Trends Neurosci, 2005, 28, 661–669.
Wędzony K, Chocyk A, Maćkowiak M. Potential roles of NCAM/PSA-NCAM proteins in depression and the mechanism of action of antidepressant drugs. Pharmacol Rep, 2013, 65, 1471–1478.
Zychowska M, Rojewska E, Przewlocka B, Mika J: Mechanisms and pharmacology of diabetic neuropathy – experimental and clinical studies. Pharmacol Rep, 2013, 65, 1601–1610.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lasoń, W., Budziszewska, B., Basta-Kaim, A. et al. New trends in the neurobiology and pharmacology of affective disorders. Pharmacol. Rep 65, 1441–1450 (2013). https://doi.org/10.1016/S1734-1140(13)71504-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/S1734-1140(13)71504-4