Abstract
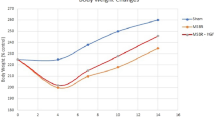
Our society is aging at a rapid rate; the effects of aging on physiologic functions (e.g., small bowel adaptation) are poorly understood. The purpose of this study was to determine the ability of the aged small bowel mucosa to adapt after resection. Young (2-month-old) and aged (24-month-old) F344 rats underwent massive (70%) proximal small bowel resection (SBR) or sham operation; rats were killed at 9 or 16 days after surgery. The remnant small bowel and corresponding sham segments were harvested, weighed, and analyzed for DNA content and villus height. To determine whether the adaptive response after SBR could be enhanced, aged rats underwent SBR or sham operation and were treated with either neurotensin or saline solution (control). SBR resulted in adaptive hyperplasia in the remaining small bowel remnant in both young and aged rats at 9 and 16 days compared with sham animals. At 9 days, significant increases were noted in weight, villus height, and DNA content of the distal remnant in young and aged rats after SBR; the increases were similar in both young and aged rats. At 16 days, both young and aged rats displayed significant increases in remnant weight after SBR. Administration of neurotensin increased the weight of the remnant intestine in aged rats after SBR compared with saline treatment. Our findings demonstrate that aged small bowel mucosa exhibits a proliferative and adaptive capacity in response to SBR that was similar to that of the young animals. In addition, neurotensin administration enhanced the normal adaptive response of the small bowel in aged rats, providing further evidence that neurotensin may be therapeutically useful to augment mucosal regeneration in the early periods after massive SBR.
Similar content being viewed by others
References
Day JC. Population projections of the United States by age, sex, race and Hispanic origin: 1993–2050. In Day JC, ed. Current Population Reports. Washington, DC:U.S. Department of Commerce Bureau of the Census, 1993, 25–1104.
Rosenthal RA, Zenilman ME. Surgery in the elderly. In Townsend CM Jr, Beauchamp RD, Evers BM, Mattox KL, eds. Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice. Philadephia: W.B. Saunders, 2001, pp 226–248.
Holt PR, Yeh KY. Colonic proliferation is increased in senescent rats. Gastroenterology 1988;95:1556–1563.
Holt PR, Yeh KY. Small intestinal crypt cell proliferation rates are increased in senescent rats. J Gerontol 1989;44:B9-B14.
Deschner EE, Godbold J, Lynch HT. Rectal epithelial cell proliferation in a group of young adults. Influence of age and genetic risk for colon cancer. Cancer 1988;61:2286–2290.
Roncucci L, Ponz de Leon M, Scalmati A, Malagoli G, Prat-issoli S, Perini M, Chahin NJ. The influence of age on colonic epithelial cell proliferation. Cancer 1988;62:2373–2377.
Stadler J, Stern HS, Yeung KS, McGuire V, Furrer R, Mar-con N, Bruce WR. Effect of high fat consumption on cell proliferation activity of colorectal mucosa and on soluble faecal bile acids. Gut 1988;29:1326–1331.
Baca I, Feurle GE, Schwab A, Mittmann U, Knauf W, Leh-nert T. Effect of neurotensin on exocrine pancreatic secretion in dogs. Digestion 1982;23:174–183.
Andersson S, Rosell S, Hjelmquist U, Chang D, Folkers K. Inhibition of gastric and intestinal motor activity in dogs by (Gln4) neurotensin. Acta Physiol Scand 1977;100:231–235.
Wood JG, Hoang HD, Bussjaeger LJ, Solomon TE. Neurotensin stimulates growth of small intestine in rats. Am J Physiol 1988;255:G813-G817.
Evers BM, Izukura M, Townsend CM Jr, Uchida T, Thompson JC. Differential effects of gut hormones on pancreatic and intestinal growth during administration of an elemental diet. Ann Surg 1990;211:630–638.
Evers BM, Izukura M, Townsend CM Jr, Uchida T, Thompson JC. Neurotensin prevents intestinal mucosal hy-poplasia in rats fed an elemental diet. Dig Dis Sci 1992;37:426–431.
Izukura M, Parekh D, Evers BM, Yoshinaga K, Greeley GH Jr, Townsend CM Jr. Neurotensin stimulates colon growth in rats. Gastroenterology 1990;98:A416.
Chung DH, Evers BM, Shimoda I, Townsend CM Jr, Ra-jaraman S, Thompson JC. Effect of neurotensin on gut mu-cosal growth in rats with jejunal and ileal Thiry-Vella fistulas. Gastroenterology 1992;103:1254–1259.
Izukura M, Evers BM, Parekh D, Yoshinaga K, Uchida T, Townsend CM Jr, Thompson JC. Neurotensin augments intestinal regeneration after small bowel resection in rats. Ann Surg 1992;215:520–527.
de Miguel E, Gomez de Segura IA, Bonet H, Rodriguez Montes JA, Mata A. Trophic effects of neurotensin in massive bowel resection in the rat. Dig Dis Sci 1994;39:59–64.
Evers BM, Izukura M, Rajaraman S, Parekh D, Thakore K, Yoshinaga K, Uchida T, Townsend CM Jr, Thompson JC. Effect of aging on neurotensin-stimulated growth of rat small intestine. Am J Physiol 1994;267:G180-G186.
Scolapio JS, Fleming CR. Short bowel syndrome. Gastro-enterol Clin North Am 1998;27:467–479, viii.
Wilmore DW, Robinson MK. Short bowel syndrome. World J Surg 2000;24:1486–1492.
Evers BM. Small bowel. In Townsend CM Jr, Beauchamp RD, Evers BM, Mattox KL, eds. Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice, 16th ed. Philadephia: W.B. Saunders, 2001, pp 873–916.
Chesky JA, Rockstein M. Life span characteristics in the male Fischer rat. Exp Aging Res 1976;2:399–407.
Khalil T, Thompson JC. Aging and gut peptides. In Thompson JC, Greeley GH Jr, Rayford PL, Townsend CM Jr, eds. Gastrointestinal Endocrinology. New York: McGraw-Hill, 1987, pp 147–157.
Burton K. A study of the conditions and mechanisms of the diphenylamine reaction for the colorimetric estimation of deoxyribonucleic acid. Biochemistry 1956;162:315–323.
Bradford MM. A rapid and sensitive method for the quantita-tion of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248–254.
Booth CC, Evans KT, Menzies T, Street DF. Intestinal hypertrophy following partial resection of the small bowel in the rat. Br J Surg 1959;46:403–410.
Dowling RH, Booth CC. Structural and functional changes following small intestinal resection in the rat. Clin Sci 1967;32:139–149.
Hanson WR. Proliferative and morphological adaptation of the intestine to experimental resection. Scand J Gastroen-terol(Suppl)1982;74:11–20.
Hanson WR, Rijke RP, Plaisier HM, Van Ewijk W, Os-borne JW. The effect of intestinal resection on Thiry-Vella fistulae of jejunal and ileal origin in the rat: evidence for a systemic control mechanism of cell renewal. Cell Tissue Ki-net 1977;10:543–555.
O’Loughlin E, Winter M, Shun A, Hardin JA, Gall DG. Structural and functional adaptation following jejunal resection in rabbits: Effect of epidermal growth factor. Gastroenterology 1994;107:87–93.
Stern LE, Erwin CR, O’Brien DP, Huang F, Warner BW. Epidermal growth factor is critical for intestinal adaptation following small bowel resection. Microsc Res Tech 2000; 51:138–148.
Fadrique B, Lopez JM, Bermudez R, Gomez de Segura IA, Vazquez I, De Miguel E. Growth hormone plus high protein diet promotes adaptation after massive bowel resection in aged rats. Exp Gerontol 2001;36:1727–1737.
Bucher NLR. The effects of age on regeneration of rat liver. Cancer Res 1950;10:324–332.
Bucher NLR. The influence of age upon the incorporation of thymadine-2-C14 into the DNA of regenerating rat liver. Cancer Res 1964;24:509–512.
Kropacova K, Misurova E. Influence of age and gamma irradiation on the proliferative activity in regenerating rat liver. Physiol Res 1992;41:135–140.
Timchenko NA, Wilde M, Kosai KI, Heydari A, Bilyeu TA, Finegold MJ, Mohamedali K, Richardson A, Darlington GJ. Regenerating livers of old rats contain high levels of C/EB-Palpha that correlate with altered expression of cell cycle associated proteins. Nucleic Acids Res 1998;26:3293–3299.
Wang X, Quail E, Hung NJ, Tan Y, Ye H, Costa RH. Increased levels of forkhead box M1B transcription factor in transgenic mouse hepatocytes prevent age-related proliferation defects in regenerating liver. Proc Natl Acad Sci USA 2001;98:11468–11473.
Tanigawa K, Nakamura S, Kawaguchi M, Xu G, Kin S, Tamura K. Effect of aging on B-cell function and replication in rat pancreas after 90% pancreatectomy. Pancreas 1997;15:53–59.
Sim FJ, Zhao C, Penderis J, Franklin RJ. The age-related decrease in CNS remyelination efficiency is attributable to an impairment of both oligodendrocyte progenitor recruitment and differentiation. J Neurosci 2002;22:2451–2459.
Lipkin M. Phase 1 and phase 2 proliferative lesions of co-lonic epithelial cells in diseases leading to colonic cancer. Cancer 1974;34(Suppl):878–888.
Ryan CK, Miller JH, Seydel AS, Jensen KM, Sax HC. Epidermal growth factor and neurotensin induce microvillus hypertrophy following massive enterectomy. JGastrointest Surg 1997;1:467–473.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by grants R37 AG10885, P01 DK35608, and T32 DK07639 from the National Institutes of Health and by a Jeane B. Kempner Scholars Award (R.P.T.).
Rights and permissions
About this article
Cite this article
Thomas, R.P., Slogoff, M., Smith, F.W. et al. Effect of aging on the adaptive and proliferative capacity of the small bowel. J Gastrointest Surg 7, 88–95 (2003). https://doi.org/10.1016/S1091-255X(02)00128-2
Published:
Issue Date:
DOI: https://doi.org/10.1016/S1091-255X(02)00128-2