Abstract
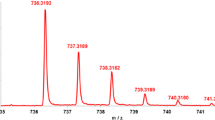
A rapid and accurate method of quantifying positional isomeric mixtures of phosphorylated hexose and N-acetylhexosamine monosacchrides by using gas-phase ion/molecule reactions coupled with FT-ICR mass spectrometry is described. Trimethyl borate, the reagent gas, reacts readily with the singly charged negative ions of phosphorylated monosaccharides to form two stable product ions corresponding to the loss of one or two neutral molecules of methanol from the original adduct. Product distribution in the ion/molecule reaction spectra differs significantly for isomers phosphorylated in either the 1- or the 6-position. As a result, the percents of total ion current of these product ions for a mixture of the two isomers vary with its composition. In order to determine the percentage of each isomer in an unknown mixture, a multicomponent quantification method is utilized in which the percents of total ion current of the two product ions for each pure monosaccharide phosphate and the mixture are used in a two-equation, two-unknown system. The applicability of this method is demonstrated by successfully quantifying mock mixtures of four different isomeric pairs: Glucose-1-phosphate and glucose-6-phosphate; mannose-1-phosphate and mannose-6-phosphate; galactose-1-phosphate and galactose-6-phosphate; N-acetylglucosamine-1-phosphate and N-acetylglucosamine-6-phosphate. The effects of mixture concentrations and ion/molecule reaction conditions on the quantification are also discussed. Our results demonstrate that this assay is a fast, sensitive, and robust method to quantify isomeric mixtures of phosphorylated monosaccharides.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Marquardt, T. Freeze, H. Congenital Disorders of Glycosylation: Glycosylation Defects in Man and Biological Models for Their Study. Biol. Chem 2001, 382, 161–177.
Saier, M. H., Jr.; Chauvaux, S. Cook, G. M. Deutscher, J. Paulsen, I. T. Reizer, J. Ye, J. J. Catabolite Repression and Inducer Control in Gram-Positive Bacteria. Microbiol 1996, 142, 217–230.
Romano, A. H. Conway, T. Evolution of Carbohydrate Metabolic Pathways. Res. Microbiol 1996, 147, 448–455.
Kotrba, P. Inui, M. Yukawa, H. Bacterial Phosphotransferase System (PTS) in Carbohydrate Uptake and Control of Carbon Metabolism. J. Biosci. Bioeng 2001, 92, 502–517.
Yarema, K. J. Bertozzi, C. R. Characterizing Glycosylation Pathways. Genome Biol 2001, 2, 1–10.
Kvam, C. Olsvik, E. S. McKinley-McKee, J. Saether, O. Studies on Recombinant Acetobacter xylinum α-Phosphoglucomutase. Biochem. J 1997, 326, Pt 1, 197–203.
Murata, T. Studies on Konjak Mannan Biosynthesis. 3. Studies on Phosphomannose Isomerase of Amorphophallus-Konjac C Koch. 1. Isolation and Some Enzymic Properties. Plant Cell Physiol 1975, 16, 953–961.
Ye, R. W. Zielinski, N. A. Chakrabarty, A. M. Purification and Characterization of Phosphomannomutase/Phosphoglucomutase from Pseudomonas aeruginosa Involved in Biosynthesis of Both Alginate and Lipopolysaccharide. J. Bacteriol 1994, 176, 4851–4857.
Cheng, P. W. Carlson, D. M. Mechanism of Phosphoacetylglucosamine Mutase. J. Biol. Chem 1979, 254, 8353–8357.
Li, Y. J. Ogata, Y. Freeze, H. H. Scott, C. R. Turecek, F. E. Gelb, M. H. Affinity Capture and Elution/Electrospray Ionization Mass Spectrometry Assay of Phosphomannomutase and Phosphomannose Isomerase for the Multiplex Analysis of Congenital Disorders of Glycosylation Types Ia and Ib. Anal. Chem 2003, 75, 42–48.
Brunt, K. Hokse, H. Liquid-Chromatographic Analysis of α-D-Glucose-1-Phosphate—Determination of the Activity of Phosphorylase. J. Chromatogr 1983, 268, 131–137.
Zhou, Q. Kyazike, J. Edmunds, T. Higgins, E. Mannose 6-Phosphate Quantitation in Glycoproteins Using high-pH Anion-Exchange Chromatography with Pulsed Amperometric Detection. Anal. Biochem 2002, 306, 163–170.
Feurle, J. Jomaa, H. Wilhelm, M. Gutsche, B. Herderich, M. Analysis of Phosphorylated Carbohydrates by High-Performance Liquid Chromatography Electrospray Ionization Tandem Mass Spectrometry Utilizing a β-Cyclodextrin Bonded Stationary Phase. J. Chromatogr. A 1998, 803, 111–119.
Leavell, M. D. Kruppa, G. H. Leary, J. A. Analysis of Phosphate Position in Hexose Monosaccharides Using Ion/Molecule Reactions and SORI-CID on an FT-ICR Mass Spectrometer. Anal. Chem 2002, 74, 2608–2611.
Leavell, M. D. Kruppa, G. H. Leary, J. A. Probing Isomeric Differences of Phosphorylated Carbohydrates Through the Use of Ion/Molecule Reactions and FT-ICR. J. Am. Soc. Mass Spectrom 2003, 14, 323–331.
Tao, W. A. Gozzo, F. C. Cooks, R. G. Mass Spectrometric Quantitation of Chiral Drugs by the Kinetic Method. Anal. Chem 2001, 73, 1692–1698.
Augusti, D. V. Carazza, F. Augusti, R. Tao, W. A. Cooks, R. G. Quantitative Chiral Analysis of Sugars by Electrospray Ionization Tandem Mass Spectrometry Using Modified Amino Acids as Chiral Reference Compounds. Anal. Chem 2002, 74, 3458–3462.
Chu, I. H. Dearden, D. V. Bradshaw, J. S. Huszthy, P. Izatt, R. M. Chiral Host–Guest Recognition in an Ion/Molecule Reaction. J. Am. Chem. Soc 1993, 115, 4318–4320.
Ramirez, J. He, F. Lebrilla, C. B. Gas-Phase Chiral Differentiation of Amino Acid Guests in Cyclodextrin Hosts. J. Am. Chem. Soc 1998, 120, 7387–7388.
Nikolaev, E. N. Goginashvili, G. T. Talrose, V. L. Kostyanovsky, R. G. Investigation of Asymmetric Gas-Phase Ion/Molecule Reactions by FT-ICR Spectrometry. Int. J. Mass Spectrom. Ion Processes 1988, 86, 249–252.
Nikolaev, E. N. Denisov, E. V. Rakov, V. S. Futrell, J. H. Investigation of Dialkyl Tartrate Molecular Recognition in Cluster Ions by Fourier Transform Mass Spectrometry: A Comparison of Chirality Effects in Gas and Liquid Phases. Int. J. Mass Spectrom 1999, 183, 357–368.
Camara, E. Green, M. K. Penn, S. G. Lebrilla, C. B. Chiral Recognition is Observed in the Deprotonation Reaction of Cytochrome c by (2R)- and (2S)-2-Butylamine. J. Am. Chem. Soc 1996, 118, 8751–8752.
Dearden, D. V. Dejsupa, C. Liang, Y. J. Bradshaw, J. S. Izatt, R. M. Intrinsic Contributions to Chiral Recognition: Discrimination Between Enantiomeric Amines by Dimethyldiketopyridino-18-Crown-6 in the Gas Phase. J. Am. Chem. Soc 1997, 119, 353–359.
Liang, Y. J. Bradshaw, J. S. Izatt, R. M. Pope, R. M. Dearden, D. V. Analysis of Enantiomeric Excess Using Mass Spectrometry: Fast Atom Bombardment/Sector and Electrospray Ionization Fourier Transform Mass Spectrometric Approaches. Int. J. Mass Spectrom 1999, 187, 977–988.
Grigorean, G. Ramirez, J. Ahn, S. H. Lebrilla, C. B. A Mass Spectrometry Method for the Determination of Enantiomeric Excess in Mixtures of D,L-Amino Acids. Anal. Chem 2000, 72, 4275–4281.
Leeck, D. T. Ranatunga, T. D. Smith, R. L. Partanen, T. Vainiotalo, P. Kenttämaa, H. I. Differentiation of Stereoisomeric Diols by Using Ch3ob + Och3 in a Small Fourier-Transform Ion-Cyclotron Resonance Mass-Spectrometer. Int. J. Mass Spectrom. Ion Processes 1995, 141, 229–240.
Thoen, K. K. Gao, L. Ranatunga, T. D. Vainiotalo, P. Kenttämaa, H. I. Stereoselective Chemical Ionization Mass Spectrometry: Reactions of CH3OPOCH3+ with Cyclic Vicinal Diols. J. Org. Chem 1997, 62, 8702–8707.
Thompson, R. S. Guler, L. P. Nelson, E. D. Yu, Y. Q. Kenttämaa, H. I. Mechanistic Study of Stereoselective Gas-Phase Reactions of Phosphenium Ions with Cis- and Trans-1,2-Diaminocyclohexanes. J. Org. Chem 2002, 67, 5076–5084.
Desaire, H. Leary, J. A. Multicomponent Quantification of Diastereomeric Hexosamine Monosaccharides Using Ion Trap Tandem Mass Spectrometry. Anal. Chem 1999, 71, 4142–4147.
Desaire, H. Leary, J. A. Utilization of MS3 Spectra for the Multicomponent Quantification of Diastereomeric N-Acetylhexosamines. J. Am. Soc. Mass Spectrom 2000, 11, 1086–1094.
Desaire, H. Leary, J. A. Detection and Quantification of the Sulfated Disaccharides in Chondroitin Sulfate by Electrospray Tandem Mass Spectrometry. J. Am. Soc. Mass Spectrom 2000, 11, 916–920.
de Koning, L. J. Nibbering, N. M. M. van Orden, S. L. Laukien, F. H. Mass Selection of Ions in a Fourier Transform Ion Cyclotron Resonance Trap Using Correlated Harmonic Excitation Fields (CHEF). Int. J. Mass Spectrom 1997, 165, 209–219.
Su, T. Chesnavich, W. J. Parametrization of the Ion–Polar Molecule Collision Rate-Constant by Trajectory Calculations. J. Chem. Phys 1982, 76, 5183–5185.
Gronert, S. O’Hair, R. A. J. Gas Phase Reactions of Trimethyl Borate with Phosphates and Their Non-Covalent Complexes. J. Am. Soc. Mass Spectrom 2002, 13, 1088–1098.
Leavell, M. D. Kruppa, G. H. Leary, J. A. Determination of Phosphate Position in Hexose Monosaccharides Using an FTICR Mass Spectrometer: Ion/Molecule Reactions, Labeling Studies, and Dissociation Mechanisms. Int. J. Mass Spectrom 2003, 222, 135–153.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online July 2, 2003
Rights and permissions
About this article
Cite this article
Gao, H., Petzold, C.J., Leavell, M.D. et al. Investigation of ion/molecule reactions as a quantification method for phosphorylated positional isomers: An FT-ICR approach. J Am Soc Mass Spectrom 14, 916–924 (2003). https://doi.org/10.1016/S1044-0305(03)00401-X
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S1044-0305(03)00401-X