Abstract
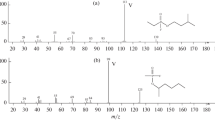
A chemical ionization method is reported for distinction of diastereomeric hydroxysteroids by using Fourier-transform ion cyclotron mass spectrometry (FT-ICR). Certain phosphenium ions are demonstrated to react with stereoisomeric steroids to yield qualitatively different product ions. For example, l,3,5(10)-estratriene-3,16β,17β-triol (cis-estriol) reacts with the dimethoxy phosphenium ion to form a diagnostic product ion (not formed for the trans-estriol) through addition followed by the loss of two molecules of methanol. In an analogous manner, the 1,3-dioxolan-2-phosphenium ion produces a diagnostic product ion through the loss of ethylene glycol from the adduct of cis-estriol only. The l,3,5(10)-estratriene-3,16α,17β-triol (trans-estriol), on the other hand, reacts with each phosphenium ion only via hydroxide abstraction-initiated pathways that indicate the presence of at least two hydroxyl groups in the molecule. These specific reactions take place for all hydroxysteroids examined, independent of their stereochemistry. Another isomer pair, cholestan-3α,5α-diol (cis-cholestandiol) and cholestan-3β,5α-diol (trans-cholestandiol), is differentiated based on selective elimination of water only from the adduct of the cis-isomer. However, the method does not allow distinction between the stereoisomeric 5β-pregnane-3α,17α,20α-triol and 5β-pregnane-3α,17α,20β-triol. The different reactivities of the three pairs of steroid isomers and of each diastereomeric compound pair are rationalized by reaction enthalpies and steric effects based on straightforward and predictable reaction mechanisms.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wainer, I. W. Drug Stereochemistry. Analytical Methods and Pharmacology. Marcel Dekker: New York, 1993.
Menges, R. A.; Armstrong, D. W. In Chiral Separations by Liquid Chromatography. ACS Symposium Series 471; Ahuja, S., Ed.; American Chemical Society: Washington, D. C., 1991; 67–100.
Tabet, J.-C. Applications of Mass Spectrometry to Organic Stereochemistry, Splitter, J. S.; Turecek, F.; Eds.; VCH Publishers: New York, 1994, Chap XIX.
Brodbelt, J. S. Mass Spec. Rev. 1997, 16, 91–110 and references within.
Winkler, F. J.; McLafferty, F. W. Tetrahedron 1974, 30, 2971–2976.
Sellier, N.; Mancel, V. Rapid Commun. Mass Spectrom. 2000, 14, 80–85.
Leeck, D. T.; Ranatunga, T. D.; Smith, R. L.; Partanen, T.; Vainiotalo, P.; Kenttämaa, H. I. Int. J. Mass Spectrom. Ion Processes 1995, 141, 229–240.
Thoen, K. K.; Gao, L.; Ranatunga, T. D.; Vainiotalo, P.; Kenttämaa, H. I. J. Org. Chem 1997, 62, 8702–8707.
Suming, H.; Yaozu, C.; Longfei, J.; Shuman, X. Org. Mass. Spectrom. 1985, 20, 719–723.
Moraes, L. A. B.; Eberlin, M. N. J. Am. Soc. Mass Spectrom. 2001, 12, 150–162.
Thompson, R. S.; Guler, L. P.; Nelson, E. D.; Yu, Y.; Kenttämaa, H. I. J. Org. Chem. In press.
Grostic, M. F.; Rinehart, K. L., Jr. J. Org. Chem. 1968, 33, 1740–1746.
Longevialle, P.; Milne, G. W. A.; Fales, H. M. J. Am. Chem. Soc. 1973, 95, 6666–6669.
Sjöberg, P. J. R.; Markides, K. E. J. Mass Spectrom. 1998, 33, 872–883.
Carlesso, V.; Fournier, F.; Tabet, J.-C. Eur. J. Mass Spectrom. 2000, 6, 421–428.
Desaire, H.; Leary, J. A. J. Am. Soc. Mass Spectrom. 2000, 11, 1086–1094.
Tao, W. A.; Wu, L.; Cooks, R. G. Chem. Commun. 2000, 2023–2024.
Zhang, D.; Tao, W. A.; Cooks, R. G. Int. J. Mass Spectrom. 2001, 204, 159–169.
Tao, A. W.; Cooks, R. G. Angew. Chem. Int. 2001, 40, 757–760.
Tao, W. A.; Gozzo, F. C.; Cooks, R. G. Anal. Chem. 2001, 73, 1692–1698.
Grigorean, G.; Lebrilla, C. B. Anal. Chem. 2001, 73, 1684–1691.
Chen, L.; Wang, T.-C. L.; Ricca, T. L.; Marshall, A. G. Anal. Chem. 1987, 59, 449–454.
Thoen, K.; Smith, R. L.; Nousiainen, J. J.; Nelson, E. D.; Kenttämaa, H. I. J. Am. Chem. Soc. 1996, 118, 8669–8676.
(a) Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Zakrzewski, V. G.; Montgomery, J. A., Jr.; Stratmann, R. E.; Burant, J. C.; Dapprich, S.; Millam, J. M.; Daniels, A.D.; Kudin, K. N.; Strain, M. C.; Farkas, O.; Tomasi, J.; Barone, V.; Cossi, M.; Cammi, R.; Menucci, B.; Pomelli, C.; Adamo, C.; Clifford, S.; Ochterski, J.; Petersson, G. A.; Ayala, P. Y.; Cui, Q.; Morokuma, K.; Malick, D. K.; Rabuck, A. D.; Raghavahari, K.; Foresman, J. B.; Cioslowski, J.; Ortiz, J. V.; Baboul, A. G.; Stefanov, B. B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Gomperts, R.; Martin, R. L.; Fox, D. J.; Keith, T.; Al-Laham, M. A.; Peng, C. Y.; Nanayakkara, A.; Gonzalez, C.; Challacombe, M.; Gill, P. M. W.; Johson, B.; Chen, W.; Wong, M. W.; Andres, J. L.; Head-Gordon, M.; Replogle, E. S.; Pople, J. A. Gaussian, Inc.: Pittsburgh, 1998.
Scott, A. P.; Radom, L. J. Phys. Chem. 1996, 100, 16502–16513.
Cowley, A. H.; Kemp, R. A. Chem. Rev. 1985, 85, 367–382.
Wolfschütz, R.; Schwarz, H.; Blum, W.; Richter, W. J. Org. Mass Spectrom. 1978, 13, 697–704.
Lin, P.; Kenttämaa, H. I. J. Phys. Org. Chem. 1992, 5, 201–208.
Kenttämaa, H. I.; Pachuta, R. R.; Rothwell, A. P.; Cooks, R. G. J. Am. Chem. Soc. 1989, 111, 1654–1665.
Donovan, T.; Brodbelt, J. J. Am. Soc. Mass Spectrom. 1992, 3, 47–59.
Sigsby, M. L.; Day, R. J.; Cooks, R. G. Org. Mass Spectrom. 1979, 14, 273–280.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Petucci, C., Guler, L. & Kenttämaa, H.I. Differentiation of stereoisomeric steroids by reactions with phosphenium ions. J Am Soc Mass Spectrom 13, 362–370 (2002). https://doi.org/10.1016/S1044-0305(02)00344-6
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S1044-0305(02)00344-6