Abstract
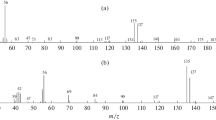
A similarity of electron ionization mass spectra of various alkyl esters of benzene-1,2-dicarboxylic (phthalic) acid places special demands on the reliability of the identification of monoalkyl phthalates, often recognized as dialkyl phthalates and vice versa. An original mass spectrometric criterion is proposed for the group identification of monoalkyl phthalates (assignment to a subgroup of homologues), which assumes the calculation of the total relative intensity of ion peaks at m/z > 170. In the mass spectra of monoalkyl esters, this value does not exceed 1%, whereas for dialkyl ethers, it varies from 1 to 172%. Within the set subgroups of isomers, it becomes effective to use the homologous increments of the retention indices, iRI = RI – 100x, where x = int (M/14), to refine the identification results. Using predefined iRI values and the ratio М ≈ 0.14(RI – iRI) + y, we can estimate the molecular weights of compounds in the mass spectra of which the molecular ion signals are not detected or are of low intensity (including monoalkyl phthalates). In addition, iRI values are in many cases informative for estimating the number of branches of the sp3 carbon skeleton of molecules (Z) based on regression relations of the form of iRI = aZ + b. The examination of homologous increments of the retention indices of dialkyl phthalates revealed an anomalous dependence of their values on the number of carbon atoms in the alkyl fragments of the molecules: from +125 (СН3) to –33 (С10Н21). In the subgroup of monoalkyl phthalates, such anomalies were not observed.

Similar content being viewed by others
Notes
Identification errors of the Ist order is the impossibility of correct identification of the compounds characterized and presented in the reference information arrays, of the IInd order is an erroneous identification of previously uncharacterized objects.
REFERENCES
The NIST 17 Mass Spectral Library (NIST17/2017/EPA/NIH), NIST Standard Reference Database no. 69, June 2017, Gaithersburg, MD: Natl. Inst. Stand. Technol. http://webbook.nist.gov. Accessed January 2020.
Zenkevich, I.G. and Lukina, V.M., Analitika Kontrol’, 2019, vol. 23, no. 3, p. 410. https://doi.org/10.15826/analitika.2019.23.3.009
Eliseenkov, E.V. and Zenkevich, I.G., Mass-Spektrom., 2020, vol. 20 (in press).
Zenkevich, I.G., Anal. Bioanal. Chem., 2013, vol. 405, p. 3075. https://doi.org/10.1007/s00216-013-6751-2
Zenkevich, I.G., Pushkareva, T.I., and Karakashev, G.V., Analitika Kontrol’, 2019, vol. 23, no. 3, p. 425. https://doi.org/10.15826/analitika.2019.23.3.012
Zenkevich, I.G. and Fakhretdinova, L.N., Analitika Kontrol’, 2015, vol. 19, no. 2, p. 175. https://doi.org/10.15826/analitika.2015.19.2.013
Zenkevich, I.G. and Fakhretdinova, L.N., J. Anal. Chem., 2016, vol. 71, no. 12, p. 1204. https://doi.org/10.7868/S004445021612015X
Hamming, M.G. and Foster, N.G., Interpretation of Mass Spectra of Organic Compounds, New York: Academic, 1979.
Kim, N.-S. and Lee, D.S., Anal. Sci., 2001, vol. 17, no. 1 (suppl.), A383.
Pasenzotto, L., Gracco, L., and Conte, L., J. Sci. Food Agric., 2001, vol. 83, p. 1037.
Syeda, F.A., Habib-ur-Rehman, Choudahry, M.I., and Ata-ur-Rahman, Int. J. Genet. Mol. Biol., 2011, vol. 3, no. 7, p. 95.
Bagavathi, P.E. and Ramasamy, N., Pharmacogn. Res., 2012, vol. 14, no. 1, p. 11. https://doi.org/10.4103/0974-8490.91028
Kawakami, T., Isama, K., and Matsuoka, A., J. Environ. Sci. Health, 2011, vol. 46, no. 8, p. 855. https://doi.org/10.1080/10934529.2011.579870
Rowdhwal, S.S.S. and Chen, J., BioMed Res. Int., 2018, 1750368. https://doi.org/10.1155/2018.1750368
Stein, S.E., Babushok, V.I., Brown, R.L., and Linstrom, P.J., J. Chem. Inf. Model., 2007, vol. 47, p. 975. https://doi.org/10.1021/ci600548y
Zenkevich, I.G. and Ioffe, B.V., Interpretatsiya mass-spektrov organicheskikh soedinenii (Interpretation of Mass Spectra of Organic Compounds), Leningrad: Khimiya, 1986.
Zenkevich, I.G., Russ. J. Gen. Chem., 2017, vol. 87, no. 4, p. 795. https://doi.org/10.1134/S1070363217040211
Zenkevich, I.G. and Nosova, V.E., J. Anal. Chem., 2019, vol. 74, no. 13, p. 1297. https://doi.org/10.1134/S1061934819130124
Zenkevich, I.G. and Nosova, V.E., J. Anal. Chem., 2019, vol. 74, no. 14, p. 1413. https://doi.org/10.1134/S1061934819140120
Zenkevich, I.G., Russ. J. Gen. Chem., 2019, vol. 89, no. 3, p. 369. https://doi.org/10.1134/S1070363219030010
ACKNOWLEDGMENTS
The experimental data discussed in this work were obtained using the equipment of the Resource Center in the field of “Chemistry” at the Institute of Chemistry, St. Petersburg State University. The author is grateful to his colleagues working at the Center for assistance. The author acknowledges Prof. Károly Héberger (Research Center for Natural Sciences, Hungarian Academy of Sciences, Budapest) for his conclusions regarding the possibilities of using chemometric methods for the differentiation of alkyl phthalates.
Funding
This work was partially supported by the Russian Foundation for Basic Research, project no. 18-03-00151/A.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by O. Zhukova
Rights and permissions
About this article
Cite this article
Zenkevich, I.G. Specific Features of the Gas Chromatography–Mass Spectrometry Identification of Monoalkyl Phthalates. J Anal Chem 75, 1322–1329 (2020). https://doi.org/10.1134/S1061934820100160
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934820100160