Abstract
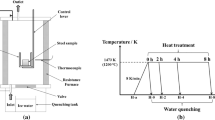
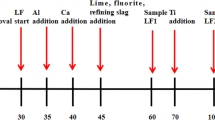
Refining process of 430 stainless steel in AOD (argon oxygen decarburization) was simulated under the experimental condition. Al was chosen as deoxidizer, Mg-Al alloy was added in process of refining, and slag used in oxidation period of AOD was chosen in the experiments. The variation of total oxygen content, the size, morphology and composition of inclusions in refining process and the mechanical properties, pitting corrosion resistance of final samples were studied. The results show that no obvious differences in total oxygen content were observed among all the experiments. Neither chain nor cluster Al2O3 inclusions were found in refining process of experiments treated by Mg-Al alloys, the average size of inclusions in the steel treated by Mg-Al alloys was less than that of inclusions in steel not treated by Mg-Al alloy. 430 stainless steel treated by Mg-Al alloy shows better tensile strength, yield strength, and pitting resistance than that in the contrast experiment.
Similar content being viewed by others
References
NIU Lin-xia. Development Direction of Production and Technology of Stainless Steel [J]. Iron Steel Research, 2007, 35 (6): 46 (in Chinese).
Mapelli P, Nolli P. Formation Mechanism of Non-Metallic Inclusions in Different Stainless Steel Grades [J]. ISU International, 2003, 43(8): 1191.
YANG Rui-cheng, BI Hai-juan, NIU Shao-rui, et al. The Resistance to Local Corrosion of 409L and 430 Stainless Steel [J]. Univ Sci Technol Beijing, 2003, 33(4): 428 (in Chinese).
Saxena S K. Production of Ultra-Clean Steels With Better Mechanical Properties With Magnesium Treatment [C] // Steel-making Conference Proceedings. Pittsburgh: Iron and Steel Soc of AIME, 1996: 89.
Saxena S K. Refining Reaction of Magnesium in Steel at Steel-making Temperature [C]//Proceedings International Symposium on the Physical Chemistry of Iron and Steelmaking. Toronto: Conference of Metallurgists, 1982: 17.
JIANG Zhou-hua, LI Shuang-jiang, LI Yang. Thermodynamic Calculation of Inclusion Formation in Mg-Al-Si-0 System of 430 Stainless Steel [J]. Journal of Iron and Steel Research, International, 2011, 18(2): 14.
Itoh H, Hino M, Ban Y S. Thermodynamics on the Formation of Non-Metallic Inclusion of Spinel in Liquid Steel [J]. Testu-to-Hagane, 1998, 84(2): 85 (in Japanese).
Nishi T, Shinme K. Formation of Spinel Inclusion in Molten Stainless Steel Under Al Deoxidation With Slag [J]. Tetsu-to-Hagane, 1998, 84(12): 837 (in Japanese).
Cha W Y, Kim D S, Lee Y D, et al. A Thermodynamic Study on the Inclusion Formation in Ferritic Stainless Steel Melt [J]. ISIJ International, 2004, 44(7): 1134.
LI Zheng-bang. New Development of Super Clean Steel [J]. Journal of Materials and Metallurgy, 2002, 1(3): 161 (in Chinese).
WANG Bo, JIANG Zhou-hua, JIANG Mao-fa. Inclusion Modification in GCrlS Bearing Steel With Al-Mg Alloy Treatment [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(10): 1736 (in Chinese).
LI Yang, ZHI Wei-jun, LI Wei-jiang, et al. Preparation of Ultra-Fine MgO • A1203 Spinel Powder and Its Metallurgy Behavior in Medium Carbon Steel [J]. Advanced Materials Research, 2009, 79-82: 111.
LI Yang, LI Shuang-jiang, ZHUANG Ying, et al. Preparation of Ultra-Fine MgO † Al2O3 Spinel Powder and Its Metallurgy Behavior in Low Carbon Steel [J]. Materials Science Forum, 2011, 675-677: 291.
Krawiec H, Vignal V. Dissolution of Chromium-Enriched Inclusion and Pitting Corrosion of Resulfurized Stainless Steels [J]. Metall Mat Trans: A Phys Metall Mat Sci, 2006, 5: 1541 (in Chinese).
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation Item: Item Sponsored by National Natural Science Foundation of China (50704010); Fundamental Research Funds for Central Universities of China (090402019)
Rights and permissions
About this article
Cite this article
Jiang, Zh., Zhuang, Y., Li, Y. et al. Effect of Modification Treatment on Inclusions in 430 Stainless Steel by Mg-Al Alloys. J. Iron Steel Res. Int. 20, 6–10 (2013). https://doi.org/10.1016/S1006-706X(13)60089-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1016/S1006-706X(13)60089-8