Abstract
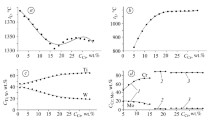
On the basis of regular solution sublattice model, thermodynamic equilibrium of austenite/carbide in Fe-Nb-C ternary system was investigated. The equilibrium volume fraction, chemical driving force of carbide precipitates and molar fraction of niobium and carbon in solution at different temperatures were evaluated respectively. The volume fraction of precipitates increases, molar fraction of niobium dissolved in austenite decreases and molar fraction of carbon increases with decreasing the niobium content. The driving force increases with the decrease of temperature, and then comes to be stable at relatively low temperatures. The predicted ratio of carbon in precipitates is in good agreement with the measured one.
Similar content being viewed by others

References
Hillert M, Jarl M A. Thermodynamic Analysis of the Iron-Nitrogen System [J]. Metall Trans, 1975, 6A: 553–558.
Lakshmanan V K. Thermodynamics of the Interstitial Phases of Niobium in Iron Austenite [D]. Hamilton: McMaster University, 1977.
Ohtani H, Hasebe M, Nishizawa T. Calculation of Fe-C, Co-C and Ni-C Phase Diagrams [J]. Trans Iron Steel Inst Japan, 1984, 24: 857–863.
Kaufman L, Bernstein H. Computer Calculation of Phase Diagrams [M]. New York: Academic Press, 1970.
Ohtani H, Hasebe M, Nishizawa T. Calculation of Fe-C-Nb Ternary Phase Diagram [J]. Calphad, 1989, 13: 187–194.
XU Yun-bo, YU Yong-mei, WU Di, et al. Thermodynamic Calculations of Precipitation Behavior in Nb Microalloyed Steels [J]. Chinese Journal of Materials Research, 2006, 20(1): 104–108 (in Chinese).
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation Item: Item Sponsored by National Natural Science Foundation of China (50504007, 50474086, 50334010); Liaoning Province Science Foundation (20041009)
Rights and permissions
About this article
Cite this article
Xu, Yb., Yu, Ym., Liu, Xh. et al. Thermodynamic Calculation of Carbide Precipitate in Niobium Microalloyed Steels. J. Iron Steel Res. Int. 13, 53–56 (2006). https://doi.org/10.1016/S1006-706X(06)60110-6
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1016/S1006-706X(06)60110-6