Abstract
This study presents a comprehensive examination of the Indo-Pacific Tarpon (IPT) population in Lake Siombak, North Sumatra Province, Indonesia. Through a year-long investigation, we analyzed various facets of IPT ecology, encompassing population structure, growth dynamics, reproductive behavior, feeding habits, and mortality parameters. Male dominance in collected specimens, coupled with consistently immature gonads in females, suggests a unique reproductive pattern influenced by the brackish nature of Lake Siombak. Utilizing the von Bertalanffy method, we determined a moderate growth rate in IPT, with a predominance of medium-sized, sexually immature individuals. The dietary analysis revealed an omnivorous nature, with a notable reliance on zooplankton, particularly Cladocera and Copepods. However, the most significant concern arises from the estimation of the exploitation rate (E), surpassing the annual optimum exploitation threshold, indicating a state of overexploitation. This underscores the urgent need for regulatory measures to ensure the sustainability of IPT resources in Lake Siombak. Our findings contribute to a nuanced understanding of IPT ecology in this specific ecosystem, emphasizing the importance of balanced conservation and management practices. The documented reproductive, growth, and feeding patterns provide crucial insights for informing targeted conservation strategies and safeguarding the enduring viability of the IPT population in Lake Siombak. This research serves as a foundational contribution to the broader field of fisheries science, advocating for evidence-based management to preserve the ecological balance of aquatic ecosystems.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
1 Introduction
Lake Siombak, a coastal water expanse covering approximately 29 hectares in Indonesia, stands as a distinctive aquatic ecosystem, diverging from recently identified anchialine lakes within the Indonesia Archipelago [1]. Its uniqueness lies in hydrodynamic conditions shaped by tidal currents from the ocean, rendering it an estuarine ecosystem replete with a myriad of flora and fauna, spanning freshwater, brackish, and marine organisms [2,3,4]. The presence of lush mangrove forests along the lake's periphery further enriches its biodiversity [5].
Within this dynamic ecosystem, the Indo-Pacific Tarpon Megalop cyprinoides (Broussonet, 1782) emerges as a prime focus for local fishermen in Lake Siombak. Notably inhabiting the Belawan estuary [4, 6], the Indo-Pacific Tarpon (IPT) is a pelagic species thriving in coastal waters and river mouths, presenting significant nutritional potential as a protein source (Genisa, 1999; Froese and Pauly, 2019). Previous investigations indicate a protein content in IPT surpassing that of Tuna and Tilapia [7]. Despite its culinary promise, comprehensive explorations of IPT's biological dimensions, including population aspects such as growth, reproduction, feeding habits, and exploitation rates, remain notably absent.
Existing research has touched upon morphometrics and length–weight relationships, yet a holistic understanding of IPT's population structure and stock status remains elusive [6, 8, 9]. This knowledge gap is particularly glaring given the economic and ecological significance of Lake Siombak, enveloping critical ecosystems like mangrove forests and coastal habitats [10]. In light of this, there is a pressing need for sustainable management practices informed by robust scientific data.
Furthermore, human activities exert significant pressures on the aquatic ecosystem. Rapid urbanization and industrialization in the surrounding areas have led to increased pollution levels, primarily from untreated sewage, industrial effluents, and agricultural runoff [11, 12]. This is also observed in Lake Siombak, where anthropogenic activities contribute to environmental degradation and pose challenges to the conservation of biodiversity and ecosystem health [13,14,15]. These pollutants degrade water quality, disrupt nutrient cycles, and pose serious health risks to aquatic organisms, including the Indo-Pacific Tarpon. Moreover, habitat destruction, primarily through deforestation and land reclamation for agriculture and urban development, further exacerbates ecological stressors. These anthropogenic pressures threaten the integrity of Lake Siombak's ecosystem, compromising its resilience and long-term sustainability [16,17,18].
The socioeconomic context surrounding Lake Siombak underscores the intertwined relationship between local communities and natural resources [19]. Fishing serves as a primary source of livelihood for many residents, providing employment opportunities and contributing to household income and food security [20]. However, socioeconomic disparities and competing interests often exacerbate resource exploitation and environmental degradation. Limited access to alternative livelihood options, coupled with population growth and urban migration, intensifies pressure on fisheries resources. Furthermore, inadequate infrastructure, such as waste management facilities and sanitation services, exacerbates pollution problems, disproportionately impacting marginalized communities [15, 21, 22]. Addressing these socioeconomic challenges requires holistic approaches that prioritize community engagement, equitable resource allocation, and sustainable development strategies to foster resilience and promote the well-being of both people and the environment [23].
To address these challenges, this study aims to conduct a comprehensive investigation into the biological aspects of IPT in Lake Siombak. Through examinations of their presence, abundance, growth, reproduction, and feeding habits, we seek to provide insights for informed decision-making in fisheries resource management. By applying the precautionary principle and considering biological indicators, we aim to promote sustainable utilization practices and ensure the ecological and economic integrity of Lake Siombak's aquatic environment [24,25,26].
2 Methods
2.1 Location and sampling
This study was conducted in Lake Siombak, North Sumatra Province, from September 2018 to August 2019, focusing on eleven specific sampling stations (see Fig. 1). Gill nets with a 1-inch mesh size were utilized for sample collection, with observations conducted twice during high and low tides to capture variations in IPT distribution and behavior associated with tidal cycles. Sampling sites 1 to 8 were situated within Lake Siombak, covering different areas to encompass the lake's spatial heterogeneity, while sites 9 to 11 were positioned in tributaries outside the lake, providing insight into Indo-Pacific Tarpon (IPT) populations in adjacent freshwater habitats. During each sampling event, gill nets were deployed at predetermined locations within each sampling site, ensuring comprehensive coverage of the aquatic habitat. Trained personnel conducted sample collection from the gill nets, following standardized protocols (i.e., involved carefully removing the fish from the nets, minimizing any potential injury or stress during handling) to minimize handling stress on the captured fish.
2.2 Fish capture and measurement
IPT were captured using gill nets during both high and low tides. Upon capture, IPT were promptly measured for length and weight. Subsequently, each fish underwent dissection to determine its sex and maturity, while the stomach and intestine contents were preserved for further laboratory analysis. This involved the careful examination of reproductive organs to differentiate between male and female specimens, and the determination of the maturity stage based on the gonadal development. Additionally, the stomach and intestine contents were carefully extracted and preserved for subsequent laboratory analysis. This step aimed to unravel the dietary habits and preferences of the fish [27]. This analysis involved sorting and identifying the various food items present in the digestive tract. Morphological and taxonomical characteristics of each item were considered to identify prey items accurately. Special attention was given to discerning the different types of organisms, such as zooplankton, small fish, and other aquatic invertebrates, that constituted the diet of the IPT.
2.3 Data analysis
Data analysis related to growth parameters, frequency distribution, recruitment patterns, mortality, and exploitation rates were performed using the FAO-ICLARM Stock Assessment Tool II (FISAT II) [28]. Theoretical age (t0) using Pauly's (1980) formula:
where:
-
Lmax is the maximum observed length.
-
Lmin is the minimum observed length.
-
k is the growth rate coefficient.
The analysis of the relationship between length and weight refers to linear regression analysis [25], expressed as:
where:
-
W is the weight.
-
L is the length.
-
a and b are constants derived from the regression analysis.
Analysis of the size of the first gonadal mature fish refers to the Udupe method [29], calculated as:
where:
-
LGML is the length at which 50% of the fish have mature gonads.
-
LGonad is the length of individual fish at gonadal maturity.
-
nGonad is the number of individuals with mature gonads.
Meanwhile, the food analysis was carried out descriptively with the help of Microsoft Excel. Furthermore, statistical analysis was performed using PAST (version 4.07b).
2.4 Limitations
The use of gill nets for fish capture may introduce some bias in the sampling, as it primarily targets larger individuals and may not accurately represent the entire population. Additionally, the reliance on morphological and taxonomical characteristics for prey identification in the food analysis may introduce some subjectivity and potential misclassification of food items. Future research could address these limitations by employing a more diverse range of sampling methods and incorporating molecular techniques for prey identification, thereby enhancing the accuracy and reliability of the findings [30, 31].
3 Results and discussion
3.1 Distribution and abundance of Tarpon fish
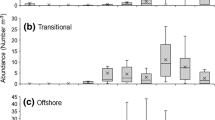
A total of 401 Indo-Pacific Tarpon (IPT) individuals were collected during the course of this study, as illustrated in Fig. 2a–c. The highest number of samples occurred in the center of the lake (sampling sites 3, 4, 5, 6, 7, and 8), while the number of samples from sampling sites near settlement areas and tributaries (sampling sites 1, 2, 9, 10, and 11) showed a low number of sample collections. The highest collection occurred in November 2018 with (n = 133 individuals, averaging 12 samples for each sampling), while the lowest was in April 2019 with only 4 individuals collected throughout this study. Notably, a discernible pattern emerged, with a majority of collections transpiring during the rainy season (August-January), constituting 67.33% (270 individuals), as opposed to the dry season, which accounted for 32.67% (131 individuals).
Gender distribution within the collected IPT specimens revealed a marked male predominance (Gender Ratio ≈ 4.99), comprising 83.29% (334 individuals), in contrast to 16.71% females (67 individuals). This skew towards males can be attributed to the behavioral inclination of female IPT, which tend to migrate to marine waters for spawning. The spawning season for this species spans the entire year and predominantly occurs in offshore areas. Consequently, the scarcity of female IPT in freshwater or brackish waters aligns with this migratory behavior. Consistent with prior research, the offshore breeding behavior of IPT manifests in transparent larvae resembling those of eels, characterized by forked tails [32, 33].
Non-metric Multidimensional Scaling (n-MDS) analysis was conducted to compare Indo-Pacific Tarpon populations between study sites and seasons (Supplementary Figure S1). The stress value of 0.1307 suggests that the MDS plot provides a reasonably good representation of the relationships between sites. Additionally, we performed cluster analysis using paired group (UPGMA) with Bray–Curtis similarity analysis to see the relationships between sampling sites and the distribution of IPT populations. The analysis revealed that sampling sites inside the lake exhibit a Bray–Curtis similarity of more than -0.5, indicating a higher degree of similarity in IPT populations compared to sites outside the lake. This suggests that IPT within Lake Siombak share more abundance compared to those in the tributaries outside the lake. The higher degree of similarity in IPT populations within Lake Siombak suggests that environmental factors or habitat characteristics within the lake may be promoting population homogeneity. This could be attributed to factors such as food availability, water quality, or habitat complexity, which are known to influence fish abundance and distribution [34,35,36]. Furthermore, the clustering pattern aligns with the idea of interconnected habitats within the lake, where fish movements and population dynamics are more closely linked.
3.2 The growth model of tarpon fish
The FISAT II analysis, employing the von Bertalanffy method, furnished critical insights into the growth dynamics of IPT in Lake Siombak. The maximum length (L∞) of IPT was determined as 49.32 cm, with a growth rate coefficient (K) of 0.51 per year and a theoretical age (to) of 0.0869 years. This K value places IPT within the realm of relatively moderate growth rates. As per Pauly's classification, fish with a high growth rate coefficient (K) exhibit rapid growth, achieving their maximum length swiftly, whereas those with a low growth rate take an extended duration to reach their peak size [37].
The monthly growth curve, illustrated in Fig. 3, further delineates the growth trajectory of IPT in Lake Siombak. Notably, fish under 5 years of age exhibit a rapid growth phase, with a subsequent deceleration in growth as they reach the age of 5 years, culminating in the attainment of maximum body length. This pattern signifies a fast early-age growth, transitioning into a slower pace with increasing age until the asymptotic length is reached. This trend aligns with broader observations, as demonstrated in other studies where young fish display a comparatively swifter growth rate, while adult fish exhibit a more gradual approach to achieving their asymptotic length [24].
3.3 The fish length-frequency distribution & length–weight relationship
The length-frequency distribution analysis of IPT in Lake Siombak revealed distinct patterns, with 33.34% of the specimens falling within the length group of 27.5–30.2 cm, while a smaller cohort inhabited the 13.7–16.5 cm range. Intriguingly, only a solitary fish was captured at a length of 38.5–41.2 cm. A closer examination of the length frequency distribution (see Supplementary Figure S2) exposed a notable trend: fish caught during the rainy season (August-January), ranging from 20 to 30 cm, tended to exhibit larger sizes compared to their counterparts in the dry season.
The IPT population in Lake Siombak predominantly comprised medium-sized, sexually immature individuals, as depicted in Fig. 4a. This observation aligns with the pelagic nature of IPT, which primarily inhabit marine waters and estuaries. The presence of juvenile IPT in estuarine zones with mangrove forests contrasts with the adult population's preference for marine environments, contributing to the scarcity of adult IPT in estuarine waters, including Lake Siombak and Belawan River [6, 32, 33].
Furthermore, the analysis of the length–weight relationship for IPT exhibited a robust correlation and determination (R value above 0.8) for both males and females. The calculated value of b, representing the slope of the relationship, was 2.773, indicating negative allometric growth. This is elucidated by the dominance of length growth over weight, substantiated by the fish's flat body shape. However, it's noteworthy that the level of gonad maturity could influence the value of b, with limited instances falling within Gonadal Maturity Level (GML) III and IV [24].
3.4 Reproduction & population recruitment
The predominant collection of IPT specimens in our study, coupled with the consistently observed presence of immature gonads in females, underscores a distinct reproductive scenario. The average gonadal maturity index, consistently below 0.5 across most months, except for an uptick in August to 0.67 (Fig. 4a–b), suggests a prevalence of sexually immature individuals. Furthermore, the analysis of the size of the first gonadal fish indicated a size of 40.10 cm, falling within the range of 39.30 cm to 40.89 cm. Notably, the largest collected IPT in Lake Siombak measured 38.5 cm, affirming the absence of spawned individuals. This collective evidence implies that the IPT population in Lake Siombak is composed of individuals that have not yet undergone spawning.
The gender-specific patterns of male and female IPT, both exhibiting immaturities, collectively indicate a lack of spawning occurrences. This can be attributed to the brackish nature of Lake Siombak, utilized by IPT solely for foraging and rearing, with spawning activities relegated to the marine environment. The association of mature IPT with marine waters is a well-established phenomenon, as adult IPT predominantly inhabit the sea, while juveniles are found in river mouths, deep bays, mangrove forests, and brackish swamps, contingent upon wave dynamics [38].
Examining the recruitment pattern of IPT in Lake Siombak revealed a notable peak in the 5th month of the study, notably January. This surge in recruitment is likely correlated with heightened food availability in the waters during this period, including abundant shrimp and crab populations [39]. Numerous studies have elucidated the multifaceted factors influencing fish growth, encompassing physiological and environmental conditions such as temperature, pH, salinity, geographical location, food availability, and sampling techniques [40,41,42,43,44,45,46,47].
3.5 Food and feeding habits
The examination of IPT stomach and intestinal contents yielded insights into the dietary preferences of this species, revealing its omnivorous nature (Fig. 4c–d). The analysis identified a diverse array of potential food sources, encompassing at least 10 types consumed by IPT. Notably, Rotifers and Cladocera constituted the majority of the gastric contents, each comprising 22% and 21%, respectively. Copepods and Chlorophyceae were also notable components, each contributing 15%. In terms of frequency, Cladocera emerged as the most frequently consumed food (35%), followed by Copepod (18%), and Shrimp (10%). The monthly average of stomach contents and food frequency collectively highlights the predominant role of zooplankton, specifically Cladocera and Copepods, in the diet of IPT in Lake Siombak. Daphnia and Diaptomus, identified as prominent food types, were consistently found in the stomachs of IPT, aligning with their abundance in Lake Siombak as reported in a previous study [39].
These findings underscore the versatility of IPT as an omnivorous species, adept at exploiting a varied diet. The reliance on zooplankton, particularly Cladocera and Copepods, indicates the significance of these components in the ecological niche of IPT in Lake Siombak. This dietary insight is crucial for understanding the trophic dynamics of IPT and its ecological role within the lake's ecosystem. Further investigations into the seasonal variations and availability of these zooplanktonic prey items would contribute to a comprehensive understanding of IPT feeding habits and its ecological interactions within the Lake Siombak environment.
3.6 Mortality and exploitation rate
The comprehensive evaluation of IPT mortality parameters revealed a total mortality (Z) estimate of 4.21 per year, with natural mortality (M) standing at 1.03 per year and fishing mortality (F) at 3.18 per year. The distinction between M and F values emphasizes the profound impact of fishing activities, as evidenced by the considerably higher F values compared to natural mortality. This discrepancy signals an elevated monthly fishing mortality rate in Lake Siombak, surpassing the natural deaths of IPT.
The exploitation rate (E), a crucial metric in understanding the impact of fishing activities on the IPT population, was calculated at approximately 0.75 per year. This notable value surpasses the annual optimum exploitation threshold of 0.5 per year. In accordance with the framework proposed by Sparre & Venema (1998), an E value exceeding 0.5 places the IPT population in Lake Siombak in the realm of overexploitation. This scenario implies a state where the rate of exploitation (E) exceeds the maximum sustainable level (MSY), leading to potential ecological imbalances.
The graphical representation (see Fig. 4e) visually underscores the critical threshold of overexploitation, emphasizing the urgency for management interventions to restore a sustainable equilibrium. Continuous unregulated fishing poses a substantial threat to the long-term viability of IPT resources in Lake Siombak, necessitating immediate attention to mitigate the adverse consequences of overexploitation. The findings underscore the imperative for implementing effective fisheries management strategies, such as regulatory measures and sustainable harvesting practices [48], to safeguard the IPT population and ensure the enduring ecological integrity of Lake Siombak. At the community level, raising awareness about the importance of sustainable fishing practices and the consequences of overexploitation can empower local fishermen to become stewards of their resources. Community-based initiatives could be established to promote responsible fishing behaviors, such as implementing catch limits, using selective fishing gear, and engaging in habitat restoration efforts. Additionally, collaboration between government agencies, local communities, and conservation organizations is essential to develop and enforce policies that regulate fishing activities, protect critical habitats, and promote sustainable resource management practices [49,50,51]. This multidimensional approach can help address the underlying drivers of overexploitation and foster a harmonious relationship between human activities and the natural environment, ensuring the long-term sustainability of IPT populations and the ecological health of Lake Siombak.
4 Conclusion
In conclusion, while this study provides valuable insights into the Indo-Pacific Tarpon (IPT) population in Lake Siombak, North Sumatra, Indonesia, there are opportunities for further research to enhance our understanding of this species. Including sampling sites that encompass both freshwater and estuarine habitats would provide a more comprehensive understanding of IPT dynamics, considering their migratory behavior between these environments. Future studies could investigate the specific environmental cues that trigger IPT migration and explore how these movements influence population dynamics and resource utilization. Additionally, examining the connectivity between freshwater and estuarine habitats would shed light on the broader ecological role of IPT within the aquatic ecosystem. By addressing these research gaps, we can better inform conservation and management strategies aimed at preserving the IPT population and maintaining the ecological integrity of Lake Siombak and its surrounding habitats.
Data availability
The datasets are available from the corresponding author on reasonable request.
References
Becking LE, Renema W, Santodomingo NK, Hoeksema BW, Tuti Y, de Voogd NJ. Recently discovered landlocked basins in Indonesia reveal high habitat diversity in anchialine systems. Hydrobiologia. 2011;677:89–105. https://doi.org/10.1007/s10750-011-0742-0.
Muhtadi A, Yulianda F, Boer M, Krisanti M, Rahmadya A. Santoso hydrodynamics of tropical tidal lake waters, Siombak Lake, Medan. Indonesia AACL Bioflux. 2020;13:2014–31.
Muhtadi A, Yunasfi Y, Leidonald R, Sandy SD, Junaidy A, Daulay AT. Status Limnologis Danau Siombak, Medan Sumatra Utara. OLDI. 2016;1:39. https://doi.org/10.14203/oldi.2016.v1i1.16.
Leidonald R, Lesmana I, Muhtadi A. Desrita Biodiversity flora and fauna in tropical tidal lake. IOP Conf Ser Earth Environ Sci. 2019;260: 012105. https://doi.org/10.1088/1755-1315/260/1/012105.
Muhtadi A, Yulianda F, Boer M, Krisanti M. Spatial distribution of mangroves in tidal lake ecosystem. IOP Conf Ser Earth Environ Sci. 2020;454: 012131. https://doi.org/10.1088/1755-1315/454/1/012131.
Khairul K, Wahyuningsih H, Jumilawati E. Distribusi dan pola pertumbuhan Ikan Bulan-bulan (Megalops cyprinoides Broussonet, 1782) di Sungai Belawan. J Perikan dan Kelaut. 2014;19(56):61.
Cahyani RT, Bija S, Sugi LTN. Karakteristik Ikan Bulan-bulan (Megalops cyprinoides) dan potensinya sebagai tepung untuk fortifikasi pangan. Teknol Pangan Media Inf dan Komun Ilm Teknol Pertan. 2020;11:182–91. https://doi.org/10.35891/tp.v11i2.2030.
Jawad LA, Al-Mamry JM. A record of the Indo-Pacific Tarpon Megalops cyprinoides (Broussonet, 1782) in Omani waters. Bull Fish Biol. 2017;17:75–7.
Coates D. Observations on the biology of Tarpon, Megalops cyprinoides (Broussonet) (Pisces : Megalopidae), in the Sepik River, Northern Papua New Guinea. Mar Freshw Res. 1987;38:529. https://doi.org/10.1071/MF9870529.
Rönnbäck P. The ecological basis for economic value of seafood production supported by mangrove ecosystems. Ecol Econ. 1999;29:235–52. https://doi.org/10.1016/S0921-8009(99)00016-6.
Cao Y, Langdon P, Chen X, Huang C, Yan Y, Yang J, Zeng L. Regime shifts in shallow lake ecosystems along an urban-rural gradient in Central China. Sci Total Environ. 2020;733: 139309. https://doi.org/10.1016/j.scitotenv.2020.139309.
Wiederkehr F, Wilkinson CL, Zeng Y, Yeo DCJ, Ewers RM, O’Gorman EJ. Urbanisation affects ecosystem functioning more than structure in tropical streams. Biol Conserv. 2020;249: 108634. https://doi.org/10.1016/j.biocon.2020.108634.
Omposunggu VM, Boleg A. Potensi dan Pengembangan Obyek Wisata Alam Terhadap Perekonomian Masyarakat di Danau Siombak, Paya Pasir, Medan Marelan. Sumatera Utara Regionomic. 2021;3:26–37.
Yulianda F, Muhtadi A, Boer M, Krisanti M, Wardiatno Y. Biological conservation of molluscs based on spatial and temporal distribution in tropical tidal lake Medan-Indonesia HAYATI. J Biosci. 2020;27:273–82. https://doi.org/10.4308/hjb.27.4.273.
Yusni E, Ifanda D. Analysis of heavy metal of copper (Cu) and lead (Pb) at Siombak Lake North Sumatera Province. IOP Conf Ser Earth Environ Sci. 2020;454: 012129. https://doi.org/10.1088/1755-1315/454/1/012129.
Sidle RC, Benson WH, Carriger JF, Kamai T. Broader perspective on ecosystem sustainability: Consequences for decision making. Proc Natl Acad Sci. 2013;110:9201–8. https://doi.org/10.1073/pnas.1302328110.
Ho LT, Goethals PLM. Opportunities and challenges for the sustainability of lakes and reservoirs in relation to the sustainable development goals (SDGs). Water. 2019;11:1462. https://doi.org/10.3390/w11071462.
Reid AJ, Carlson AK, Creed IF, Eliason EJ, Gell PA, Johnson PTJ, Kidd KA, MacCormack TJ, Olden JD, Ormerod SJ, et al. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biol Rev. 2019;94:849–73. https://doi.org/10.1111/brv.12480.
Silaen MA. Usaha Tambak Para Nelayan di Danau Siombak (Studi Kasus di Kelurahan Paya Pasir, Kecamatan Medan Marelan, Kota Medan). Medan: Universitas Sumatera Utara; 2018.
Kawarazuka N, Béné C. Linking small-scale fisheries and aquaculture to household nutritional security: an overview. Food Secur. 2010;2:343–57. https://doi.org/10.1007/s12571-010-0079-y.
Hasibuan NH, Hutabarat L, Khair H, Suryati I, Anggia Y, Manalu SP. Assessment of microplastic on tropical tidal lake waters in Medan (study cases-Siombak lake). IOP Conf Ser Earth Environ Sci. 2023;1239: 012025. https://doi.org/10.1088/1755-1315/1239/1/012025.
Muhtadi A, Leidonald R. The relationship between water quality and aquatic organisms in tidal lakes, Medan-Indonesia. IOP Conf Ser Earth Environ Sci. 2024;1302: 012061. https://doi.org/10.1088/1755-1315/1302/1/012061.
Hariram NP, Mekha KB, Suganthan V, Sudhakar K. Sustainalism: an integrated socio-economic-environmental model to address sustainable development and sustainability. Sustainability. 2023;15:10682. https://doi.org/10.3390/su151310682.
Yonvitner; Setyobudiandi, I.; Ernawati, Y.; Zairion; Mashar, A.; Muhtadi, A.; Akmal, S.G. Biologi Perikanan dan Pengelolaan. IPB Press. Bogor. Indonesia, 2020;
Sparre P, Venema SC. Introductions of tropical fish stock assessment. Rome: Food and Agriculture Organization of the United Nations; 1998.
Lane DE, Stephenson RL. Fisheries management science: the framework to link biological, economic, and social objectives in fisheries management. Aquat Living Resour. 1995;8:215–21. https://doi.org/10.1051/alr:1995021.
Baker R, Buckland A, Sheaves M. Fish gut content analysis: robust measures of diet composition. Fish Fish. 2014;15:170–7. https://doi.org/10.1111/faf.12026.
Gayanilo FCJ, Sparre P, Pauly D. FAO-ICLARM stock assessment tools II (FiSAT II) revised version user’s guide. FAO Comput Inf Ser. 2005;8:168.
Udupe KS. Statistical method of estimating the size at first maturity in fishes. Fishbyte, WorldFish Cent. 1986;4:8–10.
Russo T, Maiello G, Talarico L, Baillie C, Colosimo G, D’Andrea L, Di Maio F, Fiorentino F, Franceschini S, Garofalo G, et al. All is fish that comes to the net: metabarcoding for rapid fisheries catch assessment. Ecol Appl. 2021;31: e02273. https://doi.org/10.1002/eap.2273.
Kantoussan J, Laë R, Tine M. Review of the fisheries indicators for monitoring the impacts of fishing on fish communities. Rev Fish Sci Aquac. 2018;26:460–78. https://doi.org/10.1080/23308249.2018.1458282.
Adams AJ, Horodysky AZ, McBride RS, Guindon K, Shenker J, MacDonald TC, Harwell HD, Ward R, Carpenter K. Global conservation status and research needs for tarpons (Megalopidae), ladyfishes (Elopidae) and bonefishes (Albulidae). Fish Fish. 2014;15:280–311. https://doi.org/10.1111/faf.12017.
Froese, R.; Pauly, D. FishBase Available: www.fishbase.org (Accessed on 10 Feb, 2023).
Baltz DM, Fleeger JW, Rakocinski CF, McCall JN. Food, density, and microhabitat: factors affecting growth and recruitment potential of juvenile saltmarsh fishes. Environ Biol Fishes. 1998;53:89–103. https://doi.org/10.1023/A:1007471724608.
Bacheler NM, Paoli TJ, Schacht GM. Controls on abundance and distribution of yellow perch: predator, water quality, and density-dependent effects. Trans Am Fish Soc. 2011;140:989–1000. https://doi.org/10.1080/00028487.2011.603979.
Eklöv P. Effects of habitat complexity and prey abundance on the spatial and temporal distributions of Perch (Perca fluviatilis) and Pike (Esox lucius). Can J Fish Aquat Sci. 1997;54:1520–31. https://doi.org/10.1139/f97-059.
Pauly D. On the interrelationships between natural mortality, growth parameters, and mean environmental temperature in 175 fish stocks. ICES J Mar Sci. 1980;39:175–92. https://doi.org/10.1093/icesjms/39.2.175.
Genisa AS. Pengenalan Jenis-Jenis Ikan Laut Ekonomi penting di Indonesia. Oseana. 1999;24:17–38.
Muhtadi A, Pulungan A, Fadhlin A, Melati P, Sinaga RZ, Uliya R, Rizki M, Ifanda D, Leidonald R, et al. The dynamics of the plankton community on Lake Siombak, a tropical tidal lake in North Sumatra Indonesia Biodiversitas. J Biol Divers. 2020;21(3707):3719. https://doi.org/10.1057/biodiv/d210838.
Allen DM, Service SK, Ogburn-Matthews MV. Factors influencing the collection efficiency of estuarine fishes. Trans Am Fish Soc. 1992;121(234):244.
Rountrey AN, Coulson PG, Meeuwig JJ, Meekan M. Water temperature and fish growth: otoliths predict growth patterns of a marine fish in a changing climate. Glob Chang Biol. 2014;20:2450–8. https://doi.org/10.1111/gcb.12617.
Viadero RC. Factors affecting fish growth and production. In: Lehr Jay H, Keeley J, editors. In water Encyclopedia. New Jersey: John Wiley Sons; 2004.
Quist MC, Guy CS, Schultz RD, Stephen JL. Latitudinal comparisons of walleye growth in north america and factors influencing growth of walleyes in Kansas reservoirs. North Am J Fish Manag. 2003;23:677–92. https://doi.org/10.1577/M02-050.
Talbot C. Some aspects of the biology of feeding and growth in fish. Proc Nutr Soc. 1993;52:403–16. https://doi.org/10.1079/PNS19930081.
Claramunt RM, Wahl DH. The effects of abiotic and biotic factors in determining larval fish growth rates: a comparison across species and reservoirs. Trans Am Fish Soc. 2000;129:835–51. https://doi.org/10.1577/1548-8659(2000)129%3c0835:TEOAAB%3e2.3.CO;2.
Bœuf G, Payan P. How should salinity influence fish growth? Comp. Biochem Physiol Part C Toxicol Pharmacol. 2001;130:411–23. https://doi.org/10.1016/S1532-0456(01)00268-X.
Bertucci JI, Blanco AM, Sundarrajan L, Rajeswari JJ, Velasco C, Unniappan S. Nutrient regulation of endocrine factors influencing feeding and growth in fish. Front Endocrinol. 2019. https://doi.org/10.3389/fendo.2019.00083.
Liu OR, Thomas LR, Clemence M, Fujita R, Kritzer JP, McDonald G, Szuwalski C. An evaluation of harvest control methods for fishery management. Rev Fish Sci Aquac. 2016;24:244–63. https://doi.org/10.1080/23308249.2016.1161002.
Muawanah U, Yusuf G, Adrianto L, Kalther J, Pomeroy R, Abdullah H, Ruchimat T. Review of national laws and regulation in Indonesia in relation to an ecosystem approach to fisheries management. Mar Policy. 2018;91:150–60. https://doi.org/10.1016/j.marpol.2018.01.027.
Heikkila T, Gerlak AK. The formation of large-scale collaborative resource management institutions: clarifying the roles of stakeholders, science, and institutions. Policy Stud J. 2005;33:583–612. https://doi.org/10.1111/j.1541-0072.2005.00134.x.
Chen J-L, Hsu K, Chuang C-T. How do fishery resources enhance the development of coastal fishing communities: Lessons learned from a community-based sea farming project in Taiwan. Ocean Coast Manag. 2020;184: 105015. https://doi.org/10.1016/j.ocecoaman.2019.105015.
Acknowledgements
The author thanks Mr. Syahrik for his help in sample collection, and Azizah Fadhlin for assisting with laboratory analyses of fish food. Sincere appreciation is also extended to the two anonymous reviewers whose feedback improved this manuscript.
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Contributions
A.M. wrote the main manuscript text and collected biological data, N.M., A.S., A.S.A., H.A., and S.A.P. evaluated the data and contributed to manuscript development. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The research involving fish was conducted in compliance with ethical guidelines for the treatment of animals, and the study was approved by the ethics committee/IRB of Universitas Sumatera Utara.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Muhtadi, A., Maiyah, N., Suryanti, A. et al. Ecological dynamics and conservation implications of Indo-Pacific Tarpon (Megalops cyprinoides) in Lake Siombak, North Sumatra, Indonesia. Discov Anim 1, 6 (2024). https://doi.org/10.1007/s44338-024-00003-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44338-024-00003-4