Abstract
Quantitative MRI biomarkers are increasingly requested in clinical practice in the management of patients with chronic liver diseases, to evaluate fibrosis, steatosis, iron overloard and liver function. The proton density fat fraction (PDFF) is a quantitative biomarker for the quantification of steatosis, being the current noninvasive reference standard for steatosis grading. R2* is a biomarker of iron concentration and is bundled with PDFF. MR elastography measures liver stiffness through the quantification of mechanical waves generated by an external drive and it can be used to detect significant and advanced fibrosis. Hepatobiliary phase can provide information on residual liver function by quantitative or semiquantitative parameters. The functional liver imaging score is an emerging semiquantitative tool based on the three hepatobiliary phase features: liver parenchymal enhancement, biliary contrast excretion, and portal vein sign. This work discusses the role of quantitative MRI biomarkers, offering practical tips for the diagnosis of steatosis, iron, fibrosis and liver function.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chronic liver disease (CLD) and cirrhosis account for 2 million deaths worldwide each year, high burden of disability and increased health care utilization [1].
The epidemiology of CLD has changed over the past few years. On one side, HBV incidence has been reduced by vaccination and antiviral treatment programs and many patients with HCV infection have been treated successfully with direct acting antiviral therapy; on the other, metabolic dysfunction-associated steatotic liver disease (MASLD) [2] has become an important cause of CLD because of an increasing burden of obesity and metabolic syndrome.
In the recent years, thanks to advances in technology, quantitative imaging biomarkers of liver are increasingly emerging as important clinical tools in the management of patients with CLD, in some cases even replacing the need for liver biopsy for the diagnosis, quantitative staging and treatment monitoring.
In this article, we will review quantitative MRI biomarkers for fat and iron overload, liver fibrosis, as well as liver function estimation, showing their advantages and pitfalls and offering practical tips for the radiologists for their correct use and interpretation. We will also summarize the thresholds for levels of overload in different published studies.
Quantification of hepatic steatosis
Hepatic steatosis is characterized by an abnormal intracellular accumulation of triglycerides within hepatocytes, a common histologic characteristic of many liver diseases [3]. In particular, MASLD is the accumulation of fat in the liver in the absence of alcohol consumption, steatogenic medications and other causes of chronic liver disease.
Ultrasound has 85–87% sensitivity and 80–94% specificity for diagnosing steatosis that appears as increased hepatic echogenicity [4]. Several US techniques based on attenuation coefficient can estimate steatosis with a good correlation with the degree of steatosis, but high body mass index may affect accuracy in grading steatosis [5]. On noncontrast CT, steatosis is suggested when the density of the liver parenchyma is lower than that of the spleen. Attenuation difference at least − 10 HU or a hepatic-splenic attenuation ratio less than 0.8 are indicative of 20% hepatic steatosis, whereas difference of more than − 19 HU indicates 30% [6]. However, there are many confounder factors: deposition of copper, iron, amiodarone and glycogen can increase attenuation, whereas hepatitis, beam hardening and kilovoltage peak can reduce it.
MRI has demonstrated to be superior to US and CT in the evaluation of the presence of fat [7]. Conventional T1-weighted dual-echo in and opposed-phase MRI (same repetition time, but two different echo time values, respectively, 2.3 ms at 3T/4.6 ms at 1.5 T and 1.15 ms at 3T/2.3 ms at 1.5 T) allow a subjective assessment of hepatic steatosis with the loss of signal intensity between them that indicates the presence of fat as well as increased signal intensity with fat-only Dixon imaging. The proton density fat fraction (PDFF) is currently considered the most objective MRI sequence to evaluate tissue fat content. PDFF is the proportion of MR-visible fat proton to the sum of MR-visible fat and water protons [8]. A meta-analysis of pooled data collected from 28 studies has demonstrated excellent linearity, negligible bias and precision of PDFF across different field strengths, imager manufacturers and reconstruction methods [3].
The most used method is based on a chemical shift-encoded (CSE) multiecho spoiled gradient-recalled echo (mSGRE). Reconstruction algorithm provides calculated images, including water proton density of tissue water and proton density of tissue fat images and the PDFF map.
PDFF is expressed as absolute percentage (%) and ranges from 0 to 100% and it can be calculated by placing circular regions of interest (ROIs) on PDFF map [8]. In our experience, we typically place four ROIs of about 3 cm2 in diameter in the anterior, posterior, medial and lateral segments of the liver on PDFF maps, avoiding major vessels, liver edges and artifacts. The PDFF value across the entire liver is reported as the mean of the values of all four ROIs. A meta-analysis of 13 studies conducted to evaluate the diagnostic accuracy of MR-PDFF for the assessment of liver steatosis with histology as reference standard has summarized the PDFF values in different steatosis grades. The thresholds varied among individual studies, 3.42–6-90% for ≥ grade 1, 10.00–17.50% for ≥ grade 2 and 16.37–23.50% for grade 3 steatosis [9] (Fig. 1).
Among various imaging modalities, PDFF has been demonstrated the highest diagnostic precision in the quantitative evaluation of liver steatosis and has been validated against liver biopsy as reference standard [8]. Liver biopsy has indeed some limitations. It is invasive with a certain risk of bleeding, provides an assessment of a small portion of the liver and, therefore, it cannot be used for monitoring, whereas a control of MASLD and its complications has become a major public health priority, hence the need of developing noninvasive biomarkers [10].
A study showed that PDFF is associated with increased risks of malignant neoplasm, alcoholic disease, fibrosis and cirrhosis of liver and MASLD and that it is a causal mediator between potential risk factor and liver disease risks [11].
Quantification of liver iron
Iron is a normal component of many proteins of human organism as hemoglobin, myoglobin cytochrome and peroxidases. However, when it is in excess, it is toxic and it could to lead to end-organ damage [12]. Iron overload is a common problem and can be primary when caused by genetic diseases, most often hereditary hemochromatosis, or secondary mainly in patients who underwent transfusion therapy. These patients have high transferrin and ferritin levels and accumulation of free iron.
Liver is the main iron storage organ, the first to show iron overload and the only one to show a linear relationship between its iron concentration and total body iron. Iron overload generates the release of reactive oxygen species with toxic effects on the hepatocytes; if left untreated, patients with iron disorders will develop chronic liver disease.
Iron quantification, therefore, is crucial for the diagnosis and for patient monitoring after therapies, before tissue injury. Biopsy has a limited clinical utility in the quantification of liver iron concentration and has been largely replaced by noninvasive methods.
Among these, serologic markers (ferritin and transferrin) are sensitive, but not specific for the diagnosis since they may be elevated in other conditions. US cannot be used to evaluate liver iron overload, whereas the suspicion of iron overload is raised at non-enhanced CT when the liver attenuation is above the normal threshold of 65 HU, although this CT sign is not specific and can be observed in other conditions [13].
MRI provides a good accuracy for the diagnosis of iron overload, allowing to predict the risk of complications. The MR signal in iron-loaded liver decays more rapidly than does in the normal liver and this decay rate can be quantified by the transverse relaxation times (T2 and T2*) or rates (R2 and R2*). R2 and R2* are the preferred values as they well correlate with the liver iron concentration (LIC).
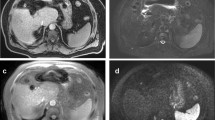
The transverse relaxation rate (R2*) is becoming the first-line biomarker for liver iron concentration and it is bundled with PDFF. Accurate R2* requires elimination of confounding effects of fat on PDFF acquisition [8]. Similar to fat fraction measurements, four ROIs of 3 cm2 are placed on maps and the mean R2* are reported. The R2* values can be converted to LIC, which estimate the iron content in milligrams per gram of dry liver tissue (mgFE/g) with field-specific equations (Figs. 2, 3).
According to the literature, a value below 1.8 mg/g is considered normal. Values between 1.8 and 3.2 mg/g are considered borderline, whereas 3.2–7.0 mg/g and 7.0–15.0 mg/g indicate mild and moderate overload, respectively. Finally, a value greater than 15 mg/g is indicative of severe deposition with increased risk of early death [12].
Monitoring of chelation therapy is based on liver iron content as assessed with MRI. Follow-up with iron quantification is recommended annually in patients with long-term transfusion. According to a calibration curve [13], the threshold of Liver R2* at 1.5 T for initiating therapy was reported > 270 sec−1, whereas the threshold indicates inadequate chelation was > 580 sec−1 [14].
Quantification of liver fibrosis
Chronic liver diseases lead over time to the development of fibrosis, which results in increased stiffness of the hepatic parenchyma. It is associated with several complications such as portal hypertension, variceal bleeding, hepatic encephalopathy and liver failure. Since studies have revealed that fibrosis is potentially reversible, its detection at an early stage and staging are essential for improving prognosis [15].
Elastography provides liver stiffness measurement and can be performed with ultrasound or MRI [16]. Ultrasound-based measurements can be obtained by vibration-controlled transient elastography (VCTE/Fibroscan) or with shear-wave elastography (SWE). Fibroscan has become the commonest noninvasive tool used as an alternative for biopsy. However, it has some limitations when it is used in patients with ascites, obesity and massive pleural effusion.
SWE gives a real-time visualization of selected area with a large surface area compared to Fibroscan and it can be used as an alternative with similar diagnostic performance, particularly in the presence of obesity and massive free ascites [17].
MR elastography is currently considerated the most accurate noninvasive method for detecting and staging hepatic fibrosis. It requires three components: a driver to generate mechanical waves, a phase-contrast pulse sequence with motion-encoding gradients to detect tissue motion, postprocessing to obtain wave images and inversion algorithms to produce elastograms.
The MR elastography equipment is composed by an active driver located outside of scanner room connected by a tube to a passive driver placed on the patient abdomen over the right lobe and held with an elastic trap that generates a continuous acoustic vibration transmitted at a frequency typically of 60 Hz. The magnitude and phase images are the raw data acquired during exam. The first ones provide anatomic information, while the second ones show shear-wave propagation. The postprocessing generates gray-scale and color elastograms without and with a 95% confidence map and a gray-scale and color wave images.
The gray-scale elastogram is generally used for quantitative evaluation in kiloPascal (kPa) and color elastogram for qualitative stiffness evaluation. Color elastograms depict violet parenchyma with normal livers, while depict parenchyma in green through red when liver stiffness is elevated.
ROIs are drawn on each image, sampling the largest portion of liver and mean liver stiffness measurements in kiloPascals are recorded. The overall mean stiffness is obtained by calculating the weighted arithmetic mean (Figs. 4, 5).
On magnitude images, areas within 1 cm of the edge, large vessels, extrahepatic tissues, fissures, masses and gallbladder fossa should be avoided, while on wave images, areas with wave distortion, low-amplitude waves and poor wave propagation should be avoided. Finally, on elastogram images crosshatched regions on the 95% confidence map should be excluded.
Several confounding factors can increase liver stiffness. It was demonstrated that in patients with chronic liver disease, liver stiffness may increase for a short time after meal. For this reason, fasting is recommended for patients 4–6 h before the exam. Other causes are acute inflammation, liver congestion and infiltrative processes. Elastogram could be non-diagnostic with a significant iron overload. In this case, spin echo sequences, that are less affected, can be used [8, 18].
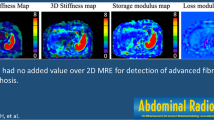
Singh et al. [19] have conducted a meta-analysis of individual participant data collected from 12 studies to evaluate diagnostic accuracy of MRE for staging liver fibrosis in patients with chronic liver disease. Excellent discriminative ability for the detection of advanced fibrosis and cirrhosis and good discriminative ability for the detection of any and significant fibrosis with a diagnostic performance comparable across patients with HCV, HBV, MASLD and alcoholic liver disease were shown.
Over the years, several studies have been conducted on single etiology groups (Table 1). In a systematic review and individual participant data with pooled analysis of diagnostic performance MR elastography in 9 studies with 232 patients with MASLD, this technique has showed from good to excellent diagnostic accuracy for discriminating grades of fibrosis with optimal cutoff for diagnosis of any (≥ stage 1), significant (≥ stage 2) and advanced fibrosis (≥ stage 3) and cirrhosis as 2.88, 3.54, 3.77 and 4.09 kPa, respectively [20].
Venkatesh et al. [21] have studied diagnostic performance of MRE for staging of liver fibrosis in chronic hepatitis B, getting optimal cutoff for diagnosis of ≥ F1, ≥ F2, ≥F3 and F4 as 2.74, 3.2, 3.7 and 4.33, respectively. They observed that the cutoff values for fibrosis and significant fibrosis were higher than those reported in chronic hepatitis C (2.42 kPa, 3.16 kPa) [22] and alcoholic liver fibrosis (2.20 kPa, 2.57 kPa) [23], whereas the cutoff values for advanced fibrosis and cirrhosis were lower than those reported in chronic hepatitis C (4.21 kPa and 6.20 kPa) [24].
Quantification of liver function
The introduction of hepatobiliary contrast agents, especially gadoxetic acid, has enabled a functional evaluation of hepatobiliary system. Gadoxetic acid has notably dual elimination, 50% is excreted by the kidneys and the other 50% is excreted into the biliary system. During the arterial and portal venous phases, gadoxetic acid is distributed into the vascular and extravascular spaces; after that, it is uptaken by organic anion transporters into hepatocytes and excreted in the biliary system during the transitional (3–5 min after injection) and hepatobiliary phases (20 min). Therefore, it allows the evaluation of liver parenchyma, hepatic vessels as well as liver excretory function, which it is now considered noninvasive biomarker of hepatobiliary disorders [25].
The estimation of liver function is a crucial tool for monitoring the progression of chronic liver disease, for determining therapeutic strategies and for the prevention of post-interventional hepatic failure.
Several quantitative parameters have been proposed to estimate hepatic function such relative liver enhancement (RLE), hepatic uptake index (HUI), contrast uptake index (CEI) and liver-to-spleen contrast index (LSI). These are based on the signal intensity on non-enhanced and hepatobiliary phases and are obtained through equation. All biomarkers have been shown a strong positive correlation with each other and to correlate with liver function parameters.
They are fairly accurate at differentiating patients with MELD score ≥ 15 versus patients with MELD < 15, where MELD score of 15 is the threshold for liver transplantation [26] [27].
Liver failure is the major cause of morbidity and mortality after liver resection. Several methods have been used to assess preoperative liver function, among these indocyanine green clearance has been widely used.
Kim et al. [28] demonstrated that gadoxetic acid-enhanced MRI is able to predict posthepatectomy liver insufficiency better than the indocyanine green clearance test. Moreover, RLE has demonstrated to be able to differentiate steatosis and steatohepatitis in patients with nonalcoholic fatty liver disease, allowing patients to be selected for biopsy [29].
The presence of liver fibrosis could be evaluated by using gadoxetic acid. Feier et al. [30] have shown that RLE is a good discriminator for the presence of fibrosis, whereas Watanabe et al. [31] have found that CEI is a good biomarker for the staging.
In the 2016, Bastati and colleagues [32] developed the functional liver imaging score (FLIS), a semiquantitative method, easier to assess function compared with other quantitative parameters due to the lack of complex quantification, being able to be easily included into routine clinical practice.
FLIS is based on the three hepatobiliary phase features on gadoxetate-disodium MRI: liver parenchymal enhancement, biliary contrast excretion, and portal vein sign, each ranging from 0 to 2 on an ordinal scale. In particular, liver parenchymal enhancement is compared to the right kidney and a score of 0, 1 or 2 is assigned if the liver is hypo-, iso- or hyperintense, respectively. For the biliary contrast excretion, it is assigned 0, 1 or 2 if there is no biliary contrast excretion, contrast in the intrahepatic ducts, or contrast in the common hepatic duct till the duodenum. Finally, portal vein sign is based on signal intensity of portal vein relative to the liver parenchyma and a score of 0, 1 or 2 is assigned if the portal vein is hyper-, iso- or hypointense. The three scores are summed, obtaining the FLIS (Fig. 6). Recent studies demonstrated that FLIS was associated with graft survival in patients who underwent transplantation and was linked with first decompensation and mortality in patients with advanced chronic liver disease. In a retrospective study of 265 patients, both decompensated and compensated patients with CLD with a reduced FLIS showed a three- to seven-fold higher risk of mortality, respectively, even after adjusting for established prognostic factors [33].
Axial and coronal contrast-enhanced DIXON images in the hepatobiliary phase. 63-year-old woman with primary sclerosing cholangitis. Liver parenchyma is isointense to the kidney (A). Portal vein isointense to the hepatic parenchyma (B) and biliary contrast excretion to the common bile duct (C). The sum of three scores is 4
It was also observed that FLIS and its three parameters have strong correlations with Child–Pugh scores, being able to stratify patients with chronic liver disease and liver cirrhosis according to the classification with a FLIS ≥ 5 as optimal cutoff for prediction of Child–Pugh A [34].
FLIS and its three parameters have also showed very strong correlation with ALBI grades [35]; however, Eryuruk et al. [36] have demonstrated that REI has a correlation with ALBI grades outperforming FLIS in predicting the ALBI grade.
It was shown that the addition of splenic craniocaudal diameter (a marker of portal hypertension) to FLIS refines the prediction of the composite endpoint hepatic/decompensation/death in patients with advanced chronic liver disease [37].
In a study of 502 patients who underwent preoperative gadoxetic acid-enhanced MRI, followed by HCC resection, FLIS has demonstrated to perform better than the MELD score, ALBI score, and ICG-R15 clearance to predict posthepatectomy liver failure [38].
Finally, it was subject of debate whether magnetic field strength influences quantitative MRI assessments, since 3T MRI is known to give higher contrast-noise ratios than 1.5 T for the liver parenchyma on contrast-enhanced MRI; however, it was demonstrated that FLIS is independent of field strength, exam parameters, scanner type and vendor (Table 2) [27].
Conclusions
MRI offers the possibility of a comprehensive evaluation of hepatobiliary disease with the use of qualitative and quantitative parameters, which represent relevant clinical tools in the management of patients with chronic liver disease, in many cases replacing liver biopsy for the diagnosis, quantitative staging and treatment monitoring.
Data and materials availability
Data sharing not applicable.
References
Moon AM, Singal AG, Tapper EB (2020) Contemporary epidemiology of chronic liver disease and cirrhosis. Clin Gastroenterol Hepatol 18(12):2650–2666. https://doi.org/10.1016/j.cgh.2019.07.060
Rinella ME, Lazarus JV, Ratziu V, Francque SM, Sanyal AJ, Kanwal F, Romero D, Abdelmalek MF, Anstee QM, Arab JP, Arrese M, Bataller R, Beuers U, Boursier J, Bugianesi E, Byrne CD, Narro GEC, Chowdhury A, Cortez-Pinto H, Cryer DR, Cusi K, El-Kassas M, Klein S, Eskridge W, Fan J, Gawrieh S, Guy CD, Harrison SA, Kim SU, Koot BG, Korenjak M, Kowdley KV, Lacaille F, Loomba R, Mitchell-Thain R, Morgan TR, Powell EE, Roden M, Romero-Gómez M, Silva M, Singh SP, Sookoian SC, Spearman CW, Tiniakos D, Valenti L, Vos MB, Wong VW, Xanthakos S, Yilmaz Y, Younossi Z, Hobbs A, Villota-Rivas M, Newsome PN, NAFLD Nomenclature consensus group (2024) A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Ann Hepatol 29(1):101133. https://doi.org/10.1016/j.aohep.2023.101133
Yokoo T, Serai SD, Pirasteh A, Bashir MR, Hamilton G, Hernando D, Hu HH, Hetterich H, Kühn JP, Kukuk GM, Loomba R, Middleton MS, Obuchowski NA, Song JS, Tang A, Wu X, Reeder SB, Sirlin CB, RSNA-QIBA PDFF Biomarker Committee (2018) Linearity, bias, and precision of hepatic proton density fat fraction measurements by using MR imaging: a meta-analysis. Radiology 286(2):486–498. https://doi.org/10.1148/radiol.2017170550
Hernaez R, Lazo M, Bonekamp S, Kamel I, Brancati FL, Guallar E, Clark JM (2011) Diagnostic accuracy and reliability of ultrasonography for the detection of fatty liver: a meta-analysis. Hepatology 54(3):1082–1090. https://doi.org/10.1002/hep.24452
Ferraioli G, Soares Monteiro LB (2019) Ultrasound-based techniques for the diagnosis of liver steatosis. World J Gastroenterol 25(40):6053–6062. https://doi.org/10.3748/wjg.v25.i40.6053
Piekarski J, Goldberg HI, Royal SA, Axel L, Moss AA (1980) Difference between liver and spleen CT numbers in the normal adult: its usefulness in pre- dicting the presence of diffuse liver disease. Radiology 137(3):727–729. https://doi.org/10.1148/radiology.137.3.6934563
Kalisz K, Navin PJ, Itani M, Agarwal AK, Venkatesh SK, Rajiah PS (2024) Multimodality imaging in metabolic syndrome: state-of-the-art review. Radiographics 44(3):e230083. https://doi.org/10.1148/rg.230083
Guglielmo FF, Barr RG, Yokoo T, Ferraioli G, Lee JT, Dillman JR, Horowitz JM, Jhaveri KS, Miller FH, Modi RY, Mojtahed A, Ohliger MA, Pirasteh A, Reeder SB, Shanbhogue K, Silva AC, Smith EN, Surabhi VR, Taouli B, Welle CL, Yeh BM, Venkatesh SK (2023) Liver fibrosis, fat, and iron evaluation with MRI and fibrosis and fat evaluation with US: a practical guide for radiologists. Radiographics 43(6):e220181. https://doi.org/10.1148/rg.220181
Qu Y, Li M, Hamilton G, Zhang YN, Song B (2019) Diagnostic accuracy of hepatic proton density fat fraction measured by magnetic resonance imaging for the evaluation of liver steatosis with histology as reference standard: a meta-analysis. Eur Radiol 29(10):5180–5189. https://doi.org/10.1007/s00330-019-06071-5
Kozaka K, Matsui O (2023) MRI biomarkers and their future impact on nonalcoholic fatty liver disease. Radiology 309(1):e232420. https://doi.org/10.1148/radiol.232420
Xia T, Du M, Li H, Wang Y, Zha J, Wu T, Ju S (2023) Association between liver MRI proton density fat fraction and liver disease risk. Radiology 309(1):e231007. https://doi.org/10.1148/radiol.231007.Erratum.In:Radiology309(2):e239027
Reeder SB, Yokoo T, França M, Hernando D, Alberich-Bayarri Á, Alústiza JM, Gandon Y, Henninger B, Hillenbrand C, Jhaveri K, Karçaaltıncaba M, Kühn JP, Mojtahed A, Serai SD, Ward R, Wood JC, Yamamura J, Martí-Bonmatí L (2023) Quantification of liver iron overload with MRI: review and guidelines from the ESGAR and SAR. Radiology 307(1):e221856. https://doi.org/10.1148/radiol.221856
Wood JC, Zhang P, Rienhoff H, Abi-Saab W, Neufeld EJ (2015) Liver MRI is more precise than liver biopsy for assessing total body iron balance: a comparison of MRI relaxometry with simulated liver biopsy results. Magn Reson Imaging 33(6):761–767. https://doi.org/10.1016/j.mri.2015.02.016
Labranche R, Gilbert G, Cerny M, Vu KN, Soulières D, Olivié D, Billiard JS, Yokoo T, Tang A (2018) Liver iron quantification with MR imaging: a primer for radiologists. Radiographics 38(2):392–412. https://doi.org/10.1148/rg.2018170079
Kromrey ML, Le Bihan D, Ichikawa S, Motosugi U (2020) Diffusion-weighted MRI-based virtual elastography for the assessment of liver fibrosis. Radiology 295(1):127–135. https://doi.org/10.1148/radiol.2020191498
Ozturk A, Olson MC, Samir AE, Venkatesh SK (2022) Liver fibrosis assessment: MR and US elastography. Abdom Radiol (NY) 47(9):3037–3050. https://doi.org/10.1007/s00261-021-03269-4
Osman AM, El Shimy A, Abd El Aziz MM (2020) 2D shear wave elastography (SWE) performance versus vibration-controlled transient elastography (VCTE/fibroscan) in the assessment of liver stiffness in chronic hepatitis. Insights Imaging 11(1):38. https://doi.org/10.1186/s13244-020-0839-y
Guglielmo FF, Venkatesh SK, Mitchell DG (2019) Liver MR elastography technique and image interpretation: pearls and pitfalls. Radiographics 39(7):1983–2002. https://doi.org/10.1148/rg.2019190034
Singh S, Venkatesh SK, Wang Z, Miller FH, Motosugi U, Low RN, Hassanein T, Asbach P, Godfrey EM, Yin M, Chen J, Keaveny AP, Bridges M, Bohte A, Murad MH, Lomas DJ, Talwalkar JA, Ehman RL (2014) Diagnostic performance of magnetic resonance elastography in staging liver fibrosis: a systematic review and meta-analysis of individual participant data. Clin Gastroenterol Hepatol 13(3):440-451.e6. https://doi.org/10.1016/j.cgh.2014.09.046
Singh S, Venkatesh SK, Loomba R, Wang Z, Sirlin C, Chen J, Yin M, Miller FH, Low RN, Hassanein T, Godfrey EM, Asbach P, Murad MH, Lomas DJ, Talwalkar JA, Ehman RL (2016) Magnetic resonance elastography for staging liver fibrosis in non-alcoholic fatty liver disease: a diagnostic accuracy systematic review and individual participant data pooled analysis. Eur Radiol 26(5):1431–1440. https://doi.org/10.1007/s00330-015-3949-z
Venkatesh SK, Wang G, Lim SG, Wee A (2014) Magnetic resonance elastography for the detection and staging of liver fibrosis in chronic hepatitis B. Eur Radiol 24(1):70–78. https://doi.org/10.1007/s00330-013-2978-8
Huwart L, Sempoux C, Salameh N et al (2007) Liver fibrosis: noninvasive assessment with MR elastography versus aspartate aminotransferase-to-platelet ratio index. Radiology 245:458–466. https://doi.org/10.1148/radiol.2452061673
Bensamoun SF, Leclerc GE, Debernard L, Cheng X, Robert L, Charleux F, Rhein C, Latrive JP (2013) Cutoff values for alcoholic liver fibrosis using magnetic resonance elastography technique. Alcohol Clin Exp Res 37(5):811–817. https://doi.org/10.1111/acer.12025
Ichikawa S, Motosugi U, Ichikawa T, Sano K, Morisaka H, Enomoto N, Matsuda M, Fujii H, Araki T (2012) Magnetic resonance elastography for staging liver fibrosis in chronic hepatitis C. Magn Reson Med Sci 11(4):291–297. https://doi.org/10.2463/mrms.11.291
Baleato-González S, Vilanova JC, Luna A, Menéndez de Llano R, Laguna-Reyes JP, Machado-Pereira DM, Bermúdez-Naveira A, Osorio-Vázquez I, Alcalá-Mata L, García-Figueiras R (2023) Current and advanced applications of gadoxetic acid-enhanced MRI in hepatobiliary disorders. Radiographics 43(4):e220087. https://doi.org/10.1148/rg.220087
Beer L, Mandorfer M, Bastati N, Poetter-Lang S, Tamandl D, Stoyanova DP, Elmer MC, Semmler G, Simbrunner B, Hodge JC, Sirlin CB, Reiberger T, Ba-Ssalamah A (2019) Inter- and intra-reader agreement for gadoxetic acid-enhanced MRI parameter readings in patients with chronic liver diseases. Eur Radiol 29(12):6600–6610. https://doi.org/10.1007/s00330-019-06182-z
Poetter-Lang S, Bastati N, Messner A, Kristic A, Herold A, Hodge JC, Ba-Ssalamah A (2020) Quantification of liver function using gadoxetic acid-enhanced MRI. Abdom Radiol (NY) 45(11):3532–3544. https://doi.org/10.1007/s00261-020-02779-x
Kim DK, Choi JI, Choi MH, Park MY, Lee YJ, Rha SE, Jung SE (2018) Prediction of posthepatectomy liver failure: MRI with hepatocyte-specific contrast agent versus indocyanine green clearance test. AJR Am J Roentgenol 211(3):580–587. https://doi.org/10.2214/AJR.17.19206
Bastati N, Feier D, Wibmer A, Traussnigg S, Balassy C, Tamandl D, Einspieler H, Wrba F, Trauner M, Herold C, Ba-Ssalamah A (2014) Noninvasive differentiation of simple steatosis and steatohepatitis by using gadoxetic acid-enhanced MR imaging in patients with nonalcoholic fatty liver disease: a proof-of-concept study. Radiology 271(3):739–747. https://doi.org/10.1148/radiol.14131890
Feier D, Balassy C, Bastati N, Fragner R, Wrba F, Ba-Ssalamah A (2016) The diagnostic efficacy of quantitative liver MR imaging with diffusion-weighted, SWI, and hepato-specific contrast- enhanced sequences in staging liver fibrosis–a multiparametric approach. Eur Radiol 26(2):539–546. https://doi.org/10.1007/s00330-015-3830-0
Watanabe H, Kanematsu M, Goshima S, Kondo H, Onozuka M, Moriyama N, Bae KT (2011) Staging hepatic fibrosis: comparison of gadoxetate disodium-enhanced and diffusion-weighted MR imaging–preliminary observations. Radiology 259(1):142–150. https://doi.org/10.1148/radiol.10100621
Bastati N, Wibmer A, Tamandl D, Einspieler H, Hodge JC, Poetter-Lang S, Rockenschaub S, Berlakovich GA, Trauner M, Herold C, Ba-Ssalamah A (2016) Assessment of orthotopic liver transplant graft survival on gadoxetic acid-enhanced magnetic resonance imaging using qualitative and quantitative parameters. Invest Radiol 51(11):728–734. https://doi.org/10.1097/RLI.0000000000000286
Bastati N, Beer L, Mandorfer M, Poetter-Lang S, Tamandl D, Bican Y, Elmer MC, Einspieler H, Semmler G, Simbrunner B, Weber M, Hodge JC, Vernuccio F, Sirlin C, Reiberger T, Ba-Ssalamah A (2020) Does the functional liver imaging score derived from gadoxetic acid-enhanced MRI predict outcomes in chronic liver disease? Radiology 294(1):98–107. https://doi.org/10.1148/radiol.2019190734
Lee HJ, Hong SB, Lee NK, Kim S, Seo HI, Kim DU, Han SY, Choo KS (2021) Validation of functional liver imaging scores (FLIS) derived from gadoxetic acid-enhanced MRI in patients with chronic liver disease and liver cirrhosis: the relationship between Child-Pugh score and FLIS. Eur Radiol 31(11):8606–8614. https://doi.org/10.1007/s00330-021-07955-1
Aslan S, Eryuruk U, Tasdemir MN, Cakir IM (2022) Determining the efficacy of functional liver imaging score (FLIS) obtained from gadoxetic acid-enhanced MRI in patients with chronic liver disease and liver cirrhosis: the relationship between Albumin-Bilirubin (ALBI) grade and FLIS. Abdom Radiol (NY) 47(7):2325–2334. https://doi.org/10.1007/s00261-022-03557-7
Eryuruk U, Tasdemir MN, Karasu HI, Aslan S (2024) Comparison of the efficacy of the gadoxetic acid MRI-derived relative enhancement index (REI) and functional liver imaging score (FLIS) in predicting liver function: validation with Albumin-Bilirubin (ALBI) grade. Abdom Radiol (NY). https://doi.org/10.1007/s00261-024-04324-6
Bastati N, Beer L, Ba-Ssalamah A, Poetter-Lang S, Ambros R, Kristic A, Lauber D, Pomej K, Binter T, Simbrunner B, Semmler G, Balcar L, Bican Y, Hodge JC, Wrba T, Trauner M, Reiberger T, Mandorfer M (2022) Gadoxetic acid-enhanced MRI-derived functional liver imaging score (FLIS) and spleen diameter predict outcomes in ACLD. J Hepatol 77(4):1005–1013. https://doi.org/10.1016/j.jhep.2022.04.032
Luo N, Huang X, Ji Y, Jin G, Qin Y, Xiang B, Su D, Tang W (2022) A functional liver imaging score for preoperative prediction of liver failure after hepatocellular carcinoma resection. Eur Radiol 32(8):5623–5632. https://doi.org/10.1007/s00330-022-08656-z
Funding
No funding was received to assist with the preparation of this manuscript. The author Roberto Cannella declares these fundings: co-funding by the European Union—FESR or FSE, PON Research and Innovation 2014–2020—DM 1062/2021.
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. The first draft of the manuscript was written by Mariasole Mazzola and all the authors commented on previous version of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors Mariasole Mazzola, Giulia Pilato, Antonino Andrea Blandino, Monica Milazzo, Francesco Matteini, Federica Vernuccio and Giuseppe Brancatelli have no competing, financial or non-financial, interests to declare that are relevant to the content of this article. The author Roberto Cannella declares support for attending meetings from Bracco and Bayer; research collaboration with Siemens Healthineers.
Ethical approval and consent
Not applicable.
Consent for publication
As written in JMIIR instructions for the authors, all the MRI images included in the present manuscript are completely anonymized, so they do not require consent for publication.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mazzola, M., Cannella, R., Pilato, G. et al. Quantitative liver MRI biomarkers: what the radiologist should know. J Med Imaging Intervent Radiol 11, 13 (2024). https://doi.org/10.1007/s44326-024-00015-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44326-024-00015-0