Abstract
Biopores formed by the actions of decaying roots of plants and soil fauna are essential for providing preferential growth pathways into deeper soil when the subsoil is compacted. Biopores help with rapid transportation of oxygen which increases microbial activity. These positive impacts help to increase the yield of crops even in adverse environmental conditions. However, the impact of biopores depends on various climatic and soil conditions. Understanding the mechanisms of different factors controlling the influences of biopores on the growth and development of plant roots is essential for getting more benefits in crop production.
While several reviews have explored biopore-root interactions in crop production, there remains a lack of systematic studies on the factors influencing these interactions. In this review, we discussed different factors affecting the impacts of biopores on various crops in different situations based on current literature. It is also explained how we can maximize these impacts for getting benefits in sustainable crop production.
Biopore improves the soil structure which has vital role on the growth of crops. On the other hand, clumping of roots within the biopores was also observed which may negatively affect crop growth. Similarly, biopores increase water infiltration as well as the leaching of nutrients. The influences of biopores depend on pore characteristics, abundance of biopores, moisture content, management practices, edaphic conditions, plant species etc. Understanding the factors influencing biopore-root interaction can contribute to improve root accessing into the deeper soil, enhancing their adaptation in stressful soil conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Feeding the large global population with the increasing scarce of natural resources is a huge challenge for humanity [1]. Although in recent years the production of food has been remarkably improved around the world, mainly due to extensive use of chemical fertilizers, pesticides, machineries and high-yielding varieties for different crops in agriculture [2]. Unfortunately, this intensification of food production leads to environmental concern because of severe loss of biodiversity and soil degradation, which is a big issue for the ensuring welfare of humanity [3, 4]. Sustainable crop production is essential for tackling this issue and it is the system in which maintaining the yield of crops by using low inputs and conserving natural resources [5]. Soil is the fundamental resource for crop production which regulates different processes such as water and nutrient movement, carbon sequestration and maintaining biodiversity from sessile flora to different soil fauna such as ground beetles, earthworms, moles etc. [6, 7].
Soil compaction is a big problem for crop production. About 68 million ha of land is degraded due to soil compaction worldwide [8]. The main cause for soil compaction is the extensive use of agricultural machineries [9]. Bulk density and penetration resistance increased in compacted soil and reduced the soil porosity and pore connectivity at the same time [10, 11]. Therefore, the roots of plants cannot efficiently use natural resources from the deeper soil due to the presence of a compacted layer [11, 12]. Schneider and Don [13] showed that subsoil contains more than half of the water and plant nutrients. Similarly, storage of organic caron is double in subsoil (20–200 cm) compared to topsoil (0–20 cm) [14]. Kautz et al. [15] reported that subsoil contributes around 10–80% of plant nutrients, particularly when topsoil is dry, and nutrients are deficient. Compacted subsoil prevents plant roots from accessing a significant portion of water and nutrients, leading to underutilization of these resources.
There are several ways for improving root growth in compacted soil. Firstly, traditional deep tillage can be used to improve rooting depth in hard soil [16]. However, the impact of deep tillage is short live [17] and this technique requires huge amounts of energy which increases the input cost [18]. On the other hand, soil biopores can be used as a preferential pathway for roots to grow into the deeper soil by providing less mechanical resistance which helps for accessing roots and the utilization of subsoil resources [19].
Biopores are the round shaped pores in soils that are created by the actions of soil living organisms mainly by the plants roots and earthworm’s activities [20]. Biopores are available across the soil profile from the topsoil to several meters depth of soil [20]. Many researchers have claimed that thick and deep roots can penetrate hard soil which after decomposition produced biopores [21, 22] that is also termed as bio-tillage [23]. Islam et al. [24] showed that biopores provide access into the deeper soil for rice roots through the compacted plough pan. Moreover, Colombi et al. [19] reported that biopores improved air permeability in compacted soil. Additionally, biopores improve the water infiltration which reduces the runoff and erosion after heavy rainfall [25].
The wall of biopores created from the decomposition of roots and earthworm’s activities is enriched with nutrient [26]. Dresemann et al. [27] found that uptake of N was greater by the roots of wheat plants grown in microcosms having biopores. Moreover, biopore acts as a hotspot for microbes [28, 29] because of having a higher availability of food and a proper environment in biopores [30]. The bacterial population was greater in the biopores wall than in the soil apart from biopores due to higher availability of carbon in bipores [31]. This improved microbial population is important for crop production because all the biogeochemical processes such as decomposition of organic matter, nutrient cycling, carbon sequestration etc. depend on microbial activities [32]. However, sometimes biopores have a negative influence on root growth particularly when the diameter is larger than the root diameters due to lack of root-soil contact [33, 34] which might reduce the water and nutrients uptake from the soil [35]. Although biopore has some negative impacts in some situations, it has great influence on improving crop productivity particularly in compacted soil. However, the impact of biopores depends on pore characteristics such as size, shape [24], abundance of pores [36], moisture content [37], compaction of soil [38], plant species [24], tillage condition [39, 40] and so on.
The influence of biopore for improving crop productivity can be maximized by using different techniques such as conservation tillage which ensure less disturbance of biopores [35], using perennial [20], tap and deep-rooted plants that have more capacity to create biopores [41], ensuring better crop establishment by timely planting of preceding and succeeding crop [39]. Maintaining reduced tillage is very important for conserving the abundance of biopores from the topsoil to subsoil [40].
Although the impact of biopore-root interaction depends on many factors, there are no comprehensive studies on various variables that influence the impact of biopores on improving crop productivity. This review aims to fill this gap by exploring the various factors affecting biopores influence on root development and crop yield. Additionally, how can we maximise the impact of biopore-root interaction on crop production is not presented in organized ways in the current literatures. Therefore, we also discussed how to maximize the impact of biopore by proper utilization of water and nutrients for sustainable crop production.
2 Methods
Relevant publications were identified using different online search resources such as Google Scholar, Wiley Online, Science Direct, Springer Link, Academia, PubMed, Taylor & Francis Online, and Research Gate platforms. The keywords used for collecting relevant publications were Biopore, Biopores, Macropores, Macropores, Pore, Soil structure, Bio-tillage, Root-soil ineteraction, Cracks, Soil burrowing, Root impacts, Soil structure, Soil pore and Porosity. Out of over 380 articles, 89 articles were found more relevant to tackle the issues mentioned in this review. The results from different field and laboratory studies were included in this review for better understanding of different factors and mechanisms related to root-biopore interactions. Additionally, attempts have been made to depict currently available and possible strategies for maximizing the impact of root-biopore interaction to improve crop productivity in a sustainable way.
3 What is biopore?
Soil pore system is formed by the process of spatial arrangement of soil separates as well as by the actions of biological activities such as growth of roots and activities of soil fauna. Islam et al. [21] noted that previously growing roots can create channels which are utilized by the roots of succeeding plants in subsoil. Additionally, Banfield et al. [28] claimed that soil fauna such as earthworms can create these channels (Fig. 1). Scientists have described that the soil pore which is formed by biological activities is termed as ‘biopore’ [20, 42]. However, biopores that are created and inhabited by earthworms are known as the drilosphere [43]. The drilosphere is not only the pores created by the earthworm’s activities, but it also includes the skin, cast, middens and gut [44]. Biopores are the part of soil macropores and it only consists of 1–10% of total soil volume [28]. On the other hand, Zhang et al. [45] showed that biopores contribute 30.1–58% and 66.3–74.1% of total macroporosity in the subsoil (20–30 cm depth) for rainfed maize and paddy fields, respectively.
The size of the biopores ranging from greater than 30 μm up to 5 mm [26, 46] depending on the factors such as crop type, compaction level, nutrient status of the soil [20]. Strong root systems of preceding crops can grow through compacted soil and after decomposition it produces biopores and this process is called bio-tillage [23]. A field study with sandy loam soil in Denmark conducted by Pulido-Moncada [47] showed that different cover crops such as lucerne and chicory can make many vertically well-connected pores (biopores) in hard subsoil. Moreover, many scientists have shown that different plants can create different types of biopores after decomposition of roots [21, 41]. One of the major alterations in soil structure is the creation of interconnected biopores through plant root degradation [48].
4 Difference between biopore and non-biopore
Soil pore system is crucial for transporting water and air which acts as niches and habitats for soil fauna and plant roots [26, 45]. However, this pore system varies in types such as biopores and non-biopores [49, 50] which are different in functions and morphological features [45]. The main differences between biopores and non-biopores are summarised below in Table 1.
5 Factors affecting biopore-root interaction
There are many factors that affect biopores-root interaction (Fig. 2).
5.1 Compactions
Soil bulk density controls the root growth in biopores, which is repeatedly addressed in many studies [12, 38] (Fig. 3). The roots of ryegrass seedlings tended to grow towards artificial biopores when soil strength was greater [56]. In a laboratory study with different bulk densities (1.4, 1.6 and 1.8 g cm−3) De Freitas et al. [57] reported that growth of maize roots was 68.8% of total roots in macropores when the bulk density was at or above 1.6 g cm−3 compared to 1.4 g cm−3 bulk density. On the other hand, growth of wheat roots was only 12.5% in biopores when the bulk density was lower than 1.2 g cm−3 [38]. Similarly, a medium-resolution microscopic analysis study by White and Kirkegaard [58] reported that about only 30–40% of the wheat roots on the inside of macropores in loose topsoil and 85–100% of the roots inside the compacted subsoil. Additionally, a column experiment by Xiong et al. [59] with high resolution X-ray CT image analysis reported that the number of biopores utilized by maize roots was 2.7 times higher in the compacted (1.6 g cm-3) soil than in the noncompacted (1.3 g cm-3) soil [59]. However, the number of biopores crossed by the maize roots was greater in non-compacted soil compared to compacted soil. The presence of biopores in loose soil did not increase the shoot and root biomass of wheat [37]. Therefore, higher bulk density stimulates the growth of roots towards biopores. The bulk density of soil is expressed as penetration resistance. Penetration resistance higher than 2 MPa has been recorded as critical value which reduces the root growth [60]. The growth of roots is prevented at 2.8 MPa penetration resistance in bulk soil then follows root growth in biopores [38].
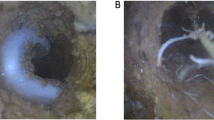
Picture showing rice roots growing through biopores in compacted plough pan in controlled greenhouse conditions. Image taken by high resolution camera. Source [24]
5.2 Size, shape and orientation of biopores
The effectiveness of biopores depends on the size of biopores. Landl et al. [36] reported that a biopore with a wider diameter than the root diameter is not effective for water and nutrient uptake by the plant roots due to lack of root-soil contact. Although more than 80% of the roots of maize were in large size (3.2 mm) biopores and depth of root length doubled, the growth of maize plants decreased compared to control without biopores [61]. Additionally, White and Kirkegaurd [58] found that the growth of wheat root has no impact if the biopores size is large. Similarly, Bouke et al. [37] showed that large size biopores limit the water and nutrient uptake due to the presence of large air gaps between the pore walls and plant roots. The length of different types of roots improved according to the size of biopores. In a field study, Han et al. [62] showed that an increased number of fine roots is present in small size biopores. On the other hand, more medium and larger root growth was observed in soil with large biopores. This study suggested that the size of the biopore is important for selection of crops with different root architectures for better utilization of biopores.
In addition to biopore size, orientation of the biopores (vertical or angled) determines the root growth within the biopores as well as re-entering roots into bulk soil. A laboratory study with ryegrass Hirth et al. [56] showed that about 13% of roots grew within the biopores before re-entering into the bulk soil when the biopores are vertically (90°) oriented [56]. On the other hand, this study also reported that 78% roots of ryegrass grew only a few millimetres before re-entering bulk soils when biopores were inclined at 40°. Similarly, a modelling study conducted by Landl et al. [63] found that 83–98% of plant roots remained in the biopores when pores were oriented at 90° and only 18–60% of the roots remained in biopores when the inclination was 40°.
Besides the size and orientation of the biopore, shape is also an important factor for the utilization of biopores for water and nutrient uptake from the deeper soil. A greenhouse study by Islam et al. [24] reported that the root length density of rice plants was 55% greater for elongated biopores compared to round biopores.
Moreover, pore wall roughness and microporosity in the pore wall affect the roots re-entering into the bulk soil. The roots of ryegrass re-enter into the bulk soil from the biopores more efficiently when the roughness and microporosity of the pore wall is greater [56]. A laboratory study using X-ray CT imaging technique showed that the tendency of maize roots to grow into biopores was greater when the wall of biopore is roughened [64].
5.3 Abundance
A column experiment with wheat plants showed that increasing the number of biopores increased root length density but did not increase the root biomass [65]. Therefore, it concluded that roots can be able to explore a greater volume of soil with the same root biomass which will be good for improving water and nutrient uptake from the greater depth of subsoil. This study also suggested that biopore not only increases the root length of plants but there is also a potential for increasing the yield of the plants. Setiawan and Rohmat [66] showed the infiltration of water depends on the number of biopores presence in the grass fields and it reduced the runoff from fields more efficiently when the abundance of biopores was greater [66].
5.4 Management practices/tillage impacts/climatic condition
Formation and maintenance of biopore mainly depend on the management and climatic condition of agricultural land [35]. In the frequently cultivated ecosystems, pore system is disrupted and damaged especially in plough layer [39, 40]. In contrast, bioppores are more stable for soil under natural conditions [39]. The impact of biopores is restricted to the deeper soil in annual cropping systems [67]. Moreover, tillage has great impact on the existence of biopores. Biopores can be continual from surface to subsoil in conservation tillage with perennial cropping systems [40]. The density of biopores is higher in zero or reduced tillage systems compared to conventional tillage practices [40] (Fig. 4) because of an increase in the number of earthworms, which encourages the creation of biopores [20]. Additionally, tillage destroys the opening of biopores, reducing their capacity to facilitate gas exchange with the atmosphere and water infiltration [20]. The utilization of biopores also depends on the climatic conditions of a region. A modelling and real experimental study by Landl et al. [36] showed that the uptake of water by the wheat plant increased during the drought period in the presence of biopores in compacted soil. The impact of biopores varies depending on climatic conditions or locations. The clumping of roots was recorded in biopores and roots were not re-entered into the bulk soil in Australia [33]. However, in Europe, it showed that roots were less clumped in the biopores, and they also observed the re-entry of roots into the bulk soil [26]. This clumping tendency of roots in the biopores differed due to the difference in climatic conditions. In Australia there is dry climatic condition [68] whereas in Germany there is a high precipitation rate. Therefore, the impact of biopores is more prominent in drought prone areas compared to wetland areas.

Adapted from Or et al. [40]
Image showing biopore abundance in different tillage systems. A Zero tillage; B Reduced tillage and C Conventional tillage fields.
5.5 Plant species
Plant species is also another important factor for getting benefits from the presence of biopores. A controlled greenhouse study by Islam et al. [21] showed that deep rooted rice plant created 20% more biopores compared to shallow rooted rice plants. Additionally, Islam et al. [24] reported that the deep rooted rice genotype produced 81% greater RLD, 30% more root numbers and 103% more branching in the subsoil with the presence of artificial biopores than the shallow rooting rice genotype. Moreover, perennial root systems are much more effective for the creation of more stable and continuous biopores compared to annual root systems [67]. Because the roots of perennial plants are better at penetrating compacted soil due to having thick and deep rooting ability [69]. In general, tap rooted crop species are more effective for drilling the soil than those of fibrous rooted plant species because roots of fibrous rooted plants remain mostly in shallower depth [70]. A field experiment conducted by Huang et al. [71] showed tap rooted precrop chicory increased the number of large sized (5–6 mm) biopores than the fibrous rooted tall fescue. The increased soil macroporosity under the tap rooted crops cultivation is frequently shown by different scientists [72,73,74]. The biopores utilized by the different root systems differently. The tap rooted oilseed rape roots grew in the centre of a biopore with only a few lateral roots attached to the pore wall, whereas fibrous rooted wheat roots grew around the biopore wall which helps for establishing higher root-soil contact [26].
5.6 Soil texture
Soil texture has a great influence on both root decomposition and the quality of biopores. Phalempin et al. [64] reported that maize roots decomposed quickly (> 78 days) in loam soil. However, some biopores were not completely decomposed after 216 days in the sandy soil. The authors also showed the biopores in loam soil were much more stable than biopores in sandy soil.
5.7 Moisture content
Soil moisture content is also a factor for determining the effects of biopores on the growth of plant roots. Because the penetration resistance of soil is modulated by the moisture content and the penetration resistance increases with the reduction of soil moisture. Volkmar [75] investigated that the growth of wheat roots was only observed in dry conditions (Negative water potential). On the other hand, root and shoot growth of spring wheat is reduced in the treatment with biopores in dry conditions compared to without biopores in well irrigated conditions [37]. A laboratory study with artificial macropores by Dresemann et al. [27] showed that the shoot dry weight of winter wheat increased by 66% when grown with the presence of biopores in low water content conditions. On the other hand, shoot dry matter was increased only by 39% when grown with the presence of macropores with high moisture content.
5.8 Depth of soil
Biopores in the deeper soil are more persistent than the biopores in the topsoil [20] because biopore system in the topsoil is frequently destroyed the tillage and machinery activities. On the other hand, biopores in the subsoil are usually intact due to less intervention by tillage activities [20]. In a study, Han et al. [76] concluded that pores size reduced with depth and the authors also claimed that smaller pores (< 0.2 mm) reduced more quickly than larger size pores (0.2–0.1 mm). Therefore, more studies are needed on the dynamics of the creation and utilizations of root-induced biopores in subsoil.
5.9 Nutrient concentration
Apart from different morphological properties and climatic conditions, higher nutrient availability may enhance the root growth in biopores. A study with artificial pores suggested that root growth was 6 times higher in pores with improved nutrient content than in pores filled with sand or empty pores [77]. A rhizotron study with wheat plants Bauke et al. [37] reported that improved water and phosphorous supply enhanced the shoot and root biomass production with the presence of biopores. However, there was no effects of biopores on the shoot and root biomass in water and phosphorous limited conditions.
6 How to improve the effects of biopore?
The improvement of biopore impact is very crucial for sustainable agriculture. The maximization of biopore benefits depends on the various factors such as soil type, crop selection, cropping duration, tillage history etc. A field study with different fodder crops Han et al. [41] showed that soil (Luvisols) with greater silt and clay contents and with bulk density of 1.45 g cm−3 is ideal for creating stable biopores compared to sandy and loose soil. However, soil (Ultisols) with lower bulk density (1.0 g cm−3) is not ideal for stable biopores formation [78] due to having greater shrinkage capacity of loose soil and at the same time, less compacted soil is more susceptible to being deformed after drying [79]. On the other hand, the soils which are extremely compacted (4 MPa) such as Kandosols which contains large amounts of kaolinite that acts as a cementing agent are not good for the creation of biopores by the plant roots and soil fauna because of greater soil strength [58]. Many studies have reported that deep and conventional tillage decreased biopores abundance [40, 42].
Despite the soil types, crop species selection is also playing a vital role in the formation and utilization of biopores. The ability to penetrate the compacted soil is different by the different plant species. Dicotyledonous plants are more effective for penetrating the hard soil compared to monocotyledonous plants [80] because of having greater diameter roots for dicots [81] which can exert greater radial pressure to grow in deeper [82]. A growth chamber study by Stirzaker et al. [81] showed that only larger (< 0.8 mm) roots of barley were able to penetrate the biopore wall which indicates that re-entry of roots from the biopores into the bulk soil depends on the root’s morphologies. Plants with large tap root systems can create more biopores than shallow and fibrous rooted plants [26, 74]. In a field study by Han et al. [41] reported that about 28% more biopores are produced by the tap rooted chicory than fibrous rooted tall fescue.
Cropping duration is another way of increasing the density of biopores. Around 6 times higher biopores density was recorded in the third year compared to the first year for different crops such as lucerne, chicory and tall fescue [41]. Therefore, by cultivating the perennial crops rather than annual crops is better for getting more biopores [20]. Yunusa and Newton [46] suggested that there are four main characteristics for good biopores formation plants: (1) thick and deep roots, (2) rapid growth rate and perennial in nature, (3) rapid decomposition of roots and (4) quick adaptivity to physical constraints.
The continuity of biopore is greater for the fields under no-tillage compared to conventional tillage [35]. Tillage operation destroys the vertical opening of biopores which affects the water and nutrient uptake as well as infiltration and exchange of gas in the biopore networks [20]. Additionally, root growth in subsoil increased in moist conditions than in dry conditions [83]. Timely planting will ensure better root growth [39] which might be helpful for the formation of more biopores by the plant roots. Therefore, maintaining proper moisture and timely planting in the fields are good strategies for getting a good number of biopores. Some other strategies can be adopted for increasing the population of earthworms in the fields by reducing tillage intensity and depth [84].
7 Research gaps and future works
There are many studies that have been recorded on the impacts of biopores on the growth of plants. However, the results are not consistent [19, 26, 33, 37]. Different plants can create different types of biopores in field conditions which vary in their shape, size and orientation due to having different root architectures [85]. Moreover, most of the studies in the literature are focused on how bulk density and penetration resistance affect biopores influences on plant growth [33, 56]. However, very little attempt has been done on how the characteristics of biopores and their pore walls affect the growth of roots in biopores due to opacity of the soils [86, 87]. Additionally, more studies should be conducted with contrasting root architectural genotypes of different crops to get higher benefits from the presence of biopores.
Some studies have reported that biopores abundance affects water infiltration and root growth [65, 66], but more studies need to be done to confirm the critical number of biopores per unit area, which will be good for maintaining water and nutrient dynamics for sustainable crop production.
The management of the field for maximizing the effects of biopores is still elucidated. For instance, Bauke et al. [37] showed that biopore has no impact in phosphorous and water limited conditions on the growth of roots and shoots for spring wheat. On the other hand, root and shoot growth of spring wheat significantly improved under sufficient water and nutrient conditions in the presence of biopores [88]. Therefore, the optimum water and nutrient status needs to be explored for maximizing the impact of biopores on the subsequent crops. However, appropriate water and nutrient status depends on the types of soils and climatic conditions [88] which also need to be further studied.
Changing the cropping pattern such as irrigated to upland for rice cultivation improved biopore production and enhanced the crop performance in upland conditions [21]. This needs more investigations in field conditions for improving the most important cereal crop rice production specially in the water limited environment.
Biopores improve the root length and root biomass for various crops [19, 24]. However, this improvement in root growth may accumulate lots of carbon which might affect the yields of plants. Therefore, more research should be conducted on the calculation of carbon balances for different plants growing with the presence or absence of biopores.
8 Conclusions
Biopores can be utilized by the plant roots as they improve the soil physical, chemical as well as biological properties. Biopores formed by the activities of plant roots and earthworms act as a preferential pathway for growing of roots in deeper soil especially in compacted soil. It also helps for utilization of water and nutrients from the subsoil during a drought period. On the other hand, leaching of nutrients from the topsoil to deeper soil may occur after heavy rainfall through biopores and sometimes roots may be clamped into the biopores, which will negatively affect the production of crops. The impact of biopores depends on many factors such as soil compaction, size of biopore, root characteristics, depth of soil, management practices for crop production and tillage operations. To maximize the effects of biopores, genotypes with deep and thick roots should be cultivated in loamy or clay soil using conservation tillage. Although biopore has great potential for improving crop production in sustainable ways, some research gaps need to be addressed related to optimum abundance of biopores, characteristics of biopores for improving root growth and management practices for maximizing the impact of biopore in real field conditions. This review will improve our understanding of how different factors influence biopore-root interaction ultimately enhances adaptation of various crops to promote higher yield in stressful environmental conditions.
Data availability
No datasets were generated or analysed during the current study.
Code availability
Not applicable.
References
Borlaug NE, Dowswell CR. Feeding a world of ten billion people: a 21st century challenge. In: Proceedings of the International Congress in the Wake of the Double Helix: From the Green Revolution to the Gene Revolution. Citeseer. 2003; 27:31.
Pretty J, Bharucha ZP. Sustainable intensification in agricultural systems. Ann Bot. 2014;114(8):1571–96.
Lanz B, Dietz S, Swanson T. The expansion of modern agriculture and global biodiversity decline: an integrated assessment. Ecol Econ. 2018;144:260–77.
Gomiero T. Soil degradation, land scarcity and food security: reviewing a complex challenge. Sustainability. 2016;8(3):281.
Brooker RW, Karley AJ, Newton AC, Pakeman RJ, Schöb C. Facilitation and sustainable agriculture: a mechanistic approach to reconciling crop production and conservation. Funct Ecol. 2016;30(1):98–107.
Adhikari K, Hartemink AE. Linking soils to ecosystem services—a global review. Geoderma. 2016;262:101–11.
Greiner L, Keller A, Grêt-Regamey A, Papritz A. Soil function assessment: review of methods for quantifying the contributions of soils to ecosystem services. Land Use Policy. 2017;69:224–37.
Batey T. Soil compaction and soil management—a review. Soil Use Manag. 2009;25(4):335–45. https://doi.org/10.1111/j.1475-2743.2009.00236.x.
Tracy SR, Black CR, Roberts JA, Mooney SJ. Soil compaction: a review of past and present techniques for investigating effects on root growth. J Sci Food Agric. 2011;91(9):1528–37. https://doi.org/10.1002/jsfa.4424.
Bottinelli N, Hallaire V, Goutal N, Bonnaud P, Ranger J. Impact of heavy traffic on soil macroporosity of two silty forest soils: initial effect and short-term recovery. Geoderma. 2014;217:10–7.
Islam MdD, Price AH, Hallett PD. Rhizosphere development under alternate wetting and drying in puddled paddy rice. Eur J Soil Sci. 2024;75(4):e13533. https://doi.org/10.1111/ejss.13533.
Valentine TA, Hallett PD, Binnie K, et al. Soil strength and macropore volume limit root elongation rates in many UK agricultural soils. Ann Bot. 2012;110(2):259–70. https://doi.org/10.1093/aob/mcs118.
Schneider F, Don A. Root-restricting layers in German agricultural soils. Part I: extent and cause. Plant Soil. 2019;442:433–51.
Guo Y, Amundson R, Gong P, Yu Q. Quantity and spatial variability of soil carbon in the conterminous United States. Soil Sci Soc Am J. 2006;70(2):590–600.
Kautz T, Amelung W, Ewert F, et al. Soil biology & biochemistry nutrient acquisition from arable subsoils in temperate climates: a review. Soil Biol Biochem. 2013;57:1003–22. https://doi.org/10.1016/j.soilbio.2012.09.014.
Bateman JC, Chanasyk DS. Effects of deep ripping and organic matter amendments on Ap horizons of soil reconstructed after coal strip-mining. Can J Soil Sci. 2011;81(1):113–20. https://doi.org/10.4141/S00-105.
Bezdicek DF, Beaver T, Granatstein D. Subsoil ridge tillage and lime effects on soil microbial activity, soil pH, erosion, and wheat and pea yield in the Pacific Northwest, USA. Soil Tillage Res. 2003;74(1):55–63. https://doi.org/10.1016/S0167-1987(03)00091-6.
Maraseni TN, Cockfield G, Apan A. A comparison of greenhouse gas emissions from inputs into farm enterprises in Southeast Queensland. Aust J Environ Sci Health Part A. 2007;42(1):11–8. https://doi.org/10.1080/10934520601015354.
Colombi T, Braun S, Keller T, Walter A. Artificial macropores attract crop roots and enhance plant productivity on compacted soils. Sci Total Environ. 2017;574:1283–93. https://doi.org/10.1016/J.SCITOTENV.2016.07.194.
Kautz T. Research on subsoil biopores and their functions in organically managed soils: a review. Renew Agric Food Syst. 2014;30(4):318–27. https://doi.org/10.1017/S1742170513000549.
Islam MdD, Price AH, Hallet PD. Effects of root growth of deep and shallow rooting rice cultivars in compacted paddy soils on subsequent rice growth. Rice Sci. 2023;30(5):459–72.
Chen G, Weil RR. Penetration of cover crop roots through compacted soils. Plant Soil. 2010;331:31–43.
Zhang Z, Peng X. Bio-tillage: a new perspective for sustainable agriculture. Soil Tillage Res. 2021;206:104844. https://doi.org/10.1016/j.still.2020.104844.
Islam MDD, Price AH, Hallett PD. Contrasting ability of deep and shallow rooting rice genotypes to grow through plough pans containing simulated biopores and cracks. Plant Soil. 2021;467(1–2):515–30. https://doi.org/10.1007/S11104-021-05131-4/FIGURES/5.
McMahon MJ, Christy AD. Root growth, calcite precipitation, and gas and water movement in fractures and macropores: A review with field observations. Published online 2000.
Athmann M, Kautz T, Pude R, Köpke U. Root growth in biopores-evaluation with in situ endoscopy. Plant Soil. 2013;371(1–2):179–90. https://doi.org/10.1007/s11104-013-1673-5.
Dresemann T, Athmann M, Heringer L, Kautz T. Effects of continuous vertical soil pores on root and shoot growth of winter wheat: a microcosm study. Agric Sci. 2018;9(6):750–64.
Banfield CC, Dippold MA, Pausch J, Hoang DTT, Kuzyakov Y. Biopore history determines the microbial community composition in subsoil hotspots. Biol Fertil Soils. 2017;53:573–88.
Athmann M, Kautz T, Banfield C, et al. Six months of L. terrestris L. activity in root-formed biopores increases nutrient availability, microbial biomass and enzyme activity. Appl Soil Ecol. 2017;120:135–42.
Kuzyakov Y, Blagodatskaya E. Microbial hotspots and hot moments in soil: concept & review. Soil Biol Biochem. 2015. https://doi.org/10.1016/j.soilbio.2015.01.025.
Uksa M, Schloter M, Endesfelder D, et al. Prokaryotes in subsoil—evidence for a strong spatial separation of different phyla by analysing co-occurrence networks. Front Microbiol. 2015;6:1269.
Jacoby R, Peukert M, Succurro A, Koprivova A, Kopriva S. The role of soil microorganisms in plant mineral nutrition—current knowledge and future directions. Front Plant Sci. 2017. https://doi.org/10.3389/fpls.2017.01617.
White RG, Kirkegaard JA. The distribution and abundance of wheat roots in a dense, structured subsoil—implications for water uptake. Plant Cell Environ. 2010;33(2):133–48. https://doi.org/10.1111/J.1365-3040.2009.02059.X.
Koebernick N, Daly KR, Keyes SD, et al. High-resolution synchrotron imaging shows that root hairs influence rhizosphere soil structure formation. New Phytol. 2017;216(1):124–35. https://doi.org/10.1111/NPH.14705.
Wendel AS, Bauke SL, Amelung W, Knief C. Root-rhizosphere-soil interactions in biopores. Plant Soil. 2022;475(1):253–77. https://doi.org/10.1007/s11104-022-05406-4.
Landl M, Schnepf A, Uteau D, et al. Modeling the impact of biopores on root growth and root water uptake. Vadose Zone J. 2019;18(1):1–20.
Bauke SL, Landl M, Koch M, et al. Macropore effects on phosphorus acquisition by wheat roots—a rhizotron study. Plant Soil. 2017;416(1–2):67–82. https://doi.org/10.1007/S11104-017-3194-0/FIGURES/7.
Atkinson JA, Hawkesford MJ, Whalley WR, Zhou H, Mooney SJ. Soil strength influences wheat root interactions with soil macropores. Plant Cell Environ. 2020;43(1):235–45. https://doi.org/10.1111/pce.13659.
Lucas M, Schlüter S, Vogel HJ, Vetterlein D. Soil structure formation along an agricultural chronosequence. Geoderma. 2019;350:61–72.
Or D, Keller T, Schlesinger WH. Natural and managed soil structure: on the fragile scaffolding for soil functioning. Soil Tillage Res. 2021;208:104912.
Han E, Kautz T, Perkons U, Lüsebrink M, Pude R, Köpke U. Quantification of soil biopore density after perennial fodder cropping. Plant Soil. 2015;394(1–2):73–85. https://doi.org/10.1007/S11104-015-2488-3/FIGURES/6.
Wuest SB. Soil biopore estimation: effects of tillage, nitrogen, and photographic resolution. Soil Tillage Res. 2001;62(3–4):111–6. https://doi.org/10.1016/S0167-1987(01)00218-5.
Bouché M, Kilbertius G, Reisinger O, Mourey A, Cancela da Fonseca JA. Humification et biodégradation. Published online 1975
Brown GG, Barois I, Lavelle P. Regulation of soil organic matter dynamics and microbial activityin the drilosphere and the role of interactionswith other edaphic functional domains. Eur J Soil Biol. 2000;36(3–4):177–98.
Zhang Z, Liu K, Zhou H, Lin H, Li D, Peng X. Three dimensional characteristics of biopores and non-biopores in the subsoil respond differently to land use and fertilization. Plant Soil. 2018;428(1–2):453–67. https://doi.org/10.1007/s11104-018-3689-3.
Yunusa IAM, Newton PJ. Plants for amelioration of subsoil constraints and hydrological control: the primer-plant concept. Plant Soil. 2003;257(2):261–81.
Pulido-Moncada M, Katuwal S, Munkholm LJ. Characterisation of soil pore structure anisotropy caused by the growth of bio-subsoilers. Geoderma. 2022;409:115571.
Angers DA, Caron J. Plant-induced changes in soil structure: processes and feedbacks. Biogeochemistry. 1998;42:55–72.
Abou Najm MR, Jabro JD, Iversen WM, Mohtar RH, Evans RG. New method for the characterization of three-dimensional preferential flow paths in the field. Water Resour Res. 2010. https://doi.org/10.1029/2009WR008594.
Katuwal S, Norgaard T, Moldrup P, Lamandé M, Wildenschild D, de Jonge LW. Linking air and water transport in intact soils to macropore characteristics inferred from X-ray computed tomography. Geoderma. 2015;237:9–20. https://doi.org/10.1016/j.geoderma.2014.08.006.
Zhang ZB, Zhou H, Zhao QG, Lin H, Peng X. Characteristics of cracks in two paddy soils and their impacts on preferential flow. Geoderma. 2014;228–229:114–21. https://doi.org/10.1016/j.geoderma.2013.07.026.
Luo L, Lin H, Li S. Quantification of 3-D soil macropore networks in different soil types and land uses using computed tomography. J Hydrol. 2010;393(1–2):53–64.
Koestel J, Larsbo M. Imaging and quantification of preferential solute transport in soil macropores. Water Resour Res. 2014;50(5):4357–78.
Bauke SL, von Sperber C, Siebers N, Tamburini F, Amelung W. Biopore effects on phosphorus biogeochemistry in subsoils. Soil Biol Biochem. 2016;111:157–65. https://doi.org/10.1016/j.soilbio.2017.04.012.
Haas C, Horn R. Impact of small-scaled differences in micro-aggregation on physico-chemical parameters of macroscopic biopore walls. Front Environ Sci. 2018;6:1–12.
Hirth JR, Mckenzie BM, Tisdall JM. Ability of seedling roots of Lolium perenne L. to penetrate soil from artificial biopores is modified by soil bulk density, biopore angle and biopore relief. Plant Soil. 2005;272:327–36. https://doi.org/10.1007/s11104-004-5764-1.
De Freitas PL, Zobel RW, Synder VA. Corn root growth in soil columns with artificially constructed aggregates. Crop Sci. 1999;39(3):725–30.
White RG, Kirkegaard JA. The distribution and abundance of wheat roots in a dense, structured subsoil—implications for water uptake. Plant Cell Environ. 2010;33(2):133–48. https://doi.org/10.1111/j.1365-3040.2009.02059.x.
Xiong P, Zhang Z, Wang Y, Peng X. Variable responses of maize roots at the seedling stage to artificial biopores in noncompacted and compacted soil. J Soils Sediments. 2022;22(4):1155–64.
Bengough AG, McKenzie BM, Hallett PD, Valentine TA. Root elongation, water stress, and mechanical impedance: a review of limiting stresses and beneficial root tip traits. J Exp Bot. 2011;62(1):59–68. https://doi.org/10.1093/jxb/erq350.
Passioura JB, Stirzaker RJ. Feedforward responses of plants to physically inhospitable soil. Int Crop Sci I. 1993. https://doi.org/10.2135/1993.internationalcropscience.c114.
Han E, Kautz T, Huang N, Köpke U. Dynamics of plant nutrient uptake as affected by biopore-associated root growth in arable subsoil. Plant Soil. 2017;415(1–2):145–60. https://doi.org/10.1007/s11104-016-3150-4.
Landl M, Huber K, Schnepf A, Vanderborght J. A new model for root growth in soil with macropores. Plant Soil. 2016;415:99–116. https://doi.org/10.1007/s11104-016-3144-2.
Phalempin M, Landl M, Wu GM, Schnepf A, Vetterlein D, Schlüter S. Maize root-induced biopores do not influence root growth of subsequently grown maize plants in well aerated, fertilized and repacked soil columns. Soil Tillage Res. 2022;221:105398.
Koch M, Boselli R, Hasler M, Zörb C, Athmann M, Kautz T. Root and shoot growth of spring wheat (Triticum aestivum L.) are differently affected by increasing subsoil biopore density when grown under different subsoil moisture. Biol Fertil Soils. 2021;57:1155–69.
Setiawan I, Rohmat D. Test the effectiveness of biopore in the framework of eco-campus development at Universitas Pendidikan Indonesia. In: IOP Conference Series: Earth and Environmental Science. Vol 286. IOP Publishing; 2019:12015.
Benjamin JG, Mikha M, Nielsen DC, Vigil MF, Calderon F, Henry WB. Cropping intensity effects on physical properties of a no-till silt loam. Soil Sci Soc Am J. 2007;71(4):1160–5.
Morand DT. The world reference base for soils (WRB) and soil taxonomy: an appraisal of their application to the soils of the Northern Rivers of New South Wales. Soil Res. 2013;51(3):167–81.
Yunusa IAM, Mele PM, Rab MA, Schefe CR, Beverly CR. Priming of soil structural and hydrological properties by native woody species, annual crops, and a permanent pasture. Aust J Soil Res. 2002. https://doi.org/10.1071/SR01038.
Blanco-Canqui H, Shaver TM, Lindquist JL, et al. Cover crops and ecosystem services: insights from studies in temperate soils. Agron J. 2015;107(6):2449–74.
Huang N, Athmann M, Han E. Biopore-induced deep root traits of two winter crops. Agriculture. 2020;10(12):634.
Kautz T, Stumm C, Kösters R, Köpke U. Effects of perennial fodder crops on soil structure in agricultural headlands. J Plant Nutr Soil Sci. 2010;173(4):490–501.
Lesturgez G, Poss R, Hartmann C, Bourdon E, Noble A, Ratana-Anupap S. Roots of Stylosanthes hamata create macropores in the compact layer of a sandy soil. Plant Soil. 2004;260:101–9.
McCallum MH, Kirkegaard JA, Green TW, et al. Improved subsoil macroporosity following perennial pastures. Aust J Exp Agric. 2004;44(3):299–307. https://doi.org/10.1071/EA03076.
Volkmar KM. Effects of biopores on the growth and N-uptake of wheat at three levels of soil moisture. Can J Soil Sci. 1996;76(4):453–8.
Han E, Kirkegaard JA, White R, et al. Deep learning with multisite data reveals the lasting effects of soil type, tillage and vegetation history on biopore genesis. Geoderma. 2022;425:116072.
McKee KL. Root proliferation in decaying roots and old root channels: a nutrient conservation mechanism in oligotrophic mangrove forests? J Ecol. 2001;89(5):876–87.
Dörner J, Sandoval P, Dec D. The role of soil structure on the pore functionality of an ultisol. J Soil Sci Plant Nutr. 2010;10(4):495–508. https://doi.org/10.4067/S0718-95162010000200009.
Peng X, Horn R, Smucker A. Pore shrinkage dependency of inorganic and organic soils on wetting and drying cycles. Soil Sci Soc Am J. 2007;71(4):1095–104. https://doi.org/10.2136/SSSAJ2006.0156.
Chen G, Weil RR. Penetration of cover crop roots through compacted soils. Plant Soil. 2009;331:31–43. https://doi.org/10.1007/s11104-009-0223-7.
Stirzaker RJ, Passioura JB, Wilms Y. Soil structure and plant growth: impact of bulk density and biopores. Plant Soil. 1996;185(1):151–62. https://doi.org/10.1007/BF02257571.
Oades JM. The role of biology in the formation, stabilization and degradation of soil structure. In: Brussaard L, Kooistra MJ, editors. Soil structure/soil biota interrelationships. Amsterdam: Elsevier; 1993. p. 377–400.
Glinski J. Soil physical conditions and plant roots. Boca Raton: CRC Press; 2018.
Chan KY. An overview of some tillage impacts on earthworm population abundance and diversity—implications for functioning in soils. Soil Tillage Res. 2001;57(4):179–91.
Pagenkemper SK, Peth S, Puschmann DU, Horn R. Effects of root-induced biopores on pore space architecture investigated with industrial X-ray computed tomography. Adv Tomogr Imaging. 2015. https://doi.org/10.2136/sssaspecpub61.c4.
Haling RE, Brown LK, Bengough AG, et al. Root hair length and rhizosheath mass depend on soil porosity, strength and water content in barley genotypes. Planta. 2014;239:643–51.
Whalley WR, Watts CW, Gregory AS, Mooney SJ, Clark LJ, Whitmore AP. The effect of soil strength on the yield of wheat. Plant Soil. 2008;306:237–47.
Cates AM, Sanford GR, Good LW, Jackson RD. What do we know about cover crop efficacy in the North Central United States? J Soil Water Conserv. 2018;73(6):153A-157A.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
M.D.I. conceptualization; design and structure of manuscript; drawing figures; and writing original draft. B.I.B., Y.H. and M.Z.U.K. editing, critical revision. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Islam, M.D., Binte, B.I., Hazzazi, Y. et al. Factors affecting biopore-root interaction: a review. Discov Agric 2, 67 (2024). https://doi.org/10.1007/s44279-024-00083-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44279-024-00083-6