Abstract
Our technologically advanced civilization has made sensors an essential component. They have potential uses in the pharmaceutical sector, clinical analysis, food quality control, environmental monitoring, and other areas. One of the most active fields of analytical chemistry research is the fabrication of electrochemical sensors. An intriguing area of electroanalytical chemistry is the modification of electrodes using polymeric films. Due to their benefits, which include high adhesion to the electrode surface, chemical stability of the coating, superior selectivity, sensitivity, and homogeneity in electrochemical deposition, polymer-modified electrodes have attracted a great deal of interest in the electroanalytical sector. Conducting polymers are an important material for sensing devices because of their fascinating features, which include high mechanical flexibility, electrical conductivity, and the capacity to be electrochemically converted between electronically insulating and conducting states. Tannin or lignin nanomaterials can be an inter-linker leading to flexible and functional polymeric networks. There is a continuing demand for fast and simple analytical methods for the determination of many clinically important biomarkers, food additives, environmental pollutants etc. This review in a comprehensive way summarizes and discusses the various metal oxide and sulfide-incorporated tannin and lignin scaffolds using electrochemical sensing and biosensing.
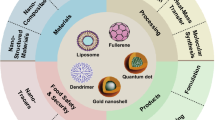
Graphical abstract





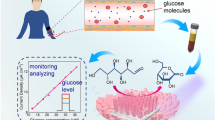
(Reproduced from reference [95] with permission from Wiley online library)





Similar content being viewed by others
Data availability
Data will be available on request.
References
R.W. Murray, Chemical modification of electrodes (1983)
J.M. Zen, A.S. Kumar, D.M. Tsai, Recent updates of chemically modified electrodes in analytical chemistry. Electroanalysis 15, 1073–1087 (2003). https://doi.org/10.1002/elan.200390130
M.A. Schneeweiss, H. Hagenström, M.J. Esplandiu, D.M. Kolb, Electrolytic metal deposition onto chemically modified electrodes. Appl. Phys. A 69, 537–551 (1999). https://doi.org/10.1007/s003399900154
P.R. Moses, L. Wier, R.W. Murray, Chemically modified tin oxide electrode. Anal. Chem. 47, 1882–1886 (1975). https://doi.org/10.1021/ac60362a043
D.C. Tiwari, R. Jain, S. Sharma, Electrochemically deposited polyaniline/polypyrrole polymer film modified electrodes for determination of furazolidone drug (2007). https://www.researchgate.net/publication/239573370
A. Moreno, M.H. Sipponen, Lignin-based smart materials: a roadmap to processing and synthesis for current and future applications. Mater. Horiz. 7, 2237–2257 (2020). https://doi.org/10.1039/D0MH00798F
J.K. Hammitt, R.J. Lempert, M.E. Schlesinger, A sequential-decision strategy for abating climate change. Nature 357, 315–318 (1992). https://doi.org/10.1038/357315a0
D. Dasgupta, D. Ghosh, S. Bandhu, D.K. Adhikari, Lignocellulosic sugar management for xylitol and ethanol fermentation with multiple cell recycling by Kluyveromyces marxianus IIPE453. Microbiol. Res. 200, 64–72 (2017). https://doi.org/10.1016/j.micres.2017.04.002
C.O. Tuck, E. Pérez, I.T. Horváth, R.A. Sheldon, M. Poliakoff, Valorization of biomass: deriving more value from waste. Science 337(2012), 695–699 (1979). https://doi.org/10.1126/science.1218930
W. Bao, D.M. O’Malley, R. Whetten, R.R. Sederoff, A laccase associated with lignification in loblolly pine xylem. Science 260(1993), 672–674 (1979). https://doi.org/10.1126/science.260.5108.672
Y. Wang, M. Chantreau, R. Sibout, S. Hawkins, Plant cell wall lignification and monolignol metabolism. Front. Plant Sci. (2013). https://doi.org/10.3389/fpls.2013.00220
A.J. Ragauskas, G.T. Beckham, M.J. Biddy, R. Chandra, F. Chen, M.F. Davis, B.H. Davison, R.A. Dixon, P. Gilna, M. Keller, P. Langan, A.K. Naskar, J.N. Saddler, T.J. Tschaplinski, G.A. Tuskan, C.E. Wyman, Lignin valorization: improving lignin processing in the biorefinery. Science (1979) (2014). https://doi.org/10.1126/science.1246843
D. Yiamsawas, G. Baier, E. Thines, K. Landfester, F.R. Wurm, Biodegradable lignin nanocontainers. RSC Adv. 4, 11661–11663 (2014). https://doi.org/10.1039/C3RA47971D
S. Sen, S. Patil, D.S. Argyropoulos, Thermal properties of lignin in copolymers, blends, and composites: a review. Green Chem. 17, 4862–4887 (2015). https://doi.org/10.1039/C5GC01066G
W. Yang, E. Fortunati, F. Dominici, G. Giovanale, A. Mazzaglia, G.M. Balestra, J.M. Kenny, D. Puglia, Effect of cellulose and lignin on disintegration, antimicrobial and antioxidant properties of PLA active films. Int. J. Biol. Macromol. 89, 360–368 (2016). https://doi.org/10.1016/j.ijbiomac.2016.04.068
E. Larrañeta, R.E.M. Lutton, A.D. Woolfson, R.F. Donnelly, Microneedle arrays as transdermal and intradermal drug delivery systems: materials science, manufacture and commercial development. Mater. Sci. Eng. R. Rep. 104, 1–32 (2016). https://doi.org/10.1016/j.mser.2016.03.001
M. Yousuf, A. Mollah, P. Palta, T.R. Hess, R.K. Vempati, D.L. Cocke, Chemical and physical effects of sodium lignosulfonate superplasticizer on the hydration of portland cement and solidification/stabilization consequences. Cem. Concr. Res. 25, 671–682 (1995). https://doi.org/10.1016/0008-8846(95)00055-H
A. Hasan, P. Fatehi, Cationic kraft lignin-acrylamide as a flocculant for clay suspensions: 1. Molecular weight effect. Sep. Purif. Technol. 207, 213–221 (2018). https://doi.org/10.1016/j.seppur.2018.06.047
S. Tan, D. Liu, Y. Qian, J. Wang, J. Huang, C. Yi, X. Qiu, Y. Qin, Towards better UV-blocking and antioxidant performance of varnish via additives based on lignin and its colloids. Holzforschung 73, 485–491 (2019). https://doi.org/10.1515/hf-2018-0134
D. Kai, M.J. Tan, P.L. Chee, Y.K. Chua, Y.L. Yap, X.J. Loh, Towards lignin-based functional materials in a sustainable world. Green Chem. 18, 1175–1200 (2016). https://doi.org/10.1039/C5GC02616D
L. Zhai, Stimuli-responsive polymer films. Chem. Soc. Rev. 42, 7148 (2013). https://doi.org/10.1039/c3cs60023h
A. Lendlein, O.E.C. Gould, Reprogrammable recovery and actuation behaviour of shape-memory polymers. Nat. Rev. Mater. 4, 116–133 (2019). https://doi.org/10.1038/s41578-018-0078-8
A. Arbenz, L. Avérous, Synthesis and characterization of fully biobased aromatic polyols – oxybutylation of condensed tannins towards new macromolecular architectures. RSC Adv. 4, 61564–61572 (2014). https://doi.org/10.1039/C4RA10691A
A. Arbenz, L. Avérous, Chemical modification of tannins to elaborate aromatic biobased macromolecular architectures. Green Chem. 17, 2626–2646 (2015). https://doi.org/10.1039/C5GC00282F
H.A.M. Bacelo, S.C.R. Santos, C.M.S. Botelho, Tannin-based biosorbents for environmental applications: a review. Chem. Eng. J. 303, 575–587 (2016). https://doi.org/10.1016/j.cej.2016.06.044
P. Schofield, D.M. Mbugua, A.N. Pell, Analysis of condensed tannins: a review. Anim. Feed Sci. Technol. 91, 21–40 (2001). https://doi.org/10.1016/S0377-8401(01)00228-0
H. Pan, Y. Zhong, Z. Zhang, C. Pan, Characteristics, industrial application, and prospects of continuous extrusion technology. Res. Appl. Mater. Sci. (2019). https://doi.org/10.33142/msra.v1i1.667
T. Ogata, S. Morisada, Y. Oinuma, Y. Seida, Y. Nakano, Preparation of adsorbent for phosphate recovery from aqueous solutions based on condensed tannin gel. J. Hazard. Mater. 192, 698–703 (2011). https://doi.org/10.1016/j.jhazmat.2011.05.073
A.-K. Koopmann, C. Schuster, J. Torres-Rodríguez, S. Kain, H. Pertl-Obermeyer, A. Petutschnigg, N. Hüsing, Tannin-based hybrid materials and their applications: a review. Molecules 25, 4910 (2020). https://doi.org/10.3390/molecules25214910
S.C.R. Santos, H.A.M. Bacelo, R.A.R. Boaventura, C.M.S. Botelho, Tannin-adsorbents for water decontamination and for the recovery of critical metals: current state and future perspectives. Biotechnol. J. 14, 1900060 (2019). https://doi.org/10.1002/biot.201900060
F.L. Braghiroli, V. Fierro, M.T. Izquierdo, J. Parmentier, A. Pizzi, A. Celzard, Nitrogen-doped carbon materials produced from hydrothermally treated tannin. Carbon N Y 50, 5411–5420 (2012). https://doi.org/10.1016/j.carbon.2012.07.027
K. Hashida, R. Makino, S. Ohara, Amination of pyrogallol nucleus of condensed tannins and related polyphenols by ammonia water treatment. Hfsg 63, 319–326 (2009). https://doi.org/10.1515/HF.2009.043
F. Braghiroli, V. Fierro, A. Pizzi, K. Rode, W. Radke, L. Delmotte, J. Parmentier, A. Celzard, Reaction of condensed tannins with ammonia. Ind. Crops Prod. 44, 330–335 (2013). https://doi.org/10.1016/j.indcrop.2012.11.024
C. Luo, W. Grigsby, N. Edmonds, A. Easteal, J. Al-Hakkak, Synthesis, characterization, and thermal behaviors of tannin stearates prepared from quebracho and pine bark extracts. J. Appl. Polym. Sci. (2010). https://doi.org/10.1002/app.31545
A. Nicollin, X. Zhou, A. Pizzi, W. Grigsby, K. Rode, L. Delmotte, MALDI-TOF and 13C NMR analysis of a renewable resource additive—thermoplastic acetylated tannins. Ind. Crops Prod. 49, 851–857 (2013). https://doi.org/10.1016/j.indcrop.2013.06.013
R. Soto, Evidence of chemical reactions between di- and poly-glycidyl ether resins and tannins isolated from Pinus radiata D. Don bark. Bioresour. Technol. 96, 95–101 (2005). https://doi.org/10.1016/j.biortech.2003.05.006
M. Gao, Z. Wang, C. Yang, J. Ning, Z. Zhou, G. Li, Novel magnetic graphene oxide decorated with persimmon tannins for efficient adsorption of malachite green from aqueous solutions. Colloids Surf A Physicochem Eng Asp 566, 48–57 (2019). https://doi.org/10.1016/j.colsurfa.2019.01.016
A.F.M. Santos, L.J.A. Macedo, M.H. Chaves, M. Espinoza-Castañeda, A. Merkoçi, F.C.A. Lima, W. Cantanhêde, Hybrid self-assembled materials constituted by ferromagnetic nanoparticles and tannic acid: a theoretical and experimental investigation. J. Braz. Chem. Soc. (2015). https://doi.org/10.5935/0103-5053.20150322
K. Rurack, R. Martínez-Máñez, Hybrid nano materials meet supramolecular chemistry: a brief introduction to basic terms and concepts, in The Supramolecular Chemistry of Organic-Inorganic Hybrid Materials. (Wiley, Hoboken, 2010), pp.1–10. https://doi.org/10.1002/9780470552704.ch1
S.I. Stupp, L.C. Palmer, Supramolecular chemistry and self-assembly in organic materials design. Chem. Mater. 26, 507–518 (2014). https://doi.org/10.1021/cm403028b
C.-C. Huang, H.-Y. Liao, Y.-C. Shiang, Z.-H. Lin, Z. Yang, H.-T. Chang, Synthesis of wavelength-tunable luminescent gold and gold/silver nanodots. J. Mater. Chem. 19, 755–759 (2009). https://doi.org/10.1039/B808594C
X. Huang, Y. Wang, X. Liao, B. Shi, Adsorptive recovery of Au3+ from aqueous solutions using bayberry tannin-immobilized mesoporous silica. J. Hazard. Mater. 183, 793–798 (2010). https://doi.org/10.1016/j.jhazmat.2010.07.096
L.S. Costa, G.P. Fidelis, S.L. Cordeiro, R.M. Oliveira, D.A. Sabry, R.B.G. Câmara, L.T.D.B. Nobre, M.S.S.P. Costa, J. Almeida-Lima, E.H.C. Farias, E.L. Leite, H.A.O. Rocha, Biological activities of sulfated polysaccharides from tropical seaweeds. Biomed. Pharmacother. 64, 21–28 (2010). https://doi.org/10.1016/j.biopha.2009.03.005
E. Binaeian, N. Seghatoleslami, M.J. Chaichi, Synthesis of oak gall tannin-immobilized hexagonal mesoporous silicate (OGT-HMS) as a new super adsorbent for the removal of anionic dye from aqueous solution. Desalin. Water Treat. 57, 8420–8436 (2016). https://doi.org/10.1080/19443994.2015.1020513
Q. Xu, Y. Wang, L. Jin, Y. Wang, M. Qin, Adsorption of Cu (II), Pb (II) and Cr (VI) from aqueous solutions using black wattle tannin-immobilized nanocellulose. J. Hazard. Mater. 339, 91–99 (2017). https://doi.org/10.1016/j.jhazmat.2017.06.005
Y. Zhang, F. He, X. Li, Three-dimensional composite hydrogel based on polyamine zirconium oxide, alginate and tannic acid with high performance for Pb(II), Hg(II) and Cr(VI) trapping. J. Taiwan Inst. Chem. Eng. 65, 304–311 (2016). https://doi.org/10.1016/j.jtice.2016.05.023
Y. Leng, L. Shi, S. Du, J. Jiang, P. Jiang, A tannin-derived zirconium-containing porous hybrid for efficient Meerwein–Ponndorf–Verley reduction under mild conditions. Green Chem. 22, 180–186 (2020). https://doi.org/10.1039/C9GC03393A
N.P. de Moraes, M.L.C.P. da Silva, T.M.B. Campos, G.P. Thim, L.A. Rodrigues, Novel synthetic route for low-cost carbon-modified TiO2 with enhanced visible light photocatalytic activity: carbon content and calcination effects. J. Solgel Sci. Technol. 87, 380–390 (2018). https://doi.org/10.1007/s10971-018-4700-4
A. Baldwin, B.W. Booth, Biomedical applications of tannic acid. J. Biomater. Appl. 36, 1503–1523 (2022). https://doi.org/10.1177/08853282211058099
E.D. Bartzoka, H. Lange, G. Poce, C. Crestini, Stimuli-responsive tannin–FeIII hybrid microcapsules demonstrated by the active release of an anti-tuberculosis agent. Chemsuschem 11, 3975–3991 (2018). https://doi.org/10.1002/cssc.201801546
P. Dharmalingam, G. Palani, R. Apsari, K. Kannan, S.K. Lakkaboyana, K. Venkateswarlu, V. Kumar, Y. Ali, Synthesis of metal oxides/sulfides-based nanocomposites and their environmental applications: a review. Mater. Today Sustain. 20, 100232 (2022). https://doi.org/10.1016/j.mtsust.2022.100232
M.B. Tahir, M. Rafique, M.S. Rafique, N. Fatima, Z. Israr, Metal oxide- and metal sulfide-based nanomaterials as photocatalysts, in Nanotechnology and Photocatalysis for Environmental Applications. (Elsevier, 2020), pp.77–96. https://doi.org/10.1016/B978-0-12-821192-2.00006-1
G.B. Subbaiah, K.V. Ratnam, S. Janardhan, K. Shiprath, H. Manjunatha, M. Ramesha, N.V.K. Prasad, S. Ramesh, T.A. Babu, Metal and metal oxide based advanced ceramics for electrochemical biosensors: a short review. Front Mater (2021). https://doi.org/10.3389/fmats.2021.682025
W. Guan, N. Tang, K. He, X. Hu, M. Li, K. Li, Gas-sensing performances of metal oxide nanostructures for detecting dissolved gases: a mini review. Front. Chem. (2020). https://doi.org/10.3389/fchem.2020.00076
Y.D. Tretyakov, E.A. Goodilin, Chemical design of metal-oxide superconductors. Phys. B: Condens. Matter 321(14), 249–256 (2002)
P. Kurzweil, Precious metal oxides for electrochemical energy converters: Pseudocapacitance and pH dependence of redox processes. J. Power. Sources 190, 189–200 (2009). https://doi.org/10.1016/j.jpowsour.2008.08.033
J.C. Védrine, Heterogeneous catalysis on metal oxides. Catalysts (2017). https://doi.org/10.3390/catal7110341
P. Rani, V. Kumar, P.P. Singh, A.S. Matharu, W. Zhang, K.-H. Kim, J. Singh, M. Rawat, Highly stable AgNPs prepared via a novel green approach for catalytic and photocatalytic removal of biological and non-biological pollutants. Environ. Int. 143, 105924 (2020). https://doi.org/10.1016/j.envint.2020.105924
J. Singh, V. Kumar, S. Singh Jolly, K.-H. Kim, M. Rawat, D. Kukkar, Y.F. Tsang, Biogenic synthesis of silver nanoparticles and its photocatalytic applications for removal of organic pollutants in water. J. Ind. Eng. Chem. 80, 247–257 (2019). https://doi.org/10.1016/j.jiec.2019.08.002
Y. Wu, C. Wadia, W. Ma, B. Sadtler, A.P. Alivisatos, Synthesis and photovoltaic application of Copper(I) sulfide nanocrystals. Nano Lett. 8, 2551–2555 (2008). https://doi.org/10.1021/nl801817d
T.-L. Li, Y.-L. Lee, H. Teng, CuInS2 quantum dots coated with CdS as high-performance sensitizers for TiO2 electrodes in photoelectrochemical cells. J. Mater. Chem. 21, 5089 (2011). https://doi.org/10.1039/c0jm04276e
M.J. Bierman, S. Jin, Potential applications of hierarchical branching nanowires in solar energy conversion. Energy Environ. Sci. 2, 1050 (2009). https://doi.org/10.1039/b912095e
H.-R. Kim, A. Haensch, I.-D. Kim, N. Barsan, U. Weimar, J.-H. Lee, The role of NiO doping in reducing the impact of humidity on the performance of SnO2-based gas sensors: synthesis strategies, and phenomenological and spectroscopic studies. Adv. Funct. Mater. 21, 4456–4463 (2011). https://doi.org/10.1002/adfm.201101154
N. Yamazoe, Toward innovations of gas sensor technology. Sens Actuators B Chem 108, 2–14 (2005). https://doi.org/10.1016/j.snb.2004.12.075
N. Barsan, D. Koziej, U. Weimar, Metal oxide-based gas sensor research: how to? Sens Actuators B Chem 121, 18–35 (2007). https://doi.org/10.1016/j.snb.2006.09.047
A. Umar, Y.-B. Hahn, Metal Oxide Nanostructures and Their Applications (American Scientific Publishers, Los Angeles, 2010)
V. Guidi, B. Fabbri, A. Gaiardo, S. Gherardi, A. Giberti, C. Malagù, G. Zonta, P. Bellutti, Metal sulfides as a new class of sensing materials. Procedia Eng. 120, 138–141 (2015). https://doi.org/10.1016/j.proeng.2015.08.586
S. Laurichesse, L. Avérous, Chemical modification of lignins: towards biobased polymers. Prog. Polym. Sci. 39, 1266–1290 (2014). https://doi.org/10.1016/j.progpolymsci.2013.11.004
A. Duval, M. Lawoko, A review on lignin-based polymeric, micro- and nano-structured materials. React. Funct. Polym. 85, 78–96 (2014). https://doi.org/10.1016/j.reactfunctpolym.2014.09.017
C. Wang, S.S. Kelley, R.A. Venditti, Lignin-based thermoplastic materials. Chemsuschem 9, 770–783 (2016). https://doi.org/10.1002/cssc.201501531
P. Figueiredo, K. Lintinen, J.T. Hirvonen, M.A. Kostiainen, H.A. Santos, Properties and chemical modifications of lignin: towards lignin-based nanomaterials for biomedical applications. Prog. Mater. Sci. 93, 233–269 (2018). https://doi.org/10.1016/j.pmatsci.2017.12.001
M.S. Ganewatta, H.N. Lokupitiya, C. Tang, Lignin biopolymers in the age of controlled polymerization. Polymers (Basel) 11, 1176 (2019). https://doi.org/10.3390/polym11071176
T.M. Budnyak, A. Slabon, M.H. Sipponen, Lignin-inorganic interfaces: chemistry and applications from adsorbents to catalysts and energy storage materials. Chemsuschem 13, 4344–4355 (2020). https://doi.org/10.1002/cssc.202000216
M.N. Collins, M. Nechifor, F. Tanasă, M. Zănoagă, A. McLoughlin, M.A. Stróżyk, M. Culebras, C.-A. Teacă, Valorization of lignin in polymer and composite systems for advanced engineering applications: a review. Int. J. Biol. Macromol. 131, 828–849 (2019). https://doi.org/10.1016/j.ijbiomac.2019.03.069
W.-J. Liu, H. Jiang, H.-Q. Yu, Thermochemical conversion of lignin to functional materials: a review and future directions. Green Chem. 17, 4888–4907 (2015). https://doi.org/10.1039/C5GC01054C
K.-T. Chung, T.Y. Wong, C.-I. Wei, Y.-W. Huang, Y. Lin, Tannins and human health: a review. Crit. Rev. Food Sci. Nutr. 38, 421–464 (1998). https://doi.org/10.1080/10408699891274273
R. Armitage, G.S. Bayliss, J.W. Gramshaw, E. Haslam, R.D. Haworth, K. Jones, H.J. Rogers, T. Searle, 360. Gallotannins. Part III. The constitution of Chinese, Turkish, Sumach, and tara tannins. J. Chem. Soc. (Resumed) (1961). https://doi.org/10.1039/jr9610001842
G. Britton, P.W. Crabtree, E. Haslam, J.E. Stangroom, Gallotannins. Part XIII. The structure of Chinese gallotannin: evidence for a polygalloyl chain. J. Chem. Soc. C: Org. (1966). https://doi.org/10.1039/j39660000783
Z. Guo, W. Xie, J. Lu, X. Guo, J. Xu, W. Xu, Y. Chi, N. Takuya, H. Wu, L. Zhao, Tannic acid-based metal phenolic networks for bio-applications: a review. J. Mater. Chem. B 9, 4098–4110 (2021). https://doi.org/10.1039/D1TB00383F
J.H. Park, S. Choi, H.C. Moon, H. Seo, J.Y. Kim, S.-P. Hong, B.S. Lee, E. Kang, J. Lee, D.H. Ryu, I.S. Choi, Antimicrobial spray nanocoating of supramolecular Fe(III)-tannic acid metal-organic coordination complex: applications to shoe insoles and fruits. Sci. Rep. 7, 6980 (2017). https://doi.org/10.1038/s41598-017-07257-x
H. Ejima, J.J. Richardson, K. Liang, J.P. Best, M.P. van Koeverden, G.K. Such, J. Cui, F. Caruso, One-step assembly of coordination complexes for versatile film and particle engineering. Science 341(2013), 154–157 (1979). https://doi.org/10.1126/science.1237265
X. Wang, W. Cai, D. Ye, Y. Zhu, M. Cui, J. Xi, J. Liu, W. Xing, Bio-based polyphenol tannic acid as universal linker between metal oxide nanoparticles and thermoplastic polyurethane to enhance flame retardancy and mechanical properties. Compos. B Eng. 224, 109206 (2021). https://doi.org/10.1016/j.compositesb.2021.109206
N.A. Che Lah, P. Murthy, M.N. Mohd Zubir, The physical and optical investigations of the tannic acid functionalised Cu-based oxide nanostructures. Sci. Rep. 12, 9909 (2022). https://doi.org/10.1038/s41598-022-14281-z
V. Kozlovskaya, E. Kharlampieva, I. Drachuk, D. Cheng, V.V. Tsukruk, Responsive microcapsule reactors based on hydrogen-bonded tannic acid layer-by-layer assemblies. Soft Matter 6, 3596 (2010). https://doi.org/10.1039/b927369g
L. Zou, P. Shao, K. Zhang, L. Yang, D. You, H. Shi, S.G. Pavlostathis, W. Lai, D. Liang, X. Luo, Tannic acid-based adsorbent with superior selectivity for lead(II) capture: adsorption site and selective mechanism. Chem. Eng. J. 364, 160–166 (2019). https://doi.org/10.1016/j.cej.2019.01.160
Z. Fu, R. Chen, Study of complexes of tannic acid with Fe(III) and Fe(II). J. Anal. Methods Chem. 2019, 1–6 (2019). https://doi.org/10.1155/2019/3894571
J. Guo, Y. Ping, H. Ejima, K. Alt, M. Meissner, J.J. Richardson, Y. Yan, K. Peter, D. von Elverfeldt, C.E. Hagemeyer, F. Caruso, Engineering multifunctional capsules through the assembly of metal-phenolic networks. Angew. Chem. Int. Ed. 53, 5546–5551 (2014). https://doi.org/10.1002/anie.201311136
S. Kalidas Sivaraman, I. Elango, S. Kumar, V. Santhanam, A green protocol for room temperature synthesis of silver nanoparticles in seconds. Curr. Sci. 97(7), 1055–1059 (2009)
X. Tian, W. Wang, G. Cao, A facile aqueous-phase route for the synthesis of silver nanoplates. Mater. Lett. 61, 130–133 (2007). https://doi.org/10.1016/j.matlet.2006.04.021
S. Aswathy Aromal, D. Philip, Facile one-pot synthesis of gold nanoparticles using tannic acid and its application in catalysis. Phys. E Low Dimens. Syst. Nanostruct. 44, 1692–1696 (2012). https://doi.org/10.1016/j.physe.2012.04.022
İ Gülçin, Z. Huyut, M. Elmastaş, H.Y. Aboul-Enein, Radical scavenging and antioxidant activity of tannic acid. Arab. J. Chem. 3, 43–53 (2010). https://doi.org/10.1016/j.arabjc.2009.12.008
J.G. Manjunatha, B.E.K. Swamy, G.P. Mamatha, O. Gilbert, B.N. Chandrashekar, B.S. Sherigara, Electrochemical studies of dopamine and epinephrine at a poly (tannic acid) modified carbon paste electrode: a cyclic voltammetric study. Int. J. Electrochem. Sci. 5, 1236–1245 (2010). https://doi.org/10.1016/S1452-3981(23)15358-2
B. Çakıroğlu, M. Özacar, Tannic acid modified electrochemical biosensor for glucose sensing based on direct electrochemistry. Electroanalysis 29, 2719–2726 (2017). https://doi.org/10.1002/elan.201700420
A.L. Suherman, S. Kuss, E.E.L. Tanner, N.P. Young, R.G. Compton, Electrochemical Hg2+ detection at tannic acid-gold nanoparticle modified electrodes by square wave voltammetry. Analyst 143, 2035–2041 (2018). https://doi.org/10.1039/C8AN00508G
M. Mehmandoust, S. Çakar, M. Özacar, N. Erk, The determination of timolol maleate using silver/tannic acid/titanium oxide nanocomposite as an electrochemical sensor in real samples. Electroanalysis 34, 1150–1162 (2022). https://doi.org/10.1002/elan.202100363
A.L. Suherman, E.E.L. Tanner, S. Kuss, S.V. Sokolov, J. Holter, N.P. Young, R.G. Compton, Voltammetric determination of aluminium(III) at tannic acid capped-gold nanoparticle modified electrodes. Sens. Actuators B Chem. 265, 682–690 (2018). https://doi.org/10.1016/j.snb.2018.03.098
M. Wang, F. Pan, L. Yang, Y. Song, H. Wu, X. Cheng, G. Liu, H. Yang, H. Wang, Z. Jiang, X. Cao, Graphene oxide quantum dots incorporated nanocomposite membranes with high water flux for pervaporative dehydration. J Memb Sci 563, 903–913 (2018). https://doi.org/10.1016/j.memsci.2018.06.062
S. Li, S. Du, S. Liu, B. Su, L. Han, Ultra-smooth and ultra-thin polyamide thin film nanocomposite membranes incorporated with functionalized MoS2 nanosheets for high performance organic solvent nanofiltration. Sep. Purif. Technol. 291, 120937 (2022). https://doi.org/10.1016/j.seppur.2022.120937
C. Zhang, D.-F. Hu, J.-W. Xu, M.-Q. Ma, H. Xing, K. Yao, J. Ji, Z.-K. Xu, Polyphenol-assisted exfoliation of transition metal dichalcogenides into nanosheets as photothermal nanocarriers for enhanced antibiofilm activity. ACS Nano 12, 12347–12356 (2018). https://doi.org/10.1021/acsnano.8b06321
Q. Wei, R. Haag, Universal polymer coatings and their representative biomedical applications. Mater. Horiz. 2, 567–577 (2015). https://doi.org/10.1039/C5MH00089K
Y. Tian, K.-H. Wu, X. Tan, Q. Zeng, R. Amal, D.-W. Wang, Hydrophilic tannic acid-modified WS 2 nanosheets for enhanced polysulfide conversion in aqueous media. J. Phys.: Energy 1, 015005 (2018). https://doi.org/10.1088/2515-7655/aaead4
A. Jędrzak, T. Rębiś, M. Kuznowicz, T. Jesionowski, Bio-inspired magnetite/lignin/polydopamine-glucose oxidase biosensing nanoplatform. From synthesis, via sensing assays to comparison with others glucose testing techniques. Int. J. Biol. Macromol. 127, 677–682 (2019). https://doi.org/10.1016/j.ijbiomac.2019.02.008
M. Martín, P. Salazar, C. Jiménez, M. Lecuona, M.J. Ramos, J. Ode, J. Alcoba, R. Roche, R. Villalonga, S. Campuzano, J.M. Pingarrón, J.L. González-Mora, Rapid Legionella pneumophila determination based on a disposable core–shell Fe3O4 @poly(dopamine) magnetic nanoparticles immunoplatform. Anal. Chim. Acta 887, 51–58 (2015). https://doi.org/10.1016/j.aca.2015.05.048
A. Jędrzak, T. Rębiś, M. Kuznowicz, A. Kołodziejczak-Radzimska, J. Zdarta, A. Piasecki, T. Jesionowski, Advanced Ga2O3/lignin and ZrO2/lignin hybrid microplatforms for glucose oxidase immobilization: evaluation of biosensing properties by catalytic glucose oxidation. Catalysts 9, 1044 (2019). https://doi.org/10.3390/catal9121044
G.A. El-Fatah, H.S. Magar, R.Y.A. Hassan, R. Mahmoud, A.A. Farghali, M.E.M. Hassouna, A novel gallium oxide nanoparticles-based sensor for the simultaneous electrochemical detection of Pb2+, Cd2+ and Hg2+ ions in real water samples. Sci. Rep. (2022). https://doi.org/10.1038/s41598-022-24558-y
A. Puangjan, S. Chaiyasith, An efficient ZrO2/Co3O4/reduced graphene oxide nanocomposite electrochemical sensor for simultaneous determination of gallic acid, caffeic acid and protocatechuic acid natural antioxidants. Electrochim. Acta (2016). https://doi.org/10.1016/j.electacta.2016.04.185
P. Bansal, G. Bhanjana, N. Prabhakar, J.S. Dhau, G.R. Chaudhary, Electrochemical sensor based on ZrO2 NPs/Au electrode sensing layer for monitoring hydrazine and catechol in real water samples. J. Mol. Liq. (2017). https://doi.org/10.1016/j.molliq.2017.10.098
R. Chokkareddy, G.G. Redhi, T. Karthick, A lignin polymer nanocomposite based electrochemical sensor for the sensitive detection of chlorogenic acid in coffee samples. Heliyon 5, 1457 (2019). https://doi.org/10.1016/j.heliyon.2019
M.R. Olthof, P.C.H. Hollman, M.B. Katan, Chlorogenic acid and caffeic acid are absorbed in humans. J. Nutr. (2001). https://doi.org/10.1093/jn/131.1.66
M.R. Olthof, P.C.H. Hollman, M.B. Katan, Human nutrition and metabolism chlorogenic acid and caffeic acid are absorbed in humans. J. Nutr. 131(1), 66–71 (2001)
R. Chokkareddy, G.G. Redhi, T. Karthick, A lignin polymer nanocomposite based electrochemical sensor for the sensitive detection of chlorogenic acid in coffee samples. Heliyon 5, e01457 (2019). https://doi.org/10.1016/j.heliyon.2019.e01457
L. Wang, X. Pan, L. Jiang, Y. Chu, S. Gao, X. Jiang, Y. Zhang, Y. Chen, S. Luo, C. Peng, The biological activity mechanism of chlorogenic acid and its applications in food industry: a review. Front. Nutr. (2022). https://doi.org/10.3389/fnut.2022.943911
S. Antherjanam, B. Saraswathyamma, Simultaneous electrochemical determination of hydrazine and hydroxylamine on a thiadiazole derivative modified pencil graphite electrode. Mater. Chem. Phys. 275, 125223 (2021). https://doi.org/10.1016/j.matchemphys.2021.125223
K.A. Mahmoud, A. Abdel-Wahab, M. Zourob, Selective electrochemical detection of 2,4,6-trinitrotoluene (TNT) in water based on poly(styreneco-acrylic acid) PSA/SiO2/Fe3O4/AuNPs/lignin-modified glassy carbon electrode. Water Sci. Technol. (2015). https://doi.org/10.2166/wst.2015.399
L. Zhang, Y. Han, J. Zhu, Y. Zhai, S. Dong, Simple and sensitive fluorescent and electrochemical trinitrotoluene sensors based on aqueous carbon dots. Anal. Chem. (2015). https://doi.org/10.1021/ac5043686
Y. Wei, M. Song, L. Yu, X. Tang, Preparation of ZnO-loaded lignin-based carbon fiber for the electrocatalytic oxidation of hydroquinone. Catalysts (2017). https://doi.org/10.3390/catal7060180
K. Ahmad, P. Kumar, S.M. Mobin, A highly sensitive and selective hydroquinone sensor based on a newly designed N-rGO/SrZrO3 composite. Nanoscale Adv. (2020). https://doi.org/10.1039/c9na00573k
A. Karthika, V. Ramasamy Raja, P. Karuppasamy, A. Suganthi, M. Rajarajan, A novel electrochemical sensor for determination of hydroquinone in water using FeWO4/SnO2 nanocomposite immobilized modified glassy carbon electrode. Arab. J. Chem. (2020). https://doi.org/10.1016/j.arabjc.2019.06.008
Y. Zhang, F. Wen, Z. Huang, J. Tan, Z. Zhou, K. Yuan, H. Wang, Nitrogen doped lignocellulose/binary metal sulfide modified electrode: preparation and application for non-enzymatic ascorbic acid, dopamine and nitrite sensing. J. Electroanal. Chem. 806, 150–157 (2017). https://doi.org/10.1016/j.jelechem.2017.10.066
F. Ghaemi, L.C. Abdullah, H. Ariffin, Lignocellulose structure and the effect on nanocellulose production, in Lignocellulose for Future Bioeconomy. (Elsevier, NY, 2019), pp.17–30
M. Vasudevan, V. Perumal, S. Karuppanan, M. Ovinis, P. Bothi Raja, S.C.B. Gopinath, T.N.J. Immanuel Edison, A comprehensive review on biopolymer mediated nanomaterial composites and their applications in electrochemical sensors. Crit. Rev. Anal. Chem. (2022). https://doi.org/10.1080/10408347.2022.2135090
Acknowledgements
The authors acknowledge Amrita Vishwa Vidyapeetham, Amritapuri campus for the internal support.
Author information
Authors and Affiliations
Contributions
Data collection was done by Devu C, Sreelekshmi S, Chandana R, Sivanand P and Santhy A. Editing and reviewing were done by Seetha Lakshmi K C and Rejithamol R.
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Devu, C., Sreelakshmi, S., Chandana, R. et al. Recent progress in tannin and lignin blended metal oxides and metal sulfides as smart materials for electrochemical sensor applications. ANAL. SCI. 40, 981–996 (2024). https://doi.org/10.1007/s44211-024-00544-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44211-024-00544-4