Abstract
Background
Depression is a complex mental health disorder marked by persistent sadness, hopelessness, and reduced interest in daily activities. The present study developed the Afghanistan National Depression Screening (ANDs) scale and examined its psychometric properties within the Afghan population.
Methods
The study enrolled 1245 Afghan individuals as participants. The items within the ANDs scale were developed through a thorough examination of pre-existing depression scales, expert assessments, and participant interviews. Multiple psychometric evaluations were administered to ensure the reliability and validity of the scale.
Results
After review and corrected item-total correlation testing, 15-items exhibiting acceptable corrected item-total correlation coefficients (ranging from 0.315 to 0.647) were retained and further validated through significant factor loadings (ranging from 0.358 to 0.725). Additionally, various properties assessed through classical test theory demonstrated satisfactory results on the 15-item scale. Specifically, measures of reliability such as internal consistency (α = 0.846) and Spearman-Brown coefficient (0.975) were deemed acceptable. Convergent validity was evidenced by strong correlations with established scales, including the DASS-21 (depression subscale, r = 0.854), GHQ-28 (depression subscale, r = 0.693), CES-D 20 (r = 0.922), and PHQ-9 (r = 0.758).
Conclusion
The 15-item ANDs scale is developed to assess depression in Afghan populations, showing promise in correlation with established measures like PHQ-9, DASS-21, CESD-20, and GHQ-28. Its potential for identifying depression symptoms warrants further investigation in epidemiological studies and clinical settings, though replication across diverse samples is necessary for generalizability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Depression constitutes a multifaceted and debilitating mental health disorder characterized by persistent and pervasive dysphoric mood states, including profound sadness, feelings of hopelessness, and a notable diminishment in the capacity to experience pleasure or interest in routine activities [1]. According to World Health Organization (WHO), approximately 5% of adults (4% men and 6% women) in the world are suffering from depression [2]. Beyond the realm of ordinary mood fluctuations, depression engenders a pervasive impact on cognitive, affective, and behavioral domains, thereby significantly impairing daily functioning [3]. Commonly observed symptoms encompass disruptions in sleep patterns, alterations in appetite, decreased energy levels, cognitive impairments such as difficulties in concentration, and a proclivity towards indecisiveness [4]. The etiology of depression is inherently complex, implicating a confluence of genetic, neurobiological, environmental, and psychosocial determinants [5]. In the realm of academic discourse, depression manifests not only through cognitive and emotional dimensions but also encompasses somatic aspects, frequently leading individuals to exhibit concurrent physical symptoms [6, 7]. Given the considerable variation in symptomatology and the deleterious ramifications on overall well-being, a holistic strategy for addressing depression involves utilizing both medication and therapy, highlighting the importance of expert evaluation and support to achieve the best treatment results [8, 9].
Existing depression scales encompass a variety of instruments designed to assess and quantify depressive symptoms across different populations. The Beck Depression Inventory (BDI) [10] and Patient Health Questionnaire (PHQ-9) [11] offer self-report measures, while the Hamilton Depression Rating Scale (HAM-D) [12] provides a comprehensive questionnaire for symptom evaluation. For elderly individuals, the Geriatric Depression Scale (GDS) [13] is valuable, and the Zung Self-Rating Depression Scale (SDS) [14] offers a self-administered option. The Center for Epidemiologic Studies Depression Scale (CES-D) [15] targets the general population, while the Montgomery-Åsberg Depression Rating Scale (MADRS) [16] aids in severity assessment. Additionally, the Quick Inventory of Depressive Symptomatology (QIDS) [17] and the 21-item Depression Anxiety Stress Scales (DASS-21) [18] offer a broader assessment of emotional states. These instruments collectively enhance the multidimensional understanding and diagnosis of depression, facilitating effective interventions and support strategies for clinicians and researchers alike.
The absence of culturally sensitive and contextually appropriate screening tools contributes to the under-recognition of depression in Afghanistan. Existing depression assessment instruments, often developed in Western contexts, may not adequately capture the unique manifestations, cultural expressions, and determinants of depression in Afghan populations. Moreover, the stigma associated with mental illness further impedes help-seeking behaviors and exacerbates the treatment gap.
To address these challenges, there is a pressing need for a validated depression screening instrument tailored specifically to the Afghan context [19]. The development of such a tool requires careful consideration of cultural nuances, linguistic diversity, and socio-economic factors prevalent in Afghanistan [19]. By creating a culturally sensitive screening scale, mental health professionals can enhance the early detection of depression, facilitate timely interventions, and ultimately alleviate the burden of this debilitating condition in Afghan society [19].
Refining existing methodologies, our research introduces the Afghanistan National Depression Screening (ANDs) Scale, meticulously crafted to align with Afghanistan’s distinct cultural and linguistic landscape. This innovation transcends the limitations of generic depression assessments, ensuring precise detection of culturally specific manifestations of distress. The ANDs Scale not only advances contextually nuanced mental health diagnostics but also pioneers a paradigm shift towards the development of universally adaptable, culturally sensitive assessment tools. The development of the ANDs scale involved rigorous processes of adaptation, translation, validation, and psychometric evaluation, guided by established principles of cross-cultural assessment and collaborative research with local stakeholders.
2 Materials and methods
2.1 Methods
2.1.1 Procedure
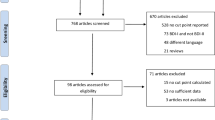
A team of clinicians and researchers conducted a thorough review of existing scales for depression. The final selection for developing the ANDs scale comprised four scales: DASS-21, CES-D 20, GHQ-28, and PHQ-9. The resulting questionnaire, with a total of 85 items, was carefully finalized. Based on a comprehensive literature review and expert advice, the major components of depressive symptomatology were identified. These components include depressed mood, feelings of helplessness, worthlessness, hopelessness, and psychomotor retardation. To capture the current state, respondents were asked, “During the past two weeks, how often have you…”. Responses were then evaluated on a scale ranging from zero (never) to three (almost always), reflecting the frequency of symptom occurrence. Initial tests on small convenience samples demonstrated the scale's performance and guided revisions to enhance clarity and acceptability. The final scale used in this study, consisting of 15 items, is detailed in Table 2. Scores on this scale can range from zero to 45, with higher scores indicating a greater prevalence of symptoms. The weighting of scores takes into account the frequency of symptom occurrence over the preceding two weeks.
After curating items deemed suitable for inclusion in the questionnaire, it was subsequently implemented in a self-rating format among a cohort of 50 participants. The sampled individuals included both ordinary community residents without reported depressive symptoms or a history of mental disorders and individuals under hospitalization for depression. The inclusion criteria encompassed both male and female participants sourced from various parts of Herat city in Afghanistan. It is noteworthy that all participants in the pilot study were aged 18 years or older. The results of the pilot study indicated that no changes were needed.
Comprehensive investigation was conducted to validate measurement instruments, involving a convenience sample of 1245 adult participants from diverse regions of Afghanistan. This study’s data collection transpired between June and September 2023, incorporating meticulous efforts to ensure linguistic and cultural appropriateness. a total of 75 questionnaires were collected for each item in the ANDs scale. To enhance the robustness of the data and account for potential incomplete responses, an additional 10% of questionnaires were also collected, resulting in a final sample size of 1245 participants. This approach aimed to ensure a sufficiently large and representative dataset for the psychometric testing and validation of the ANDs scale.
Skilled data collectors engaged in face-to-face interactions following rigorous training that emphasized the standardized administration of the ANDs scale. The training program addressed study objectives, questionnaire nuances, and ethical considerations, fostering uniformity in data collection procedures. Regular debriefing sessions facilitated ongoing feedback and refinement of methods. Consistency was maintained through standardized scripts, minimizing variations in question presentation. To mitigate biases, data collectors adopted a neutral and non-judgmental approach. Overall, this comprehensive training, coupled with standardized procedures and ongoing support, ensured the reliability and validity of collected data while minimizing potential sources of bias in participant responses.
In order to participate in the study, individuals were required to meet specific criteria, including providing informed consent, being 18 years old or older, and demonstrating the ability to read and comprehend the Dari language. These prerequisites were established to ensure the ethical inclusion of participants who could actively engage in the self-administered questionnaire, fostering a comprehensive and meaningful assessment of depressive symptomatology within the targeted population.
2.1.2 Analysis
Data extraction from the questionnaire was carried out, with information meticulously organized in Excel format for Windows. Statistical analyses were conducted using IBM SPSS Statistics software (version 26) for Windows. Internal Consistency Reliability was assessed using Cronbach’s Alpha, with values exceeding 0.70 considered satisfactory. Pearson correlation analyses were employed to assess convergent validity, and criterion validity, comparing results with the DASS-21, GHQ-28, CES-D 20, and PHQ-9. Test–retest reliability was evaluated using the Interclass Correlation Coefficient, and a value of 0.70 or above was deemed satisfactory. Exploratory factor analysis was performed using the principal component analysis with oblimin rotation (Kaiser normalization) for the factor structure. The Kaiser–Meyer–Olkin (KMO) statistic and Bartlett’s test of sphericity were carried out to check for sampling suitability and factor analysis. Factor loading greater than 0.30 was considered statistically meaningful.
The validation process extended to confirmatory factor analysis, gauging the goodness-of-fit of the four construct models of DASS-21 within the Afghan population. Various parameters, including minimum discrepancy over degree of freedom (CMIN/df), root mean square error of approximation (RMSEA), standardized root mean residual (SRMR), goodness-of-fit index (GFI), adjusted goodness-of-fit index (AGFI), comparative fit index (CFI), and Tucker-Lewis index (TLI), were utilized for this assessment, with adherence to established criteria such as CMIN/df < 5; Standardized RMR < 0.05; RMSEA < 0.08; GFI > 0.90; AGFI > 0.90; CFI > 0.95; and TLI > 0.95.
2.1.3 Ethical approval
The present investigation obtained ethical clearance from the Ethical Committee of the Afghanistan Center for Epidemiological Studies, as denoted by reference number #21.1.041.
2.2 Measures
2.2.1 Socio-demographics
A demographic information sheet, encompassing details such as age, gender, marital status, residency, economic standing, and recent exposure to a traumatic event within the past month, was employed to gather background information about the participants. The assessment of participants’ levels of depression was conducted using the Persian version of the following scales:
2.2.2 DASS-21
The Dari version of the DASS-21 [18] is a 21-item instrument used to assess levels of depression, anxiety, and stress. Responses are given on a four-point scale, with subscale scores calculated independently. Higher scores indicate greater severity in depression, anxiety, or stress. The DASS-21 has shown acceptable reliability, with Cronbach's α coefficients indicating satisfactory validity: 0.79 for anxiety, 0.91 for stress, and 0.93 for depression [20].
2.2.3 GHQ-28
The Persian version of the GHQ-28 [21] is a self-report tool designed to evaluate psychological well-being and identify potential mental health issues [21]. With 28 items, participants rate their symptoms on a four-point scale, generating scores ranging from 0 to 21 for each of the four subscales: somatic symptoms, anxiety and insomnia, social dysfunction, and severe depression [22].
2.2.4 CES-D-20
The CES-D 20 is a self-report questionnaire used to gauge the presence and intensity of depressive symptoms [15]. It's a condensed version of the original CES-D, featuring 20 items with responses on a four-point scale. Scores range from 0 to 60, with higher scores indicating more severe symptoms. Covering a range of depressive symptoms, it offers a thorough assessment for clinical and research use. Translations, like the Dari version, extend its utility across diverse linguistic and cultural settings [23].
2.2.5 PHQ-9
The Persian version of the PHQ-9 [24] is a concise self-report tool crafted to evaluate the presence and intensity of depressive symptoms [11]. With nine items, participants rate symptom frequency over the past two weeks on a four-point scale. Scores range from 0 to 27, with higher scores indicating greater depressive symptom severity.
3 Results
3.1 Characteristics
This study involved 1,245 participants, with a mean age of 32.49 years (SD ± 13.24). A majority of the participants were female (n = 625; 50.2%), and a substantial proportion were married (n = 742; 59.6%). Less than half of the participants hailed from rural areas, constituting 512 individuals (41.1%) (Table 1).
Table 2 displays the frequency of item responses, along with Classical Test Theory (CTT) statistics. Respondents endorsed all four response categories. Notably, a substantial proportion (25.9%) acknowledged occasional experiences of being unable to become enthusiastic about anything. Approximately half of the participants (45.8%) reported no instances of thinking their life had been a failure in the past week. Similarly, about half of the respondents (45.1%) indicated an absence of feelings of loneliness in the past week. The overall internal consistency (reliability) of the 15-item scale, as measured by Cronbach’s alpha, was 0.846, signifying excellent reliability (Table 2).
The Interclass Correlation Coefficient (ICC) for the ANDs scale was found to be 0.178 (95% CI 0.150–0.214), indicating a moderate level of reliability. The F Test [F(1, 223) = 7.517, p < 0.001] further supports the reliability of the measurements. The Spearman-Brown Coefficient, a measure of internal consistency, was calculated to be 0.975, suggesting high reliability between the two rounds of assessment (Table 3).
As shown in Table 4, significant positive correlations were observed between the ANDs scale and related measures, providing evidence for both convergent and criterion validity. Notably, strong correlations were found with DASS-21-Depression (r = 0.854, p < 0.001), GHQ-28-Depression (r = 0.693, p < 0.001), CES-D-20 (r = 0.922, p < 0.001), and PHQ-9 (r = 0.758, p < 0.001). These findings support the construct validity of the ANDs scale in relation to measures of depression (Table 4).
The exploratory factor analysis revealed the factor loadings and communalities (h2) for each item. Factor loadings represent the strength and direction of the relationship between items and factors, while communalities indicate the proportion of variance in each item explained by the factors. Notably, item 12 demonstrated a high factor loading of 0.725, suggesting a strong association with the underlying factor. The overall pattern of factor loadings supports the construct validity of the scale, indicating that the items align well with the underlying factors (Table 5).
In the examination of the ANDs scale's dimensionality through confirmatory factor analysis, the fit indices revealed a varied fit. Specifically, CFI was 0.819, TLI was 0.789, AGFI was 0.868, RMSEA was 0.087, NFI was 0.804, GFI was 0.901, and SRMR was 0.081. While certain indices fell below conventional thresholds for excellent fit, such as CFI, TLI, AGFI, NFI, and GFI, the RMSEA and SRMR values suggest a reasonable fit. The chi-square test (χ2) was statistically significant (p < 0.001) (Table 6) (Fig. 1).
4 Discussion
The current investigation introduced the development of the Afghanistan National Depression Screening (ANDs) Scale. Results revealed a consistent unidimensional framework and robust psychometric characteristics of the ANDs scale. Initial psychometric evaluations indicated favorable properties across various testing methodologies, including Classical Test Theory and Item Response Theory. Additionally, the cumulative score of the scale's items reliably reflects the severity of depression, as evidenced by its positive correlations with scores on the DASS-21 depression subscale, CES-D 20, GHQ-28 Depression subscale, and PHQ-9. Hence, higher scores on the ANDs scale correspond to more pronounced depressive symptoms, suggesting its utility in assessing and addressing psychological concerns among both genders.
Various research studies have elucidated the nuanced impact of psychological responses on the health and overall well-being of individuals, highlighting the influence of diverse contextual and cultural factors [25,26,27,28]. The assessment of convergent validity, employing established measures such as the DASS-21 depression subscale, CES-D 20, GHQ-28 depression subscale, and PHQ-9, underscores the validity of the ANDs scale as a reliable instrument for gauging both depression symptoms and the broader construct of depression.
The results of the item analysis indicated that the items on the ANDs Scale exhibited robust discrimination indices, as evidenced by the corrected item-total correlation coefficients. Such indices provide empirical support for the scale's effectiveness in differentiating between individuals who score high and those who score low. This capability is crucial for ensuring that the scale accurately reflects varying degrees of the attribute it measures. Comparative analysis with other research on depression scales and questionnaires further corroborates the strength of item discrimination in assessment tools employed in the development of the ANDs Scale [29,30,31,32]. These findings collectively underscore the precision and reliability of the ANDs Scale in assessing nuances within individual responses, thus enhancing its utility in both clinical and research settings.
The findings of the current investigation revealed that the ANDs Scale is characterized by a unidimensional factor structure. This aligns with the configurations documented in several established depression assessment instruments, including the CES-D 20 [15], the BDI [10], the PHQ-9 [11], the HRSD [12], and the MADRS [16]. Furthermore, the analysis indicated that each item on the ANDs Scale demonstrated robust factor loadings, a finding that echoes the structural integrity observed in the QIDS [17]. These consistencies underline the methodological soundness of the ANDs Scale in assessing symptoms of depression, thereby reinforcing its utility in both clinical and research contexts.
The ANDs Scale exhibited an acceptable test–retest reliability, as evidenced by its robust psychometric properties. This was evidenced by comparison with other established measures such as DASS-21 by Osman et al. [33], the CES-D 20 by Ohno et al. [34], GHQ-28 by Roty et al. [35], and PHQ-9 by Son et al. [36].
The ANDs Scale demonstrated a high degree of correlation with several well-established measures, including CES-D 20, GHQ-28, PHQ-9, and DASS-21. Such correlations are consistent with patterns observed with other recognized scales in the field. For instance, similar associations have been documented with the Brief Psychiatric Rating Scale [37] and the Behavioral Activation for Depression Scale (BADS) [38]. This convergence in correlations substantiates the scale’s validity and underscores its comparative reliability alongside these established instruments, thereby confirming its efficacy in capturing relevant psychological dimensions parallel to those assessed by the referenced scales.
This study is subject to several limitations. Firstly, the participants under study were drawn from the broader Afghan populace, without formal diagnostic assessments for mood disorders such as depression. Consequently, the assessment of the scale's sensitivity and specificity remains unfeasible. Secondly, the subjective nature of depression symptoms and inherent limitations in their objective assessment via self-reporting raise concerns regarding the potential influence of social desirability biases on respondents' responses. Lastly, the utilization of convenience sampling undermines the extent to which the findings of the present study can be generalized.
5 Conclusion and implications
The ANDs scale, consisting of 15 items, is developed to assess depression within the Afghan population. Its alignment with established measures such as the DSM-V based PHQ-9, as well as other widely used depression subscales like the DASS-21, CESD-20, and GHQ-28, suggests its potential utility in identifying depression symptomatology. The implications of the ANDs scale for clinical practice and public health are significant. Clinically, it could improve early detection and diagnosis of depression in the Afghan population, particularly by addressing cultural nuances in symptom expression. This may allow for better monitoring and tailoring of treatment. In public health terms, the scale supports efforts to enhance mental health services in Afghanistan, aiding in policy development and resource allocation to improve healthcare worker training and infrastructure. However, it is crucial for these findings to be replicated across diverse clinical and demographic samples to ensure the generalizability of the one-factor solution identified in this study. Further research is necessary to validate the use of the ANDs scale in clinical settings.
Data availability
The datasets during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Chen C. Recent advances in the study of the comorbidity of depressive and anxiety disorders. Adv Clin Exp Med. 2022;31(4):355–8.
Depressive disorder (depression). World Health Organization. https://www.who.int/news-room/fact-sheets/detail/depression. Accessed 2 Mar 2024.
Malhi GS, Bell E, Bassett D, Boyce P, Bryant R, Hazell P, Hopwood M, Lyndon B, Mulder R, Porter R, Singh AB. The 2020 Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for mood disorders. Aust N Z J Psychiatr. 2021;55(1):7–117.
Wong JJ, Tricco AC, Cote P, Liang CY, Lewis JA, Bouck Z, Rosella LC. Association between depressive symptoms or depression and health outcomes for low back pain: a systematic review and meta-analysis. J Gen Intern Med. 2022;37(5):1233–46.
Iyer VJ. Modern psychiatry: confluence of mind, science, and society. In: Rezaei N, editor. Multi-disciplinarity and interdisciplinarity in health. Cham: Springer International Publishing; 2022. p. 105–22.
Marx W, Penninx BW, Solmi M, Furukawa TA, Firth J, Carvalho AF, Berk M. Major depressive disorder. Nat Rev Dis Primers. 2023;9(1):44.
Jurado-González F, García-Torres F, Contreras A, Muñoz-Navarro R, González-Blanch C, Medrano LA, Ruiz-Rodríguez P, Cano-Vindel A, Moriana JA. Mapping bridges between anxiety, depression, and somatic symptoms in primary care patients: a network perspective. Curr Psychol. 2023;19:1–3.
Ahmed A, Patil PS. Navigating the shadows: a comprehensive review of suicide in the geriatric population. Cureus. 2024. https://doi.org/10.7759/cureus.53352.
Bertolín-Guillén JM. Psychiatric psychobiological treatment versus exclusive psychological treatment in depressive disorders. Am J Psychiatr Neurosci. 2023;1(1):13–21.
Beck AT, Steer RA, Brown GK. Beck depression inventory. San Antonio: The Psychological Corporation; 1987.
Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606–13.
Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56.
Greenberg SA. The geriatric depression scale (GDS). Best Pract Nurs Care Older Adults. 2012;4(1):1–2.
Zung WW. A self-rating depression scale. Arch Gen Psychiatr. 1965;12(1):63–70.
Radloff LS. The CES-D scale: a self-report depression scale for research in the general population. Appl Psychol Meas. 1977;1(3):385–401.
Montgomery SA, Åsberg MA. A new depression scale designed to be sensitive to change. Br J Psychiatr. 1979;134(4):382–9.
Rush AJ, Gullion CM, Basco MR, Jarrett RB, Trivedi MH. The inventory of depressive symptomatology (IDS): psychometric properties. Psychol Med. 1996;26(3):477–86.
Buchanan DB. Depression anxiety stress scales—short form (DASS-21). 2024. https://novopsych.com.au/assessments/depression/depression-anxiety-stress-scales-short-form-dass-21/. Accessed 25 Feb 2024.
Westerman TG, Dear GE. The need for culturally valid psychological assessment tools in Indigenous mental health. Clin Psychol. 2023;27(3):284–9.
Ali AM, Alkhamees AA, Hori H, Kim Y, Kunugi H. The depression anxiety stress scale 21: development and validation of the depression anxiety stress scale 8-item in psychiatric patients and the general public for easier mental health measurement in a post COVID-19 world. Int J Environ Res Public Health. 2021;18(19):10142.
Malakouti SK, Fatollahi P, Mirabzadeh A, Zandi T. Reliability, validity and factor structure of the GHQ-28 used among elderly Iranians. Int Psychogeriatr. 2007;19(4):623–34.
Sterling M. General health questionnaire–28 (GHQ-28). J Physiother. 2011;57(4):259.
Neyazi A, Tosun N, Hosaini F, Saokaew S. Validity and reliability of the Dari version of the CES-D scale among the general population of Herat, Afghanistan: a methodological study. Razi Int Med J. 2022;2(1):10–9.
Maroufizadeh S, Omani-Samani R, Almasi-Hashiani A, Amini P, Sepidarkish M. The reliability and validity of the Patient Health Questionnaire-9 (PHQ-9) and PHQ-2 in patients with infertility. Reprod Health. 2019;16(1):1–8.
Kleinman A. Do psychiatric disorders differ in different cultures? In: Brown JP, Closser S, editors. Understanding and applying medical anthropology. London: Routledge; 2016. p. 300–10.
Kirmayer LJ. Cultural variations in the response to psychiatric disorders and emotional distress. Soc Sci Med. 1989;29(3):327–39.
De Vaus J, Hornsey MJ, Kuppens P, Bastian B. Exploring the East-West divide in prevalence of affective disorder: a case for cultural differences in coping with negative emotion. Pers Soc Psychol Rev. 2018;22(3):285–304.
Sartorius N. Depressive disorders in different cultures: report on the WHO collaborative study on standardized assessment of depressive disorders. Geneva: World Health Organization; 1983.
Cao CH, Liao XL, Jiang XY, Li XD, Chen IH, Lin CY. Psychometric evaluation of the depression, anxiety, and stress scale-21 (DASS-21) among Chinese primary and middle school teachers. BMC Psychol. 2023;11(1):209.
Ramírez-Vélez R, Olabarrieta-Landa L, Rivera D, Izquierdo M. Factor structure of the 10-item CES-D Scale among patients with persistent COVID-19. J Med Virol. 2023;95(1): e28236.
Nadeem MU, Kulich SJ, Bokhari IH. The assessment and validation of the depression, anxiety, and stress scale (DASS-21) among frontline doctors in Pakistan during fifth wave of COVID-19. Front Public Health. 2023;16(11):1192733.
Oliveira TA, Gouveia VV, Ribeiro MG, Oliveira KG, Melo RL, Montagna E. General Health Questionnaire (GHQ12): new evidence of construct validity. Cien Saude Colet. 2023;6(28):803–10.
Osman A, Wong JL, Bagge CL, Freedenthal S, Gutierrez PM, Lozano G. The depression anxiety stress Scales—21 (DASS-21): further examination of dimensions, scale reliability, and correlates. J Clin Psychol. 2012;68(12):1322–38.
Ohno S, Takahashi K, Inoue A, Takada K, Ishihara Y, Tanigawa M, Hirao K. Smallest detectable change and test-retest reliability of a self-reported outcome measure: results of the Center for Epidemiologic Studies Depression Scale, General Self-Efficacy Scale, and 12-item General Health Questionnaire. J Eval Clin Pract. 2017;23(6):1348–54.
Roy K, Muhammad N. Adaptation and validation of the general health questionnaire (GHQ-28) in Bangladeshi context. Jagannath university. J Psychol. 2021:11(1):11–30.
Sun Y, Fu Z, Bo Q, Mao Z, Ma X, Wang C. The reliability and validity of PHQ-9 in patients with major depressive disorder in psychiatric hospital. BMC Psychiatr. 2020;20:1–7.
Overall JE, Gorham DR. The brief psychiatric rating scale. Psychol Rep. 1962;10(3):799–812.
Kanter JW, Mulick PS, Busch AM, Berlin KS, Martell CR. The Behavioral Activation for Depression Scale (BADS): psychometric properties and factor structure. J Psychopathol Behav Assess. 2007;29:191–202.
Acknowledgements
We would like to express our sincere gratitude to all the participants who enrolled in this study.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
AN designed the study. AN, HF, and MNo and MN contributed in data collection of this study. AN analyzed the data. AN, BAR, AQM, NR, HF, MNo, and MN prepared the draft of the manuscript. AN critically reviewed, rewrote, edited, and finalized the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
The current study received ethical approval from the Ethical Committee of the Afghanistan Center for Epidemiological Studies, identified by reference number #21.1.041. Upon initial engagement with participants, a comprehensive overview of the study was provided, and informed consent was secured from all participants. Participants were explicitly informed of their right to withdraw from the study at any juncture. The execution of all methods adhered to pertinent ethical guidelines and regulations.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Neyazi, A., Mohammadi, A.Q., Rahimi, B.A. et al. Afghanistan National Depression Screening (ANDs) scale: development and psychometric testing. Discov Psychol 4, 60 (2024). https://doi.org/10.1007/s44202-024-00172-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44202-024-00172-z