Abstract
The water buffalo breed known as the Murrah buffalo (Bubalus bubalis) is primarily raised for its milk. It is preserved in the Indian states of Haryana and Punjab, namely in the districts of Bhiwani, Agra, Hisar, Rohtak, Jind, Jhajhar, Fatehabad, Gurgaon, and the Delhi capital city. The fat from buffalo milk also contains ketoglycerides. While β-lactoglobulin, which contains branched chain and essential amino acids as well as a retinol-binding protein that may modulate lymphatic processes, is absent from human milk, buffalo milk has somewhat more of this protein than cow’s milk and is also the primary source of whey protein. It has the ability to create antimicrobial peptides that work well against gram-positive bacteria and are released by proteolytic enzymes during trypsin-catalyzed digestion. International dairy product production is increasingly shifting to domestic buffalo as a substitute source. Buffalo milk contains a high amount of calcium, a mineral that is necessary for the development of bones. Moreover, it contains casein-derived peptides, which may promote bone health and reduce the likelihood of osteoporosis, a disorder characterized by weakening bones and an increased risk of fractures. Customers love buffalo milk because of its excellent nutritional value. It may be eaten raw or used to manufacture premium products like cheese, yogurt, ice cream, and other traditional dairy products.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Bubalus bubalis the water buffalo is a bovid that originate from the Indian subcontinent and Southeast Asia [1]. These days, it can also be found in Italy, the Balkans, and Australia, North and South America and various African nations as well. Based on morphological and behavioural characteristics, two distinct subspecies of water buffalo are recognized: the river buffalo, which is found in the Indian subcontinent and further west in the Balkans, Egypt, and Italy and the swamp buffalo, which is found in Assam in the west through Southeast Asia to the Yangtze valley of China in the east [2]. According to phylogenetic analysis, it is possible that the river-type water buffalo originated in western India and was brought under domestication some 6300 years ago [2]. On the other hand, water-based buffalo of the swampy kind most likely originated spontaneously in Western Southeast Asia and became domesticated about 3000–7000 years ago [3]. The Meluhhas exported water buffaloes from Mesopotamia, which is modern-day Iraq, to the Indus Valley civilization approximately 2500 BC [4]. In comparison to dairy cattle, water buffaloes' milk is higher in fat and protein, making them particularly suited for cultivating rice fields [5]. Buffaloes living in rivers favor deep water. Swamp buffaloes love to spend their days wallowing in mudholes that they have dug themselves. They develop a heavy layer of mud on them when they are wallowing. Both are able to withstand hot, humid weather with winter lows of 0 ℃ (32 °F) and summer highs of 30 ℃ (86 °F) or more. Water accessibility is crucial in hot areas because they require wallows, rivers, or splashing water to help regulate temperature. Some kinds of water buffalo are able to survive on salty sandy areas and salty seashores [6]. Numerous aquatic plants support the growth of water buffalo. They forage submerged during floods, lifting their heads above the water and bringing large amounts of food plants with them [7]. Reeds, a Cyperaceae plant called Arundo donax, Eichhornia crassipes, and Juncaceae are all food items consumed by water buffalo [8]. Green fodder is often employed for intense milk manufacturing and fattening processes. Many fodder plants are dried and stored as hay, chaffed, or pulp [9]. Some fodders include alfalfa, leaves of bananas, stems, or trimmings of any kind cassava, Mangelwurzel, esparto, leucocephala Leucaena kenaf plant, grain, oats, pandanus seed, peanut, wheat, soybean, sugarcane, bagasse, and carrots. Buffalo have been safely fed pineapple trash and citrus pulp. Whole sun-dried dates, up to 25% of the usual feed mixture, are fed to milk buffalo in Egypt [10]. There were over 172 million water buffaloes in the world as of 2011. There are 208,098,759 water buffalo in the world, which are spread out throughout 77 nations and five continents [11]. The river-type and swamp-type water buffaloes, which make up more than 95.8% of the water buffalo population worldwide, are reared in Asia [12]. In 2003, there were more than 97.9 million water buffalo in India, making about 56.5% of the total population. They are largely of the river variety. As of 2003, there were about 773,000 carabao buffaloes in Bangladesh, approximately 3 million swamp buffaloes in Vietnam, and 3.3 million swamp-type carabao buffaloes in the Philippines [13]. With 22.76 million people, all of whom were swamp-type, Chinese had the world's second-greatest population in 2003. Certain breeds were only maintained in the lowlands, while others were only maintained in the highlands. In 1997, Sri Lanka’s population was predicted to be at 750,000. Although it is now practically extinct and primarily employed as a tourist attraction. In the past, water buffalo were domesticated pets in Okinawa prefecture and the Ryukyu Islands of Japan [14]. There were 23.47 million water buffalo in Pakistan, and they were raised for dairy products. By 2021, Karachi will have more than 400,000 water buffalo, which will supply the local population with both dairy products and meat [15]. The 1950s saw the hunting of water buffaloes for their meat and hides, which were consumed at home and internationally. Live exports began in the late 1970s and lasted into the early 1980s to Cuba and other nations. Insemination by artificial means techniques involve crossing swamp buffaloes with river buffaloes in various regions of Australia. Some of these hybrids are utilized to produce milk. River buffalo are the ideal animals for laboring in the dense muck of rice fields because of their large hooves and flexibility foot joints [16]. Their nickname is “the living tractor of the East.” They are employed for threshing and sheaf transportation in the majority of nations that grow rice. The power equipment for raising water as well as presses for sugarcane and oilseeds. Every year, more than a staggering 26 million water buffaloes are murdered for their meat. They furnish the world's food supply with more than 72 million tonnes of milk and three million thousand metric tons of meat annually, the most of which are produced in areas that are vulnerable to malnutrition. In India, the main purposes of keeping river buffaloes are movement and the manufacture of milk. You’ll discover that buffalo milk has higher levels of fat, protein, lactose, vitamins, and minerals than cow’s milk. It is ideal for the manufacturing of dairy products made from fat since it is also whiter and thicker in consistency [17]. A significant portion of vitamin B12 is present in whole buffalo milk, which has a number of positive health effects including a decreased risk of heart attack, stroke, and other cardiovascular conditions. It also includes additional crucial minerals like vitamins A, C, and B6 [18]. The purpose of this study is to review the most recent information on the qualities, possibilities, and numerous buffalo milk products that have been created on a global scale.
2 Methodology
The scientific papers used for this review were retrieved from several databases from like Science Direct (https://www.sciencedirect.com/), Web of Science (https://mjl.clarivate.com), Pubmed (https://Pubmed.ncbi.nln.nih.gov), SPIE Digital Library (https://www.spiedigitallibrary.org/?SSO=1), Springer open (https://www.springeropen.com), “ACS publications (https://pubs.acs.org/journal/esthag) and JSTOR (www.jstor.org) databases. Using the following keywords: “properties of buffalo milk,” “vitamin content of buffalo milk,” “buffalo milk products,” “differences between buffalo milk and cow milk”, “ origin of buffalo milk” “ therapeutic value of buffalo milk”,”Countries with highest buffalo milk production capacity”, “ medical applications of buffalo milk.
3 Milk production potential of buffalo breeds
With an annual production over 75 million metric tons and a consistent growth rate of approximately 3%, milk from buffaloes is the second- highest producer of milk in the world [19]. Typical cow milk contains an average of 3–4% fat and 3.5% of protein [20]. In contrast, buffalo milk has an average protein content measuring between 4 and 4.5% and an average fat content of about 6–8%. As a result, the food contribution represented by the volume of buffalo milk produced annually is approximately doubled when measured in terms of energy-corrected milk.46 nations in Asia, Africa, Latin America, and Australia have a combined population of 175 million [21]. Ninety-six percent or 167 million of the world’s buffaloes live in Asia, Pakistan, China, and India. These countries are also the three top buffalo rearing nations in the world [22]. There are several promising dairy buffalo breeds in India, including the Murrah, Jaffarabadi, Surati, Mehsana, Nagpuri/Pandharpuri, and Marathwadi. India produces 70% of the buffalo milk (BM), with the rest 30% coming from Pakistan (Kundhi, Azi kheli and Nili-Ravi), 20% from China (Nili-Ravi breed), and 4% from Egypt (Murrah breed) [23]. If the right processing technologies are used, buffalo milk’s richness makes it very ideal for processing. To make one kilogram of cheese, a cheese manufacturer needs eight kilograms of milk from cows but only five kilograms of buffalo milk [24]. While just 10 kg of buffalo milk is needed to make 1 kg of butter, the same kilogram of butter will require 14 kg of cow milk. Processors recognize the importance of buffalo milk as a result of these large yields [25]. A system of milk cooperatives in India and Pakistan is used to market milk, ensuring a consistent price for farmers all year round. Butterfat content in buffalo milk is twice that of cow milk [26]. In addition to ghee, buffalo milk is also used to make butter, cheese, full-cream milk powder, skim milk powder, and new-born formula [27]. High levels of calcium, a mineral essential for the formation of bones, are present in buffalo milk [28]. Additionally, it contains peptides produced from casein, which may strengthen bones and reduce the likelihood of osteoporosis, a disorder characterized by weakening bones and an increased risk of injuries [29]. Vitamins A and C are abundant in buffalo milk, and they are vital for the normal functioning of the immune system [30]. These two vitamins serve as antioxidants, ridding the body of risky free radicals that might result in chronic diseases [31]. Table 1 shows the countries that produces the most buffalo milk in the world.
3.1 Buffalo milk properties
Buffalo milk fat differs quantitatively in the distribution of triglycerides and in its physical attributes from cow milk fat, with a slightly higher concentration of saturated lipids [32]. The milk from buffalo is exceptionally smooth and white because of the lack of carotene in it. The acidity of buffalo milk ranges between 0.05% to 0.20%, its colostrum also has a higher acidity than mature milk [33] Nevertheless, the freezing point is raised by vacuum treatment, cold storage, and the addition of water, and it is lowered by boiling and souring. According to [34], the maximal buffering index for cow milk was 0.035 and for buffalo milk it was 0.042 at a pH range of 4.9 to 5.1. 0.035 at pH 5.1–5.2 for cow milk and 0.035 at pH 5.1–5.2 for buffalo milk. Cow milk’s refractive index ranges from 1.345 to 1.348 at 40 degrees Celsius, while buffalo milk's ranges from 1.346 to 1.353, with proteins and lactose playing a part. Buffalo milk (32–85 g) has curd tension that increases toward the end of lactation and is nearly 1.5 times higher than cow milk (28–54 g), according to [34]. Curd tension is, however, reduced by heating techniques such pasteurization, boiling, sterilization, and homogenization, and adding the salt citrate or sodium hexametphosphate, which decrease it by 10–28%, 58%, and 87%, respectively [35]. Other characteristics of buffalo milk include a freezing point of -0.528, viscosity of 1.79, specific gravity of 1.032, electrical conductivity of 4.46 and surface tension of 49 [36].
3.1.1 Shelf life
Most milk products are sterile when they are first produced, and only get contaminated following production while being stored causing them to lose quality [37]. Preventing any kind of contamination before, during, or after the processing of the products is therefore crucial from an economic as well as a safety point of view [38]. No matter where it comes from, due to its extremely nutritive quality and short shelf life, raw milk from is highly susceptible for pathogenic and spoilage microbes to proliferate [39]. In order to increase the shelf-life of dairy products, intervention is frequently required because certain milk enzymes can also result in unfavourable changes during storage [40]. Pasteurization conditions are meant to inactivate lipoprotein lipase, Mycobacterium tuberculosis; which are milk spoilage bacteria. Alkaline phosphatise and Coxiella burnetii, are resistant to heat and do not form spores and so are used as indicators of effective pasteurization [41]. Nevertheless, they are not potent enough to deactivate plasmin or bacterial lipases and proteases [42]. Buffalo milk had a slightly shorter shelf life (6 days) than bovine milk which lasts between 7 and 10 days after pasteurization [43]. Beyond these variables, the acidity level of buffalo milk can also impact its shelf life. As opposed to cow’s milk, buffalo milk usually has a higher pH, making it more prone to spoilage. Still, this can be lessened by adding natural preservatives to change the pH [44]. Moreover, the milk's shelf life is also influenced by its own quality [45]. When milk is produced under sanitary circumstances from healthy buffaloes, it usually keeps longer than milk from uncontrolled environments [46]. Any impurities or adulterants might hasten the deterioration process and reduce the shelf life [47].
3.1.2 Gross chemical composition
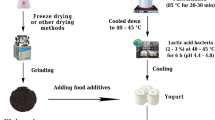
Buffalo milk is the milk obtained from domesticated buffalo, or Bubalus bubalis. The attributes of buffalo milk—namely, its richer fat content, fatter globule size and whiter milk colour—make it simple to identify [48]. The amount of milk produced by buffalo is lower (7–11 litters per day) than that of cows (15–20 litters). But buffalo milk has a higher nutritious grade than cow’s milk, making it a potential better product. Buffalo milk is more cost-effective to process and has a thicker consistency. Buffalo milk is simple to process as it contains less water (83% as against 88% for bovine milk) [49]. An individual with a cow milk allergy may also be intolerant to buffalo milk. Buffalo milk collected from various districts in West Sumatra had the following nutritional values: pH 6.06–6.39, protein 7.22–7.83%, fat 7.18–7.88%, and moisture content 80.62–81.03%. Buffalo milk has a protein content of 6.03% and a fat content of 12.40%, according to [18]. Lactic acid bacteria, which may have probiotic properties, are present in buffalo milk [50]. Probiotics can be utilized as the primary element in curds since they have the ability to inhibit pathogenic microorganisms. Probiotics may be made from the Lactic Acid Bacteria (LAB) (Lactobacillus fermentum L23) that is present in buffalo milk. Fermented milk contains lactic acid bacteria that produces antimicrobial chemicals [51]. These are used as natural antibiotics to treat infections, improve the gastrointestinal tract, and extend the shelf life of food products [52]. Table 2 shows the chemicals characteristics of buffalo and cow milk.
3.1.3 Protein
When comparing buffalo milk to cow’s milk, the protein level of buffalo milk is usually higher. Protein content in cow’s milk is usually 3–4%, whereas protein content in buffalo milk is typically 4–5% [53]. Like cow’s milk, buffalo milk has two primary protein classes that are identified by how soluble they are at pH 4.6 and 20 ℃ [54]. Approximately 80% of the protein in milk is made up of insoluble caseins, and the remaining 20% is made up of soluble whey proteins [55]. Buffalo milk has larger amounts of caseins and whey proteins since it has higher protein content compared to bovine milk [56]. They are a heterogeneous polymorphic group of proteins made up of many enzymes, lactoferrin, proteose peptone, and other small proteins (< 10%), as well as β- and α-Lactalbumin (50%, 20%, 10%, and 10%, respectively) and serum albumin (10%) [57]. There have been reports that the whey proteins from buffalo milk are more resilient to denaturation caused by heat than those from cow milk and this is particularly because of the behaviour of its immunoglobulin; IgG, IgM and IgA which are incompletely denatured even at high temperatures of 88 ℃ [58]. Bovine milk, casein micelles are supramolecular colloidal aggregates formed by four distinct gene products (αS1-, αS2-, β-, and κ-casein) found in buffalo milk [59]. Older adults and babies shouldn’t drink buffalo milk because of its high protein content. High-quality complete proteins, or those that include all the essential amino acids in the amounts needed by the body, are found in buffalo milk proteins [60]. Proteins from buffalo milk have an energy value of 17.2 J g − 1. Because of its high nutritional content, consumers greatly like buffalo milk, which may be consumed raw or used to make high-quality goods like cheese, yogurt, ice cream, and other conventional dairy products [61]. While human milk lacks β-lactoglobulin, that includes essential as well as branched chain amino acids, buffalo milk has slightly higher amounts of this protein which is also the main source of whey protein [32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62]. It can produce antibacterial peptides that are effective against gram-positive microorganisms and are released during trypsin-catalysed digestion through proteolytic enzymes. Table 3 shows the protein content of buffalo, Cow and camel milk.
3.1.4 Fat
The consistency of the milk is actually determined by its fat content. When compared to buffalo milk, cow milk has less fat in it. Cow milk is thinner than buffalo milk because of this [63]. Cow milk only has 3–4% fat, but buffalo milk has about 7–8% fat [64]. Buffalo milk fills you up more completely because of its weight, which also makes it take longer to digest [21]. Due to its higher concentration of high melting triglycerides (9–12%) than cow milk’s (5–6%), buffalo milk fat is more solid than cow milk protein. Triglycerides that include butyric acid are found in higher proportion in buffalo milk (50%) than in cow milk (37%) [65]. Because buffalo milk fat has a larger percentage of triglycerides and thus stronger emulsifying potential than cow milk fat [40]. The size of fat globules in buffalo milk is larger (4.15–4.6 μm) compared to cow milk (3.36–4.15 μm) [41]. Compared to bovine milk fat, which has 0.33% free fatty acids, buffalo milk fat has 0.22%. Bovine milk contains higher amounts of unsaponifiable matter (414–450 mg 100 ml-1) than buffalo milk (392–398 mg 100 ml-1) [66]. Like this, buffalo milk has smaller phospholipid concentration (21 mg/100 ml) than 37.37 mg/100 ml) [67]. The amount of total and free cholesterol in 100 g of buffalo milk ghee is 275 mg and 210 mg, respectively [68]. This is significantly less than the comparable amounts in cow milk ghee, which are 330 mg and 280 mg per 100 g, respectively [69]. Conversely, Buffalo milk has significantly more esterified cholesterol (64 mg 100 g-1) than cow milk (48 mg 100 g-1) [70]. The amount of ubiquinone in buffalo milk fat is around 6.5 μg g-1, which is higher than the amount in cow’s milk fat, which is 5 μg g-1 [71]. But lutein and lanosterol, which are found in buffalo milk fat at 3.0 and 83 μg g-1, respectively, are much less abundant than they are in cow milk fat, which has 4 and 93 μg g-1, respectively [72]. In addition, buffalo milk fat has less ether content—about 0.8 μg g-1—than bovine milk fat, which has 0.9 μg g-1. In buffalo milk, there are 9.8 μg of total carbonyls per gram of fat. Buffalo milk fat has 1.85 μg g-1 of alkanols (ethanol, methanol, and butanol) compared to 2.29 μg g-1 in bovine milk fat [73]. Buffalo milk fat also includes ketoglycerides. Research studies have shown that buffalo milk can help decrease lipoprotein levels [74]. This happens because it has a higher proportion of heart-healthy lipids, like fatty acids called omega-3 and omega-6. Table 4 shows the amount of fat content in Buffalo, cow and camel Milk.
3.1.5 Lactose
Milk’s lactose content differs based on what species it is. The amount of lactose in the milk of humans is substantial; it makes up between 6.5 and 7.5% of the milk [75]. The amount of lactose in raw cow milk is approximately 4.7%, in raw goat milk it is approximately 4.7%, and in raw buffalo milk it is approximately 4.86% [76]. Less than 5% is the average for goats and bovine milk, although this varies by breed [62]. It is common practice to supplement human babies’ diets with lactose, sucrose, fructose, or malto-dextrin [77]. Many women find that their capacity to digest lactose improves throughout pregnancy, particularly as the pregnancy progresses [78]. As a result of this, you may be able to consume milk and other milk and yogurt without experiencing symptoms even if you are typically lactose sensitive [79]. Table 5 shows the lactose contents of buffalo, Cow and Camel milk.
3.1.6 Minerals
Milk from buffalo is a great source of minerals like calcium, magnesium, phosphorus and zinc [80]. Although buffalo milk provides more calories per serving and more of the vital elements than cow’s milk, both are very nutrient-dense and deliver a significant amount of minerals and vitamins [81]. Plus, buffalo milk contains greater nutritional value and vitamins. However, it provides 41% of the Daily Value (DV) for phosphorus, which includes 32% of the DV for calcium, 19% for magnesium, and 14% for vitamin A, whereas cow's milk only provides 29%, 21%, 6%, and 12% of these vitamins. According to reports, buffalo milk has an average calcium level of 180 mg/100 g. Buffalo milk's phosphorous and magnesium contents were 16–30 mg/100 g and 89–138 mg/100 g, respectively [82]. The building blocks of bodily tissue, as well as the constituents of several enzyme and other physiologically active molecules, are among the nutritionally necessary macrominerals and trace elements found in milk [83]. Additional small amounts of elements found in milk are unknown in their function and are not considered essential; however, information from investigations on the nutrition of humans and animals indicates that certain elements, including Li, B, Ti, Rb, and Sr, have positive biological effects [84]. While the amounts of Mn, Se, and Co are equal in milk from both species, the concentrations of Ca, P, Mg, Zn, Fe, Cu, B, Ti, and Sr are higher in buffalo milk than in cow milk, while the percentages of K, Na, Mo, Li, and Rb are lower [85]. Milk’s functional value and antioxidant capability were increased by glutathione and selenium [86]. Table 6 presents the mineral content of milk from cows, buffalo, goats, and sheep.
3.1.7 Vitamins
Vitamins play a variety of functions in the body, such as antioxidants, and necessary co-factors in metabolism, and oxygen transport [87]. They support the body’s utilization of fat, protein, and carbohydrates. itamins that are soluble in water and found in milk include thiamin (B1), riboflavin (B2), niacin (B3), pantothenic acid (B5), vitamin B6 (pyridoxine), vitamin B12 (cobalamin), vitamin C, and folate [86]. Vitamin B12, riboflavin, and thiamin are all found in good amounts in milk. Despite having minimal amounts of pantothenic acid, vitamin B6, vitamin C, [88] and folate, milk is not thought to be a major dietary source of these vitamins [89]. Dairy products contain fat-soluble vitamins A, D, E, and K [90].The amount of fat in milk and cheese determines the amount of fat-soluble vitamins it contains. For reduced fat (2% fat), low fat (1% fat), and skim milk to have the same nutritional value as whole milk, vitamin A fortification is required [91]. According to a dietary standpoint, the fact that raw buffalo milk has a comparatively greater concentration of vitamin C is relevant since the vitamin has potent antioxidant properties and can serve as a substitute source of the vitamin in extreme conditions such as regions that are semi-arid or arid [92]. Although heating can usually prolong the freshness of milk, subject to the intensity of heat treatment, heating significantly reduces the amount of B-vitamins (B1, B2, B6, and folic acid) in the milk products of buffalo and cows [93]. The milk fat division is linked to the lard-soluble A, D, E, and K vitamins, whilst the water-soluble vitamins such as B complex and C are linked to the water phase. As a result of their instability, treatment can lower milk's effective nutritional value [94]. While the nutrients that are water- stay in skim milk or whey, the vitamins that dissolve in fat are maintained by the cream throughout processing [95]. Lactic acid bacteria, also known as LAB, are employed on a commercial basis to boost dairy products’ B vitamin synthesis. A reroute intricate biosynthetic processes in order to produce metabolites that are beneficial to the customer’s health, including B vitamins, is one of the latest advancements in LAB metabolic science [96]. Table 7 shows the Comparison of the vitamin content of buffalo, Cow, goat and sheep below.
3.1.8 Milk enzymes, protective protein and hormones
Glutathione, which functions as an anti-oxidant in the body, is present in milk. Glutathione is a tripeptide consisting of a combination of glutamic acid, glycine, and the sulphur amino acid known as cysteine [97]. It can oxidize to form oxidized glutathione, which might reduce the number of reactive oxygen species (ROS) in the cells and possibly even eradicate them. Since ROS prevent the pro-insulin gene from being expressed, cysteine plays a role in controlling the synthesis of insulin in pancreatic cells [98].Cysteine appears to play a variety of significant roles in leukocytes, including enhancement of growth, anti-apoptotic activity, and structure regulation of the production of cytokines [99]. Native digestive enzymes are components of milk that come from four primary sources such as somatic cells (leucocytes), milk fat globule membrane (MFGM), secretory cell the cytoplasm, and blood plasma [100]. Y-glutamy transferase (Y-GGT), glutathione peroxidase, also known as (GSH), lactoperoxidase (LPO), lipase enzyme (LPL), xanthine oxidoreductase (XOR), plasmin, ribonuclease (RNase), sulphydryI oxidase (SHOx), super oxide dismutase (SOD), and y-glutamy transferase enzyme (Y-GGT) are some of the best-characterized digestive enzymes found in milk [101]. There aren’t clear vital functions for the majority of the natural enzymes in milk when it comes to biosynthesis and emission [102]. Moreover, since those enzymes do not significantly improve the nutritional value or organoleptic qualities of milk, one of the main objectives of many dairy operations is to heat-kill them [103]. The products will suffer from hot weather processes, especially if milk products are made using several heat treatment techniques [104]. These techniques won’t be enough to eliminate every enzyme. Protein enzymes and lipases that survive sterilization and spray-drying treatments have the potential to significantly alter the taste and activity of milk and other dairy products [105]. A class of enzymes known as proteinases work internally on polypeptide chains as opposed to cutting individual amino acids or dipeptides from the terminals of the polypeptide chains [106]. Additionally, this class of proteinases is typically divided into four subgroups according to the enzyme’s mode of function. The main naturally occurring proteinase in buffalo milk is called plasmin [107]. The zymoger plasminogen receptor plasmin inhibitors, plasminogen activators (PAs), and regulators of plasminogen activators are the five components that make up the the plasmin protein system [108]. When a mastitic illness or late lactating occurs, this system enters the milk from the blood, and plasmin activity rises [109]. Mostly present as the dormant antecedent a protein known as plasmin is found in milk [110]. Better bacterial quality, longer storage times, and the advent of elevated temperatures processed milk have all resulted in modifications in the dairy business’s practices [111]. As a result, the plasmin protein in buffalo has become more significant because of its association with microbe proteases, which making it possible for the regulation of its levels in order to enhance the nutritional value of milk and milk products [112]. As of right now, there is no distinct distinction made regarding growth factors, cytokines, and milk hormones [113]. In an infant or adult, each of these compounds is necessary for the development, development, or repairing of several cell types [114]. Add to that the fact that all of these agents that encourage growth are chemicals that cells release as a means of communication [115]. Hormones, to put it shortly, are chemicals that the cells of a certain tissue transfer into the extracellular media in order for them to go to a new location of action (endocrine activity) and trigger a particular reaction. When present in small amounts, proteins or glycoproteins known as cytokines—produced by a variety of cell types—have a significant pharmacological impact on other cells nearby [116]. Compared to immune globulin or lactoferrin, the amounts of the more than 50 growth stimulants and enzymes found in milk are significantly smaller [117]. Many hormones with minimal nutritional or diagnostic relevance that are present in milk at trace quantities can be found there [118]. The biological functions of the hormones in human beings and milk from cattle have, however, been the subject of numerous investigations [119]. The active transport process within the gland that produces breast milk is where hormones from the blood are released into milk [120]. The mammary gland can also produce a number of hormones that are then expelled as milk. The four primary hormone groups—pituitary, hypothalamus, adrenal, and gonadal hormones—represent the majority of hormones found in milk [121].
3.2 Functional properties and medicinal applications of buffalo milk
Buffalo milk will be a great substitute for cow milk especially for body builders and those who need to gain weight because of its higher carbohydrate and fat content [21]. Buffalo milk is said to provide therapeutic benefits and to act as an adjuvant. Numerous elements included in buffalo milk may provide numerous health advantages for people [75]. Its ingestion has the potential to reduce blood pressure, triglyceride levels, and cholesterol levels. Consequently, regular ingestion of fermented buffalo milk may benefit the heart. Many of buffalo milk's bioactive ingredients contribute to these possible health advantages [122]. These substances are obtained from buffalo milk protein hydrolysates by fermentation with lactic acid bacteria (LAB) or by employing proteolytic enzymes. Alternatively, they are found naturally in buffalo milk [123]. Protein from buffalo milk and its derivatives is abundant and may have favorable health-regulating impacts on humans, including anticancer, antibacterial (fights germs), and antihypertensive (lowers blood pressure) properties [124].
3.2.1 Buffalo milk for gastrointestinal health
Food intake has increased over the last 20 years as a result of claims about its health, including better immunity, gastrointestinal health, and general wellbeing [125]. Nowadays, the dairy industry is the one using probiotics the most, especially in fermented milk products like cheese, yoghurt, butter, and dairy drinks. There are still several challenges in terms of their stability and functionality in food. The use of buffalo milk, taking into account its probiotic properties as an energy source and natural cure for a range of illnesses, highlighting the opportunities for creativity and the significance of dairy-based goods as wellness boosters, by maintaining a healthy gut microbiota, probiotic consumption helps to modulate the host’s health and lowers the chance of developing a number of illnesses, including cancer, colitis, lactose intolerance, heart disease, and overweight [18]. Although bovine milk also possesses some probiotics, does not measure to the probiotic content of buffalo milk. Peptides are primarily found in milk and dairy products. These substances are tiny molecules that can be found in food items and have a good impact on the well-being of humans by having antioxidant, antibacterial, anticancer, antidiabetic, and immunomodulatory qualities, among other benefits [126]. Peptides thus play a role in multiple biological processes, including immunological, gastrointestinal, cardiovascular, endocrine, and neurological. Protein and fat contents, post-acidification profile, viability of Lactobacillus bulgaricus, Lactobacillus acidophilus, and Streptococcus thermophilus during a 21-day chilled storage period were evaluated in this study [127]. Additionally, resistance to in vitro gastrointestinal infections caused by probiotic dairy beverages made from buffalo and cow milks with varying levels of whey (0, 26, and 50%) was evaluated [128]. When whey was present, the progressive acidification that happened to all dairy products during storage was lessened. By the time the product’s shelf life was coming to an end, lactic acid bacteria were still viable, with buffalo dairy products exhibiting the highest levels (7.33 to 8.83 log cfu/mL) [129]. When gastrointestinal digestion was reproduced in vitro, fermented items created from buffalo milk exhibited superior bacterial viability compared to those made from cow milk, indicating a positive protective influence on the human microbiome [130].
3.3 Development of buffalo milk products
When it comes to cholesterol levels and the amount of unsaturated fatty acids it contains, buffalo milk is superior to cow milk. Like cow’s milk, buffalo milk can be used to make a broad range of dairy products, including UHT cream, ice cream, yogurt, clarified fat (ghee), cream, and butter, without requiring adjustments to manufacturing methods or technology [92]. Buffalo and milk from cows differ in composition, physical, chemical, and functional qualities, hence processing technology and machinery meant for cow milk product manufacturing is frequently ill-suited to handling buffalo milk [131]. The following considerations must be made while refining goods made from buffalo milk. Variations in the milk of buffalo are significantly influenced by age, breed, season, and nursing stage. Buffalo milk has higher fat, calcium, and total solids content [132]. These will impact how some products are processed and produced. The calcium content and casein micelles of buffalo milk are higher than those of cow milk [133]. The curd tension of buffalo milk is higher than that of cow milk, and the clotting time of its rennet is much shorter. Buffalo milk either ferments or ripens more slowly overall [134]. Because buffalo milk contains more saturated fatty acids than cow milk, the butter prepared from it is tougher. After cows, buffalo provide the second-highest milk supply in the world [135]. Their capacity to produce “toned” milk—a mixture of buffalo milk and milk made from reconstituting dry skim milk powder—makes them important to the Indian economy. Buffalo milk's richness makes it a great option for manufacturing, provided the proper processing techniques are applied [136]. To make one kilogram of cheese, a cheese maker needs eight kilograms of cow’s milk but only five kilograms of buffalo milk. One kilogram of butter can be made with just 10 kg of buffalo milk as opposed to 14 kg of cow’s milk [137]. Buffalo milk’s significant yield has led producers to acknowledge its worth. Buffalo cheese is widely valued as a specialty product because of its silky texture and pure white appearance. Cheeses like Pecorino in Bulgaria, the salty cheeses of Egypt, Gemir in Iraq, and mozzarella and ricotta in Italy are one of the most sought-after in many countries [138]. The Hilton Hotel Caracas purchases all of the cheese with other buffalo products like ghee, paneer produced by the small La Guanota milking herd in the Apure River basin (about 100 kg per day), which is sold for double the price of cheese, ghee and paneer manufactured from cow’s milk in Venezuela [18].
3.4 Swamp buffaloes milk production and composition
The domestic water buffalo (Bubalus bubalis), which produces the majority of milk in many nations, makes a substantial contribution to the world’s milk output. There are two subspecies of water buffaloes: the river buffalo and the swamp buffalo. In addition to being significant in the Near East, river buffalo milk makes up a large portion of the milk produced in Pakistan and India. Comparing swamp buffaloes to river buffaloes, the former are smaller and produce less milk. They are mostly raised for draught strength and are found mostly in Eastern Asia. Throughout the course of a 270–305 day lactation, the average milk supply per head ranges from 1.0 to 1.5 kg per day [139]. It appears that better feeding and management practices have little effect on their potential for milk yield. Nonetheless, their immense worth lies in their ability to serve as the fundamental genetic foundation for the development of animals with increased meat, milk, and draught potential. To maximize the milking potential of the current meat/draught animals in Thailand and other Asian countries, one method has been to cross-breed riverine buffaloes with swamp buffaloes. In a thorough analysis of the swamp-Murrah crossbreed’s milk yield and draught ability. Dias et al. [140] found that the crossbreed’s average milk yield for the 256-day average lactation period was 4 kg/day (2.0–6.3 kg/day). In his independent case study, which examined six herds of crossbred Murrah cows in a community, he found that the average milk output throughout a 242-day lactation was 6.9 kg/day. Body weight has been used as an indirect indicator of the crossbred’s working ability. Table 8 shows the variations in the milk production of buffalo breed.
3.5 Buffalo milk as probiotics
Probiotics are meals or supplements containing live microorganisms designed to preserve or enhance the body’s “good” bacteria, or natural microflora [141]. Foods known as prebiotics, which are usually high in fiber, serve as nourishment for the human microbiota. Probiotics, or good bacteria, may be present in milk. Probiotic-rich dairy products may be good for your health and help control weight, lower cholesterol, and lessen the risk of heart disease and stroke [142]. A bacteria called L. plantarum has been shown to improve the quality of dairy products and can be present in buffalo milk. This strain of L. Plantarum is recognized for its ability to improve vitamin and mineral absorption while also promoting protein synthesis [143]. These probiotics have the potential to strengthen immunity and improve defences against gastrointestinal problems.
3.6 Therapeutic value of buffalo milk
Twelve percent of the world’s milk produced is produced from buffalo milk. While both cow’s milk and milk from buffaloes are very nutrient-dense and offer a substantial number of vitamins and minerals, buffalo milk has a higher nutrient content and a greater number of calories per serving [144]. Compared to total cow’s milk, the milk from buffaloes has higher protein, fat, and lactose. Eating milk that has greater amounts of protein in it makes you feel fuller longer. This could help you eat less during the day, which would aid in weight loss and the reduction of body fat. Conversely, choosing cow's milk can be preferable if you wish to cut back on your fat consumption or if you have a moderate intolerance to lactose. Buffalo milk may offer a number of health advantages, according to research. Buffalo milk has a large amount of calcium, which is essential for the growth of bones [145]. Furthermore, it contains peptides produced from casein, which may strengthen your bones and lower your chance of developing osteoporosis, a condition marked by weakness of the bones and an elevated frequency of fracturing. The vitamins, minerals, and bioactive components in buffalo milk give it its antioxidant qualities. Free radicals are a class of chemicals that have negative effects on your body and have been connected to a number of ailments. Antioxidants are molecules that combat these damaging components. Buffalo milk contains beta-lactoglobulin and potassium, which may lower high blood pressure. The main whey protein, beta-lactoglobulin, is a significant source of bioactive substances that have positive effects on health. beta-lactoglobulin, sometimes known as beta-Lg. These inactive peptides are retained within the sequence of the precursor protein but can be released through enzymatic proteolysis in vivo or in vitro. These peptides have several positive health impacts on humans, including antihypertensive, antioxidant, and antimicrobial qualities. They can also reduce cholesterol levels in the body and have qualities similar to those of opioids. Studies have shown that bacteria like Lactis, which may be beneficial to the heart, may be found in fermented buffalo milk. Its ingestion has the potential to reduce blood pressure, triglyceride levels, and cholesterol levels. Consequently, consuming fermented buffalo milk on a daily basis may benefit the heart. Buffalo milk commerce in the nation is necessary because of the positive impact it has on people with osteoporosis and other conditions.
4 Conclusion
People who live in arid and semiarid regions can benefit from fresh buffalo milk and products made from them as a bioactive adjuvant. The benefits of using buffalo milk as a health adjuvant are becoming more widely known. This is because buffalo milk has distinct qualities that set it apart from other animal milks, like cow milk, including higher levels of fatty acids, proteins, and vitamin C. Characterizing the fat globule membrane, bioactive proteins, and peptides will require more research. Future research is necessary to address issues related to the coagulation of buffalo milk proteins by chymosin enzyme and acid in order to make cheese. Buffalo milk, both fresh and fermented, has been shown to boost immunity and offer specific health advantages to consumers based on the milk's distinct bioactive ingredients. For buffalo milk and products to gain acceptance in the marketplace and society, more thorough study is needed to validate the anticipated health benefits and describe the characteristics of this natural adjuvant. It is anticipated that this study would create new opportunities for modern buffalo dairies and foster interest in buffalo milk research.
Availability of data and materials
All data will be made available upon reasonable request.
References
Minervino AHH, Zava M, Vecchio D, Borghese A. Bubalus bubalis: a short story. Front Vet Sci. 2020. https://doi.org/10.3389/fvets.2020.570413.
Zhang Y, Colli L, Barker JSF. Asian water buffalo: domestication, history and genetics. Anim Genet. 2020;51(2):177–91. https://doi.org/10.1111/age.12911.
Rehman ur S, Hassan F, Luo X, Li Z, Liu Q. Whole-genome sequencing and characterization of buffalo genetic resources: recent advances and future challenges. Animals. 2021;11(3):904. https://doi.org/10.3390/ani11030904.
Hwabejire HO, Akpoghelie PO, Edo GI, Onoharigho FO, Agbo JJ. Microbiological properties, anti-nutritional and nutritional composition of spontaneously and starter culture fermented malted acha flour. Proc Indian Natl Sci Acad. 2023. https://doi.org/10.1007/s43538-023-00219-0.
Verduci E, D’Elios S, Cerrato L, Comberiati P, Calvani M, Palazzo S, Peroni DG. Cow’s milk substitutes for children: nutritional aspects of milk from different mammalian species special formula and plant-based beverages. Nutrients. 2019;11(8):1739. https://doi.org/10.3390/nu11081739.
Galloso-Hernández MA, Soca-Pérez M, Dublin D, Alvarez-Díaz CA, Iglesias-Gómez J, Díaz-Gaona C, Rodríguez-Estévez V. Thermoregulatory and feeding behavior under different management and heat stress conditions in heifer water buffalo (Bubalus bubalis) in the tropics. Animals. 2021;11(4):1162. https://doi.org/10.3390/ani11041162.
Wang L, Han Y, Yu H, Fan S, Liu C. Submerged vegetation and water quality degeneration from serious flooding in Liangzi Lake China. Front Plant Sci. 2019. https://doi.org/10.3389/fpls.2019.01504.
Sánchez E, Rivera-Vargas P, Serrat X, Nogués S. Arundo donax L.: how high photosynthetic capacity is maintained under water scarcity conditions. Agronomy. 2021;11(6):1089. https://doi.org/10.3390/agronomy11061089.
Sahoo A. Silage for climate resilient small ruminant production. Rumin Husb, Econ Health Asp. 2018. https://doi.org/10.5772/intechopen.74667.
Nath PC, Ojha A, Debnath S, Sharma M, Nayak PK, Sridhar K, Inbaraj BS. Valorization of food waste as animal feed: a step towards sustainable food waste management and circular bioeconomy. Animals. 2023;13(8):1366. https://doi.org/10.3390/ani13081366.
Ugbune U, Avwenaghegha JO, Edo GI. Geochemical distribution and ecological risk level of inorganic pollutant of River Ethiope sediment. Dutse J Pure Appl Sci. 2023;9(3a):187–93. https://doi.org/10.4314/dujopas.v9i3a.19.
Borghese A, Moioli B. Management of dairy animals: buffalo: Mediterranean Region. In: Encyclopedia of dairy sciences. Amsterdam: Elsevier; 2016. p. 845–9. https://doi.org/10.1016/B978-0-08-100596-5.21232-8.
Pineda PS, Flores EB, Herrera JRV, Low WY. Opportunities and challenges for improving the productivity of swamp buffaloes in Southeastern Asia. Front Genet. 2021. https://doi.org/10.3389/fgene.2021.629861.
Kayanne H, Hara T, Arai N, Yamano H, Matsuda H. Trajectory to local extinction of an isolated dugong population near Okinawa Island Japan. Sci Rep. 2022;12(1):6151. https://doi.org/10.1038/s41598-022-09992-2.
Di Stasio L, Brugiapaglia A. Current knowledge on river buffalo meat: a critical analysis. Animals. 2021;11(7):2111. https://doi.org/10.3390/ani11072111.
Singh I, Balhara AK. New approaches in buffalo artificial insemination programs with special reference to India. Theriogenology. 2016;86(1):194–9. https://doi.org/10.1016/j.theriogenology.2016.04.031.
Abdel-Hamid M, Huang L, Huang Z, Romeih E, Yang P, Zeng Q, Li L. Effect of buffalo breed on the detailed milk composition in Guangxi China. Foods. 2023;12(8):1603. https://doi.org/10.3390/foods12081603.
Vargas-Ramella M, Pateiro M, Maggiolino A, Faccia M, Franco D, De Palo P, Lorenzo JM. Buffalo milk as a source of probiotic functional products. Microorganisms. 2021;9(11):2303. https://doi.org/10.3390/microorganisms9112303.
Herron J, O’Brien D, Shalloo L. Life cycle assessment of pasture-based dairy production systems: current and future performance. J Dairy Sci. 2022;105(7):5849–69. https://doi.org/10.3168/jds.2021-21499.
Verduci E, Di Profio E, Corsello A, Scatigno L, Fiore G, Bosetti A, Zuccotti GV. Which milk during the second year of life: a personalized choice for a healthy future? Nutrients. 2021;13(10):3412. https://doi.org/10.3390/nu13103412.
Jiang M, Meng Z, Cheng Z, Zhan K, Ma X, Yang T, Zhao G. Effects of buffalo milk and cow milk on lipid metabolism in obese mice induced by high fat. Front Nutr. 2022. https://doi.org/10.3389/fnut.2022.841800.
Napolitano F, De Rosa G, Chay-Canul A, Álvarez-Macías A, Pereira AMF, Bragaglio A, Braghieri A. The challenge of global warming in water buffalo farming: physiological and behavioral aspects and strategies to face heat stress. Animals. 2023;13(19):3103. https://doi.org/10.3390/ani13193103.
Rafiq A, Shah MH, Mohany M, Tahir AA, Elsadek MF, Qayyum MA, Abbasi AM. Evaluation of potentially toxic trace metals and associated health risk assessment in buffalo milk. Int J Environ Res Public Health. 2022;19(22):14678. https://doi.org/10.3390/ijerph192214678.
Evangelista C, Bernabucci U, Basiricò L. Effect of antioxidant supplementation on milk yield and quality in italian mediterranean lactating buffaloes. Animals. 2022;12(15):1903. https://doi.org/10.3390/ani12151903.
Nwosu LC, Edo GI, Ozgor E. The phytochemical, proximate, pharmacological, GC-MS analysis of Cyperus esculentus (Tiger nut): a fully validated approach in health, food and nutrition. Food Biosci. 2022. https://doi.org/10.1016/j.fbio.2022.101551.
Tariq, M. Opportunities for Improving feed use efficiency for sustainable dairy production in Pakistan. In: The 1st International Electronic Conference on Animals—Global Sustainability and Animals: Science, Ethics and Policy. Basel Switzerland: MDPI. 2020. p. 11. https://doi.org/10.3390/IECA2020-08826
Onyibe PN, Edo GI, Nwosu LC, Ozgor E. Effects of vernonia amygdalina fractionate on glutathione reductase and glutathione-S-transferase on alloxan induced diabetes wistar rat. Biocatal Agric Biotechnol. 2021;36:102118. https://doi.org/10.1016/j.bcab.2021.102118.
Amr M, Mohie-Eldinn M, Farid A. Evaluation of buffalo, cow, goat and camel milk consumption on multiple health outcomes in male and female Sprague Dawley rats. Int Dairy J. 2023;146:105760. https://doi.org/10.1016/j.idairyj.2023.105760.
Ratajczak AE, Zawada A, Rychter AM, Dobrowolska A, Krela-Kaźmierczak I. Milk and dairy products: good or bad for human bone? practical dietary recommendations for the prevention and management of osteoporosis. Nutrients. 2021;13(4):1329. https://doi.org/10.3390/nu13041329.
Bakshi S, Paswan VK, Yadav SP, Bhinchhar BK, Kharkwal S, Rose H, Bunkar DS. A comprehensive review on infant formula: nutritional and functional constituents, recent trends in processing and its impact on infants’ gut microbiota. Front Nutr. 2023. https://doi.org/10.3389/fnut.2023.1194679.
Edo GI, Makinde MG, Nwosu LC, Ozgor E, Akhayere E. Physicochemical and Pharmacological properties of palm oil: an approach for quality, safety, and nutrition evaluation of palm oil. Food Anal Methods. 2022;15(8):2290–305. https://doi.org/10.1007/s12161-022-02293-4.
Edo GI. Antibacterial, phytochemical and GC-MS analysis of Thevetia peruviana extracts: an approach in drug formulation. Nat Resour Human Health. 2022;2(4):418–26. https://doi.org/10.53365/nrfhh/146543.
Chakraborty P, Singh T, Shivhare US, Basu S. Understanding the effect of milk composition and milking season on quality characteristics of chhana. J Texture Stud. 2021;52(1):45–56. https://doi.org/10.1111/jtxs.12558.
Gulzar N, Rafiq S, Nadeem M, Imran M, Khalique A, Muqada Sleem I, Saleem T. Influence of milling pH and storage on quality characteristics, mineral and fatty acid profile of buffalo Mozzarella cheese. Lipids Health Dis. 2019;18(1):33. https://doi.org/10.1186/s12944-019-0976-9.
Hassan F, Edo GI, Nwosu LC, Jalloh AA, Onyibe PN, Itoje-akpokiniovo LO, Irogbo PU. An inventory of medicinal plants used as sedative, analgesic and blood tonic in Abeokuta, Ogun State Nigeria. Acta Ecol Sin. 2021. https://doi.org/10.1016/j.chnaes.2021.11.003.
Pesce A, Salzano C, De Felice A, Garofalo F, Liguori S, De Santo A, Guarino A. Monitoring the freezing point of buffalo milk. Ital J Food Saf. 2016. https://doi.org/10.4081/ijfs.2016.5691.
Calahorrano-Moreno MB, Ordoñez-Bailon JJ, Baquerizo-Crespo RJ, Dueñas-Rivadeneira AA, Montenegro MCBSM, Rodríguez-Díaz JM. Contaminants in the cow’s milk we consume? Pasteurization and other technologies in the elimination of contaminants. F1000Research. 2022;11:91. https://doi.org/10.12688/f1000research.108779.1.
Akpoghelie PO, Edo GI, Akhayere E. Proximate and nutritional composition of beer produced from malted sorghum blended with yellow cassava. Biocatal Agric Biotechnol. 2022;45:102535. https://doi.org/10.1016/j.bcab.2022.102535.
Edo GI. Effects of paraquat dichloride on adult male wistar rat. an approach in the toxicity of body weights and hematological tissues. J Anal Pharm Res. 2022;11(1):1–7. https://doi.org/10.15406/japlr.2022.11.00394.
Melini F, Melini V, Luziatelli F, Ruzzi M. Raw and heat-treated milk: from public health risks to nutritional quality. Beverages. 2017;3(4):54. https://doi.org/10.3390/beverages3040054.
Erhonyota C, Edo GI, Onoharigho FO. Comparison of poison plate and agar well diffusion method determining the antifungal activity of protein fractions. Acta Ecol Sin. 2023;43(4):684–9. https://doi.org/10.1016/j.chnaes.2022.08.006.
Edo GI, Onoharigho FO, Akpoghelie PO, Emakpor OL, Ozgor E, Akhayere E. Physicochemical, phytochemical, antioxidant, and inhibition properties of key enzymes linked to raw and regular honey. Chem Afr. 2022. https://doi.org/10.1007/s42250-022-00401-9.
Li Y, Weng P, Wu Z, Liu Y. Extending the shelf life of raw milk and pasteurized milk with plantaricin FB-2. Foods. 2023;12(3):608. https://doi.org/10.3390/foods12030608.
Edo GI, Ugbune U, Onoharigho FO, Ezekiel GO, Agbo JJ. Antioxidant activities of reissantia indica willd. (mopane paddle-pod) and nephroprotective effect on paracetamol-induced nephrotoxicity in male wistar rats. Nutrire. 2023;48(1):26. https://doi.org/10.1186/s41110-023-00214-x.
Ogheneoruese Onoharigho F, Ahuose Ighede P, Othuke Akpoghelie P, Oghenekome Akpoghelie E, Edo GI. Isolation and identification of bacterial and fungal spoilage organisms in branded and unbranded milk; consumer perception of safety hazard for milk. Appl Microbiol Theory Technol. 2022. https://doi.org/10.37256/amtt.3220221766.
Alothman M, Bremer PJ, Lusk K, Silcock P. When does milk spoil? The use of rejection threshold methodology to investigate the influence of total microbial numbers on the acceptability of fresh chilled pasteurised milk. Beverages. 2023;9(2):53. https://doi.org/10.3390/beverages9020053.
Edo GI, Nwosu LC. Association of alcohol use and dietary lifestyle of commercial drivers during the COVID-19 pandemic in Nigeria. Bull Natl Res Cent. 2022;46(1):197. https://doi.org/10.1186/s42269-022-00885-y.
Juvinal JG, De Steur H, Schouteten JJ, Muhammad DRA, De Leon AA, Dewettinck K, Gellynck X. Physico-chemical property, sensory profile and consumer acceptability of water buffalo (Bubalus bubalis L.) chocolate milk using alkalized and natural cocoa powder. Foods. 2023;12(9):1797. https://doi.org/10.3390/foods12091797.
Becskei Z, Savić M, Ćirković D, Rašeta M, Puvača N, Pajić M, Paskaš S. Assessment of water buffalo milk and traditional milk products in a sustainable production system. Sustainability. 2020;12(16):6616. https://doi.org/10.3390/su12166616.
Yuliana T, Tyano FN, Harlina PW, Cahyana Y, Marta H, Krama A. Characterizing probiotic lactic acid bacteria from buffalo milk fermentation (Dadih) for beef Biopreservation. Appl Sci. 2023;13(24):13089. https://doi.org/10.3390/app132413089.
Abdullah D, Poddar S, Rai RP, Purwati E, Dewi NP, Pratama YE. Molecular identification of lactic acid bacteria an approach to sustainable food security. J Publ Health Res. 2021;10(2_suppl):jphr.2021.2508. https://doi.org/10.4081/jphr.2021.2508.
Edo GI, Samuel PO, Ossai S, Nwachukwu SC, Okolie MC, Oghenegueke O, Agbo JJ. Phytochemistry and pharmacological compounds present in scent leaf: a review. Food Chem Adv. 2023;3:100300. https://doi.org/10.1016/j.focha.2023.100300.
Yasmin I, Iqbal R, Liaqat A, Khan WA, Nadeem M, Iqbal A, Khaliq A. Characterization and comparative evaluation of milk protein variants from Pakistani dairy breeds. Food Sci Animal Resour. 2020;40(5):689–98. https://doi.org/10.5851/kosfa.2020.e44.
Owheruo JO, Edo GI, Oluwajuyitan DT, Faturoti AO, Martins IE, Akpoghelie PO, Agbo JJ. Quality evaluation of value-added nutritious biscuit with high antidiabetic properties from blends of wheat flour and oyster mushroom. Food Chem Adv. 2023;3:100375. https://doi.org/10.1016/j.focha.2023.100375.
Jikah AN, Edo GI. Moringa oleifera: a valuable insight into recent advances in medicinal uses and pharmacological activities. J Sci Food Agric. 2023. https://doi.org/10.1002/jsfa.12892.
Mahadev GM, Meena GS. Milk protein concentrates 80: does composition of buffalo milk matter for its poor functionality? LWT. 2020;131:109652. https://doi.org/10.1016/j.lwt.2020.109652.
Singh A, Zapata RC, Pezeshki A, Knight CG, Tuor UI, Chelikani PK. Whey protein and its components lactalbumin and lactoferrin affect energy balance and protect against stroke onset and renal damage in salt-loaded, high-fat fed male spontaneously hypertensive stroke-prone rats. J Nutr. 2020;150(4):763–74. https://doi.org/10.1093/jn/nxz312.
Minj S, Anand S. Whey proteins and its derivatives: bioactivity, functionality, and current applications. Dairy. 2020;1(3):233–58. https://doi.org/10.3390/dairy1030016.
Runthala A, Mbye M, Ayyash M, Xu Y, Kamal-Eldin A. Caseins: versatility of their micellar organization in relation to the functional and nutritional properties of milk. Molecules. 2023;28(5):2023. https://doi.org/10.3390/molecules28052023.
Owheruo JO, Akpoghelie PO, Edo GI, Ojulari AE, Agbo JJ. Proximate, mineral, sensorial and microbiological properties of chin-chin produced from okra seed and wheat flour blends. Food Chem Adv. 2023;2:100298. https://doi.org/10.1016/j.focha.2023.100298.
Zebib H, Abate D, Woldegiorgis AZ. Nutritional quality and adulterants of cow raw milk, pasteurized and cottage cheese collected along value chain from three regions of Ethiopia. Heliyon. 2023;9(5): e15922. https://doi.org/10.1016/j.heliyon.2023.e15922.
Pietrzak-Fiećko R, Kamelska-Sadowska AM. The comparison of nutritional value of human milk with other mammals’ milk. Nutrients. 2020;12(5):1404. https://doi.org/10.3390/nu12051404.
Ménard O, Ahmad S, Rousseau F, Briard-Bion V, Gaucheron F, Lopez C. Buffalo vs. cow milk fat globules: Size distribution, zeta-potential, compositions in total fatty acids and in polar lipids from the milk fat globule membrane. Food Chem. 2010;120(2):544–51. https://doi.org/10.1016/j.foodchem.2009.10.053.
Pegolo S, Stocco G, Mele M, Schiavon S, Bittante G, Cecchinato A. Factors affecting variations in the detailed fatty acid profile of Mediterranean buffalo milk determined by 2-dimensional gas chromatography. J Dairy Sci. 2017;100(4):2564–76. https://doi.org/10.3168/jds.2016-11696.
Bakry IA, Yang L, Farag MA, Korma SA, Khalifa I, Cacciotti I, Wang X. A comprehensive review of the composition, nutritional value, and functional properties of camel milk fat. Foods. 2021;10(9):2158. https://doi.org/10.3390/foods10092158.
Edo GI, Ugbune U, Onoharigho FO, Ezekiel GO, Ugbuwe E, Agbo JJ. Investigation of the metal complexes and bioactive compound formed by coordination of bioactive phytochemical from ginger (Zingiber officinale) extracts to metal ions. Food Chem Adv. 2023;3:100337. https://doi.org/10.1016/j.focha.2023.100337.
Lindmark Månsson H. Fatty acids in bovine milk fat. Food Nutr Res. 2008;52(1):1821. https://doi.org/10.3402/fnr.v52i0.1821.
Iruoghene Edo G. Analysis of phytochemical constituents and antioxidant potential of bitter kola leaf extract towards bioactive food, nutrition and health resources. Org Med Chem Int J. 2022. https://doi.org/10.19080/OMCIJ.2022.11.555823.
Pasaoglulari Aydinlik N, Abubakar J, Edo G. Phytochemical and GCMS analysis on the ethanol extract of Foeniculum Vulgare and Petroselinum crispum leaves. Int J Chem Technol. 2021. https://doi.org/10.32571/ijct.911711.
Asif A, Sarker M, Deb G, Habib M, Arefin S, Bari M, Islam M. Fatty acid and amino acid profiles of cheese, butter, and ghee made from buffalo milk. J Adv Vet Animal Res. 2022;9(1):144. https://doi.org/10.5455/javar.2022.i579.
Owheruo JO, Edo GI, Bashir ZA, Akpoghelie PO, Agbo JJ. Quality evaluation of breakfast cereal meal produced from finger millet (Eleusine coracana) and Roasted African yam beans (Sphenostylis stenocarpa) flour blends. Food Sci Eng. 2023. https://doi.org/10.37256/fse.4220232328.
Park J-E, Kim T-E, Shin K-H. Quantitative analysis of four catechins from green tea extract in human plasma using ultra-performance liquid chromatography-tandem mass spectrometry for pharmacokinetic studies. Molecules. 2018;23(4):984. https://doi.org/10.3390/molecules23040984.
Manik S, Meena GS, Singh AK, Khetra Y, Singh R, Arora S, Vishweswaraiah RH. Valorization of sour buttermilk (a potential waste stream): conversion to powder employing reverse osmosis and spray drying. Membranes. 2023;13(9):799. https://doi.org/10.3390/membranes13090799.
Edo GI, Samuel PO, Jikah AN, Oloni GO, Ifejika MN, Oghenegueke O, Essaghah AEA. Proximate composition and health benefit of Roselle leaf (Hibiscus sabdariffa). Insight on food and health benefits. Food Chem Adv. 2023;3:100437. https://doi.org/10.1016/j.focha.2023.100437.
Edo GI, Ugbune U, Ezekiel GO, Onoharigho FO, Agbo JJ. Cyperus esculentus (tiger nut): its application in agriculture, food, health and nutrition. A review. Vegetos. 2023. https://doi.org/10.1007/s42535-023-00672-8.
Kapadiya DB, Prajapati DB, Jain AK, Mehta BM, Darji VB, Aparnathi KD. Comparison of Surti goat milk with cow and buffalo milk for gross composition, nitrogen distribution, and selected minerals content. Vet World. 2016;9(7):710–6. https://doi.org/10.14202/vetworld.2016.710-716.
Landi N, Ragucci S, Di Maro A. Amino acid composition of milk from cow, sheep and goat raised in ailano and valle agricola, two localities of ‘Alto Casertano’ (Campania Region). Foods. 2021;10(10):2431. https://doi.org/10.3390/foods10102431.
Romero-Velarde E, Delgado-Franco D, García-Gutiérrez M, Gurrola-Díaz C, Larrosa-Haro A, Montijo-Barrios E, Geurts J. The importance of lactose in the human diet: outcomes of a Mexican consensus meeting. Nutrients. 2019;11(11):2737. https://doi.org/10.3390/nu11112737.
Szilagyi A, Ishayek N. Lactose intolerance, dairy avoidance, and treatment options. Nutrients. 2018;10(12):1994. https://doi.org/10.3390/nu10121994.
Kandhro F, Kazi TG, Afridi HI, Baig JA. Compare the nutritional status of essential minerals in milk of different cattle and humans: estimated daily intake for children. J Food Compos Anal. 2022;105:104214. https://doi.org/10.1016/j.jfca.2021.104214.
Woźniak D, Cichy W, Dobrzyńska M, Przysławski J, Drzymała-Czyż S. Reasonableness of enriching cow’s milk with vitamins and minerals. Foods. 2022;11(8):1079. https://doi.org/10.3390/foods11081079.
Garau V, Manis C, Scano P, Caboni P. Compositional characteristics of mediterranean buffalo milk and whey. Dairy. 2021;2(3):469–88. https://doi.org/10.3390/dairy2030038.
Almeida CC, dos Baião DS, de Rodrigues PA, Saint’Pierre TD, Hauser-Davis RA, Leandro KC, Conte-Junior CA. Macrominerals and trace minerals in commercial infant formulas marketed in brazil: compliance with established minimum and maximum requirements, label statements, and estimated daily intake. Front Nutr. 2022. https://doi.org/10.3389/fnut.2022.857698.
Mandiá N, Bermejo-Barrera P, Herbello P, López-Suárez O, Fraga JM, Fernández-Pérez C, Couce ML. Human milk concentrations of minerals, essential and toxic trace elements and association with selective medical, social. Demogr Environ Fact Nutr. 2021;13(6):1885. https://doi.org/10.3390/nu13061885.
Samuel PO, Edo GI, Emakpor OL, Oloni GO, Ezekiel GO, Essaghah AEA, Agbo JJ. Lifestyle modifications for preventing and managing cardiovascular diseases. Sport Sci Health. 2023. https://doi.org/10.1007/s11332-023-01118-z.
Fantuz F, Todini L, Ferraro S, Fatica A, Marcantoni F, Zannotti M, Salimei E. Macro minerals and trace elements in milk of dairy buffaloes and cows reared in mediterranean areas. Beverages. 2022;8(3):51. https://doi.org/10.3390/beverages8030051.
Edo GI, Samuel PO, Jikah AN, Onoharigho FO, Idu LI, Obasohan P, Owigho JE. Biological and bioactive components of bitter leaf (Vernonia amygdalina leaf): insight on health and nutritional benefits. A review. Food Chem Adv. 2023;3:100488. https://doi.org/10.1016/j.focha.2023.100488.
Tardy A-L, Pouteau E, Marquez D, Yilmaz C, Scholey A. Vitamins and minerals for energy, fatigue and cognition: a narrative review of the biochemical and clinical evidence. Nutrients. 2020;12(1):228. https://doi.org/10.3390/nu12010228.
Edo GI, Ugbune U, Akpoghelie PO, Owheruo JO. Evaluation of physicochemical, phytochemical, anti-bacterial and antioxidant potential of kola nut (cola acuminata): an approach in food, health and nutritional benefits. Vegetos. 2023. https://doi.org/10.1007/s42535-023-00715-0.
Górska-Warsewicz H, Rejman K, Laskowski W, Czeczotko M. Milk and dairy products and their nutritional contribution to the average polish diet. Nutrients. 2019;11(8):1771. https://doi.org/10.3390/nu11081771.
Yeh EB, Barbano DM, Drake M. Vitamin fortification of fluid milk. J Food Sci. 2017;82(4):856–64. https://doi.org/10.1111/1750-3841.13648.
Khan IT, Nadeem M, Imran M, Ayaz M, Ajmal M, Ellahi MY, Khalique A. Antioxidant capacity and fatty acids characterization of heat treated cow and buffalo milk. Lipids Health Dis. 2017;16(1):163. https://doi.org/10.1186/s12944-017-0553-z.
Edo GI, Onoharigho FO. A comparative study of biological and chemical composition of African spinach (Amaranthus cruentus) extract: an approach in drug formulation, food and nutrition. Adv Tradit Med. 2023. https://doi.org/10.1007/s13596-023-00718-x.
Vincenzetti S, Santini G, Polzonetti V, Pucciarelli S, Klimanova Y, Polidori P. Vitamins in human and donkey milk: functional and nutritional role. Nutrients. 2021;13(5):1509. https://doi.org/10.3390/nu13051509.
Edo GI, Ugbune U, Ezekiel GO, Nwosu LC, Onoharigho FO, Agbo JJ. Medicinal plants used for the treatment of sexual dysfunction; ethnobotanical study and phytochemical analysis. Acta Ecol Sin. 2023. https://doi.org/10.1016/j.chnaes.2023.05.008.
Sørensen HM, Rochfort KD, Maye S, MacLeod G, Loscher C, Brabazon D, Freeland B. Bioactive ingredients from dairy-based lactic acid bacterial fermentations for functional food production and their health effects. Nutrients. 2023;15(22):4754. https://doi.org/10.3390/nu15224754.
Labarrere CA, Kassab GS. Glutathione: a Samsonian life-sustaining small molecule that protects against oxidative stress, ageing and damaging inflammation. Front Nutr. 2022. https://doi.org/10.3389/fnut.2022.1007816.
Bhatti JS, Sehrawat A, Mishra J, Sidhu IS, Navik U, Khullar N, Reddy PH. Oxidative stress in the pathophysiology of type 2 diabetes and related complications: current therapeutics strategies and future perspectives. Free Radic Biol Med. 2022;184:114–34. https://doi.org/10.1016/j.freeradbiomed.2022.03.019.
Giri B, Dey S, Das T, Sarkar M, Banerjee J, Dash SK. Chronic hyperglycemia mediated physiological alteration and metabolic distortion leads to organ dysfunction, infection, cancer progression and other pathophysiological consequences: an update on glucose toxicity. Biomed Pharmacother. 2018;107:306–28. https://doi.org/10.1016/j.biopha.2018.07.157.
Lee H, Padhi E, Hasegawa Y, Larke J, Parenti M, Wang A, Slupsky C. Compositional dynamics of the milk fat globule and its role in infant development. Front Pediatr. 2018. https://doi.org/10.3389/fped.2018.00313.
Yan F, Yang W, Li X, Lin T, Lun Y, Lin F, Luo G. A trifunctional enzyme with glutathione S-transferase, glutathione peroxidase and superoxide dismutase activity. Biochim Biophys Acta (BBA) Gen Subj. 2008;1780(6):869–72. https://doi.org/10.1016/j.bbagen.2008.03.003.
Jalloh AA, Edo GI, Onyibe PN, Nwosu LC. Farmers’ perception of climate change: a case study from subsistence farming in lalehun kovama of Kailahun in Eastern Sierra Leone. Environ Protection Res. 2023. https://doi.org/10.37256/epr.3120231905.
Koksal Z, Gulcin I, Ozdemir H. An important milk enzyme: lactoperoxidase. Milk Proteins Struct Biol Prop Health Asp. 2016. https://doi.org/10.5772/64416.
Edo GI, Onoharigho FO, Emakpor GL, Akpoghelie PO. The physicochemical analysis and health benefits of fresh and branded honey produced in delta state Nigeria. J Anal Pharm Res. 2022;11(2):66–72. https://doi.org/10.15406/japlr.2022.11.00403.
Deeth HC. Effects of high-temperature milk processing. Encyclopedia. 2021;1(4):1312–21. https://doi.org/10.3390/encyclopedia1040098.
Edo GI, Onoharigho FO, Jikah AN, Oloni GO, Samuel PO, Rapheal OA, Akpoghelie EO. Cyperus esculentus (tiger nut): an insight into its bioactive compounds, biological activities, nutritional and health benefits. Food Chem Adv. 2023;3:100511. https://doi.org/10.1016/j.focha.2023.100511.
Samuel PO, Edo GI, Oloni GO, Ugbune U, Ezekiel GO, Essaghah AEA, Agbo JJ. Effects of chemical contaminants on the ecology and evolution of organisms a review. Chem Ecol. 2023. https://doi.org/10.1080/02757540.2023.2284158.
Bharadwaj AG, Holloway RW, Miller VA, Waisman DM. Plasmin and plasminogen system in the tumor microenvironment: implications for cancer diagnosis, prognosis, and therapy. Cancers. 2021;13(8):1838. https://doi.org/10.3390/cancers13081838.
Jikah AN, Edo GI. Mechanisms of action by sulphur compounds in Allium sativum. A review. Pharmacol Res Mod Chin Med. 2023;9:100323. https://doi.org/10.1016/j.prmcm.2023.100323.
Ogheneoruese Onoharigho F, Oyinegberi Akpodimo E, Edo GI. The effect of uncontrolled dumping of solid waste on groundwater in Osun State, Nigeria. Fine Chem Eng. 2022. https://doi.org/10.37256/fce.3220221668.
Edo GI, Onoharigho FO, Jikah AN, Ezekiel GO, Essaghah AEA, Ekokotu HA, Owheruo JO. Evaluation of the physicochemical, phytochemical and anti-bacterial potential of Zingiber officinale (ginger). Food Chem Adv. 2024;4:100625. https://doi.org/10.1016/j.focha.2024.100625.
Bouroutzika E, Proikakis S, Anagnostopoulos AK, Katsafadou AI, Fthenakis GC, Tsangaris GT. Proteomics analysis in dairy products: cheese, a review. Appl Sci. 2021;11(16):7622. https://doi.org/10.3390/app11167622.
Edo GI, Jikah AN, Onoharigho FO, Akpoghelie PO, Agbo JJ, Ekokotu HA, Owheruo JO. The ameliorative effects of Vernonia amygdalina extract on superoxide dismutase and glutathione s-transferase on alloxan induced diabetes on male Wistar rats. Food Chem Adv. 2024;4:100620. https://doi.org/10.1016/j.focha.2024.100620.
Egbune EO, Anigboro AA, Edeche G, Edo GI, Onoharigho FO, Emakpor OL, Tonukari NJ. Effect of inoculum size on solid state fermentation of cassava (Manito esculenta Crantz). Chem Afr. 2022. https://doi.org/10.1007/s42250-022-00434-0.
Almeida CC, Mendonça Pereira BF, Leandro KC, Costa MP, Spisso BF, Conte-Junior CA. Bioactive compounds in infant formula and their effects on infant nutrition and health: a systematic literature review. Int J Food Sci. 2021;2021:1–31. https://doi.org/10.1155/2021/8850080.
Ryan RYM, Seymour J, Loukas A, Lopez JA, Ikonomopoulou MP, Miles JJ. Immunological responses to envenomation. Front Immunol. 2021. https://doi.org/10.3389/fimmu.2021.661082.
Lis-Kuberka J, Berghausen-Mazur M, Orczyk-Pawiłowicz M. Lactoferrin and immunoglobulin concentrations in milk of gestational diabetic mothers. Nutrients. 2021;13(3):818. https://doi.org/10.3390/nu13030818.
Owheruo JO, Edo GI, Ifesan BO, Bolade MK, Origbemisoye BA, Akpoghelie PO, Agbo JJ. Evaluation of nutraceutical property of extruded breakfast cereal produced from blends of malted finger millet (Eleusine coracana) and watermelon (Citrullus lanatus) seed flour. Vegetos. 2023. https://doi.org/10.1007/s42535-023-00728-9.
Kuraku S, Kaiya H, Tanaka T, Hyodo S. Evolution of vertebrate hormones and their receptors: insights from non-osteichthyan genomes. Ann Rev Animal Biosci. 2023;11(1):163–82. https://doi.org/10.1146/annurev-animal-050922-071351.
Edo GI. The German energy system: analysis of past, present, and future developments. Adv Energy Convers Mater. 2023. https://doi.org/10.37256/aecm.4120232207.
Tatiana Z. Gonadal sex steroids: production, action and interactions in mammals. Steroids Physiol Clin Med. 2012. https://doi.org/10.5772/52994.
Castellone V, Bancalari E, Rubert J, Gatti M, Neviani E, Bottari B. Eating fermented: health benefits of LAB-fermented foods. Foods. 2021;10(11):2639. https://doi.org/10.3390/foods10112639.
Edo GI, Samuel PO, Nwachukwu SC. Bioactive compounds and biological activities of tiger nut (Cyperus esculentus L.). Berlin: Springer; 2023. p. 1–28. https://doi.org/10.1007/978-3-031-29006-0_34-1.
Samtiya M, Samtiya S, Badgujar PC, Puniya AK, Dhewa T, Aluko RE. Health-promoting and therapeutic attributes of milk-derived bioactive peptides. Nutrients. 2022;14(15):3001. https://doi.org/10.3390/nu14153001.
Williams GM, Tapsell LC, Beck EJ. Gut health, the microbiome and dietary choices: an exploration of consumer perspectives. Nutr Diet. 2023;80(1):85–94. https://doi.org/10.1111/1747-0080.12769.
Agbo JJ, Onayemi TO, Edo GI. The effect of COVID-19 pandemic on the academic performance of nursing students. Soc Educ Res. 2022. https://doi.org/10.37256/ser.4120232043.
Ali MA, Kamal MM, Rahman MH, Siddiqui MN, Haque MA, Saha KK, Rahman MA. Functional dairy products as a source of bioactive peptides and probiotics: current trends and future prospectives. J Food Sci Technol. 2022;59(4):1263–79. https://doi.org/10.1007/s13197-021-05091-8.
Abesinghe AMNL, Priyashantha H, Prasanna PHP, Kurukulasuriya MS, Ranadheera CS, Vidanarachchi JK. Inclusion of probiotics into fermented buffalo (Bubalus bubalis) milk: an overview of challenges and opportunities. Fermentation. 2020;6(4):121. https://doi.org/10.3390/fermentation6040121.
Simões da Silva TM, Piazentin ACM, Mendonça CMN, Converti A, Bogsan CSB, Mora D, de Souza Oliveira RP. Buffalo milk increases viability and resistance of probiotic bacteria in dairy beverages under in vitro simulated gastrointestinal conditions. J Dairy Sci. 2020;103(9):7890–7. https://doi.org/10.3168/jds.2019-18078.
Wang S, Naumovski N, Ajlouni S, Ayyash M, Silva R, Balthazar CF, Ranadheera CS. Nonbovine milk and its products as sources of probiotics delivery: An overview of its viability, functionality and product quality characteristics. Int J Dairy Technol. 2023;76(3):482–511. https://doi.org/10.1111/1471-0307.12976.
Yang TX, Li H, Wang F, Liu XL, Li QY. Effect of cattle breeds on milk composition and technological characteristics in China. Asian Australas J Anim Sci. 2013;26(6):896–904. https://doi.org/10.5713/ajas.2012.12677.
Nwachukwu SC, Edo GI, Jikah AN, Emakpor OL, Akpoghelie PO, Agbo JJ. Recent advances in the role of mass spectrometry in the analysis of food: a review. J Food Meas Charact. 2024. https://doi.org/10.1007/s11694-024-02492-z.
Nayik GA, Jagdale YD, Gaikwad SA, Devkatte AN, Dar AH, Ansari MJ. Nutritional profile, processing and potential products: a comparative review of goat milk. Dairy. 2022;3(3):622–47. https://doi.org/10.3390/dairy3030044.
Jikah AN, Edo GI. Moringa oleifera: its industrial and pharmaceutical applications. A review. Vegetos. 2024. https://doi.org/10.1007/s42535-024-00866-8.
Edo GI, Samuel PO, Jikah AN, Ekokotu HA, Ugbune U, Oghroro EEA, Owheruo JO. Petroleum discovery, utilization and processing in the World and Nigeria: a comprehensive literature review. Sustain Chem Eng. 2024. https://doi.org/10.37256/sce.5120243970.
Kumari K, Chakraborty SK, Sudhakar A, Kishore A. Dielectric spectroscopy-based characterisation of different types of paneer (Indian cottage cheese) in terms of texture, microstructure and functional groups. Int J Dairy Technol. 2023;76(1):4–14. https://doi.org/10.1111/1471-0307.12913.
Edo GI, Samuel PO, Oloni GO, Ezekiel GO, Ikpekoro VO, Obasohan P, Agbo JJ. Environmental persistence, bioaccumulation, and ecotoxicology of heavy metals. Chem Ecol. 2024. https://doi.org/10.1080/02757540.2024.2306839.
Akpoghelie EO, Ishioro BO, Edo GI. Effects of energy consumption on human development and industrial sector performance in selected Sub-Saharan Africa and OECD countries: comparative analysis. Int J Sustain Dev World Ecol. 2024. https://doi.org/10.1080/13504509.2023.2301373.
Michael P, de Cruz CR, Mohd Nor N, Jamli S, Goh YM. The potential of using temperate-tropical crossbreds and agricultural by-products, associated with heat stress management for dairy production in the tropics: a review. Animals. 2021;12(1):1. https://doi.org/10.3390/ani12010001.
Dias K, Garcia S, (Rafiq) Islam M, Clark C. Milk yield, milk composition, and the nutritive value of feed accessed varies with milking order for pasture-based dairy cattle. Animals. 2019;9(2):60. https://doi.org/10.3390/ani9020060.
Bodke H, Jogdand S. Role of Probiotics in Human Health. Cureus. 2022. https://doi.org/10.7759/cureus.31313.
You S, Ma Y, Yan B, Pei W, Wu Q, Ding C, Huang C. The promotion mechanism of prebiotics for probiotics: a review. Front Nutr. 2022. https://doi.org/10.3389/fnut.2022.1000517.
Mehaya FM, El-Shazly AI, El-Dein AN, Farid MA. Evaluation of nutritional and physicochemical characteristics of soy yogurt by Lactobacillus plantarum KU985432 and Saccharomyces boulardii CNCMI-745. Sci Rep. 2023;13(1):13026. https://doi.org/10.1038/s41598-023-40207-4.
Mohd Azmi AF, Ahmad H, Mohd Nor N, Goh Y-M, Zamri-Saad M, Abu Bakar MZ, Abu Hassim H. The impact of feed supplementations on Asian buffaloes: a review. Animals. 2021;11(7):2033. https://doi.org/10.3390/ani11072033.
Rizzoli R. Dairy products and bone health. Aging Clin Exp Res. 2021;34(1):9–24. https://doi.org/10.1007/s40520-021-01970-4.
Chirone R, Paulillo A, Salatino P, Salzano A, Cristofaro B, Cristiano T, Neglia G. Life cycle assessment of buffalo milk: a case study of three farms in southern Italy. J Clean Prod. 2022;365:132816. https://doi.org/10.1016/j.jclepro.2022.132816.
Acknowledgements
We will like to express our profound gratitude to Maureen Marris Dinzei (dinzeimaureen@gmail.com) of the Department of Educational Administration (English) Faculty of Education, Delta State University, Abraka who took her time to proofread and edit this manuscript before it was published.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
OE, GE, AJ, VI, JA, IA, AE, HE, EO, PA were responsible for the conception and design of the study; OE, GE, AJ performed data collection. OE, GE, AJ performed data analysis and drafted the article. GE supervised the study, contributed to data analysis, interpretation, and critical revisions. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Emakpor, O.L., Edo, G.I., Jikah, A.N. et al. Buffalo milk: an essential natural adjuvant. Discov Food 4, 38 (2024). https://doi.org/10.1007/s44187-024-00114-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44187-024-00114-7