Abstract
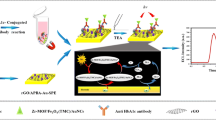
Multiplex detection of biomarkers in clinical matrices on a single sensor platform is the need for a detailed disease diagnosis and patient prognosis. Herein, we demonstrate immunoassay/enzyme detection assay formats on a 3D-printed device for measuring insulin, C peptide, and glucose in two-fold diluted human serum electrochemically. The sensor platform comprised of a graphene–gold (Gr–Au) framework to immobilize insulin and C peptide specific antibodies and glucose oxidase to detect clinically relevant levels of diabetes biomarkers with good sensitivity, specificity, and reproducibility. The fabrication of the 3D-printed sensor platform is characterized by field emission scanning electron microscopy (FESEM) and electron impedance spectroscopy (EIS). The current signals for detection of free serum insulin, C peptide were monitored by oxidation of ferro/ferricyanide redox probe, while glucose was detected directly using square wave voltammetry (drop-based) and amperometry (real-time). The designed electrochemical platform offered a limit of detection (LOD) 4 pM insulin, 0.02 nM C peptide, and 0.05 mM glucose. The Gr–Au framework was ~ 2.5× more sensitive in detecting diabetes biomarkers in human serum when compared to pristine Gr framework. Additionally, the sensor does not show any cross-reactivity to non-specific biomarkers in human serum.








Similar content being viewed by others
References
World Health Organization (WHO). WHO Diabetes Programme. http://www.who.int/diabetes/en/. Accessed 1 Apr 2022
G. Freckmann, S. Hagenlocher, A. Baumstark, N. Jendrike, R.C. Gillen, K. Rössner, C. Haug, Continuous glucose profiles in healthy subjects under everyday life conditions and after different meals. J. Diabetes Sci. Technol. 1, 695–703 (2007)
D.M. Muoio, C.B. Newgard, Mechanisms of disease: molecular and metabolic mechanisms of insulin resistance and beta-cell failure in type 2 diabetes. Nat. Rev. Mol. Cell Biol. 9, 193–205 (2008)
C. Weyer, R.L. Hanson, P.A. Tataranni, C. Bogardus, R.E. Pratley, A high fasting plasma insulin concentration predicts type 2 diabetes independent of insulin resistance: evidence for a pathogenic role of relative hyperinsulinemia. Diabetes 49, 2094–2101 (2000)
F.C. Goetz, L.R. French, W. Thomas, R.L. Gingerich, J.P. Clements, Are specific serum insulin levels low in impaired glucose tolerance and type II diabetes?: measurement with a radioimmunoassay blind to proinsulin, in the population of Wadena, Minnesota. Metabolism 44, 1371–1376 (1995)
K.C. Ronald, G.C. Weir, Joslin’s Diabetes Mellitus, fourteenth. (Lippincott Williams & Wilkins, Philadelphia, 2005)
D.F. Steiner, P.E. Oyer, The biosynthesis of insulin and a probable precursor of insulin by a human islet cell adenoma. Proc. Natl. Acad. Sci. USA 57, 473–480 (1967)
M. Liu, J. Wright, H. Guo, Y. Xiong, P. Arvan, Proinsulin entry and transit through the endoplasmic reticulum in pancreatic beta cells. Vitam. Horm. 95, 35–62 (2014)
G.L.C. Yosten, C. Maric-Bilkan, P. Luppi, J. Wahren, Physiological effects and therapeutic potential of proinsulin C-peptide. Am. J. Physiol. Endocrinol. Metab. 307, E955–E968 (2014)
K.W. Cheng, A radioreceptor assay for follicle-stimulating hormone. J. Clin. Endocrinol. Metab. 41, 581–589 (1975)
L. Andersen, B. Dinesen, P.N. Jorgensen, F. Poulsen, M.E. Roder, Enzyme immunoassay for intact human insulin in serum or plasma. Clin. Chem. 39, 578–582 (1993)
S.E. Manley, I.M. Stratton, P.M. Clark, S.D. Luzio, Comparison of 11 human insulin assays: implications for clinical investigation and research. Clin. Chem. 53, 922–932 (2007)
P.M. Clark, Assays for insulin, proinsulin(s) and C-peptide. Ann. Clin. Biochem. 36, 541–564 (1999)
J.P. Ashby, B.M. Frier, Circulating C peptide: measurement and clinical application. Ann. Clin. Biochem. 18, 125–130 (1981)
P. Koskinen, Non-transferability of C-peptide measurements with various commercial radioimmunoassay reagents. Clin. Chem. 34, 1575–1578 (1988)
J.P. Palmer, G.A. Fleming, C.J. Greenbaum, K.C. Herold, L.D. Jansa, H. Kolb, J.H. Lachin, K.S. Polonsk, P. Pozzilli, J.S. Skyler, M.W. Steffes, C-peptide is the appropriate outcome measure for type 1 diabetes clinical trials to preserve beta-cell function: report of an ADA workshop, 21–22 October 2001. Diabetes 53, 250–264 (2004)
V. Singh, S. Krishnan, Electrochemical mass sensor for diagnosing diabetes in human serum. Analyst 139, 724–728 (2014)
V. Singh, S. Krishnan, Voltammetric immunosensor assembled on carbon-pyrenyl nanostructures for clinical diagnosis of type of diabetes. Anal. Chem. 87, 2648–2654 (2015)
V. Singh, Quantum dot decorated multi-walled carbon nanotube modified electrochemical sensor array for single drop insulin detection. Mater. Lett. 254, 415–418 (2019)
W. Yang, K.R. Ratinac, S.P. Ringer, P. Thordarson, J.J. Gooding, F. Braet, Carbon nanomaterials in biosensors: should you use nanotubes or graphene? Angew. Chem. Int. Ed. 49, 2114–2138 (2010)
Y. Si, E.T. Samulski, Exfoliated graphene separated by platinum nanoparticles. Chem. Mater. 20, 6792–6797 (2008)
H.-W. Tien, Y.-L. Huang, S.-Y. Yang, J.-Y. Wang, C.-C.M. Ma, The production of graphene nanosheets decorated with silver nanoparticles for use in transparent conductive films. Carbon 49, 1550–1560 (2011)
B. Gross, S.Y. Lockwood, D.M. Spence, Recent advances in analytical chemistry by 3D printing. Anal. Chem. 89, 57–70 (2017)
H.N. Chia, B.M. Wu, Recent advances in 3D printing of biomaterials. J. Biol. Eng. 9, 4 (2015)
S. Bose, S. Vahabzadeh, A. Bandyopadhyay, Bone tissue engineering using 3D printing. Mater. Today. 16, 496–504 (2013)
Why 3D printing could be a manufacturing and logistics game changer. https://www.manufacturing.net/blog/2013/10/why-3d-printing-could-be-manufacturing-and-logistics-game-changer. Accessed 30 Apr 2018
J.V. Crivello, E. Reichmanis, Photopolymer materials and processes for advanced technologies. Chem. Mater. 26, 533–548 (2014)
N.P. Macdonald, J.M. Cabot, P. Smejkal, R.M. Guijt, B. Paull, M.C. Breadmore, Comparing microfluidic performance of three-dimensional (3D) printing platforms. Anal. Chem. 89, 3858–3866 (2017)
C. Tang, A. Vaze, J.F. Rusling, Automated 3D-printed unibody immunoarray for chemiluminescence detection of cancer biomarker proteins. Lab Chip 17, 484–489 (2017)
K. Kadimisetty, S. Malla, J.F. Rusling, Automated 3D-printed arrays to evaluate genotoxic chemistry: e-cigarettes and water samples. ACS Sens. 2, 670–678 (2017)
B.V. Chikkaveeraiah, A.A. Bhirde, N.Y. Morgan, H.S. Eden, X. Chen, Electrochemical immunosensors for detection of cancer protein biomarkers. ACS Nano 6, 6546–6561 (2012)
M.-I. Mohammed, M.P.Y. Desmulliez, Lab-on-a-chip based immunosensor principles and technologies for the detection of cardiac biomarkers: a review. Lab Chip 11, 569–595 (2011)
B. Li, H. Tan, D. Jenkins, V.S. Raghavan, B.G. Rosa, F. Güder, G. Pan, E. Yeatman, D.J. Sharp, Clinical detection of neurodegenerative blood biomarkers using graphene immunosensor. Carbon 168, 144–162 (2020)
V. Singh, 3D-printed device for synthesis of magnetic and metallic nanoparticles. J. Flow Chem. 11, 135–142 (2021)
M. Zhang, C. Mullens, W. Gorski, Insulin oxidation and determination at carbon electrodes. Anal. Chem. 77, 6396–6401 (2005)
K.-J. Huang, D.-F. Luo, W.-Z. Xie, Y.-S. Yu, Sensitive voltammetric determination of tyrosine using multi-walled carbon nanotubes/4-aminobenzeresulfonic acid film-coated glassy carbon electrode. Colloids Surf. B 61, 176–181 (2008)
C. Li, Voltammetric determination of tyrosine based on an l-serine polymer film electrode. Colloids Surf. B 50, 147–151 (2006)
Acknowledgements
The author is grateful to the Department of Science and Technology (DST), Government of India for the financial support under the Women Scientist Scheme (Project Number: SR/WOS-A/ET-46/2018) to carry out this work. The author gratefully acknowledges Dr. Venkataiah Gorige for allowing the use of electrochemical workstation in his laboratory.
Author information
Authors and Affiliations
Contributions
VS: Conceptualization, Methodology, Validation, Formal analysis, Investigation, and Writing—review & editing.
Corresponding author
Ethics declarations
Conflict of interest
The author declares no conflict of interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, V. 3D-Printed Electrochemical Platform for Detection of Diabetes Biomarkers: Drop-Based and Time-Based Readout for Clinical Diagnosis. Biomedical Materials & Devices 1, 861–870 (2023). https://doi.org/10.1007/s44174-022-00054-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44174-022-00054-9