Abstract
Materials and developments of medical devices are discussed based on trying to understand nature’s construction principles. A roadmap explaining the papertopics is shown in Fig. 1 of the introduction. Guidelines for producing biomedical materials and devices are discussed. Finally, future development of new technological procedures are mentioned helping human beings to become older and remain healthier during their higher age. Serious problems are pointed out, which will lead to serious social conflicts.

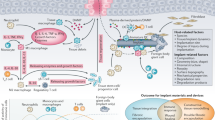
Road-map through passages in the text. The chapters where the different bio-medical materials and devices are discussed are added to the images. Lotuseffect (a), Spider silk (b), Stents (c) [59], Nacre structure (d), Femurale fracture of children (e), Bio-degradable implants (f) [35], Rats’ implants (g) [37], Giraffe’s blood circulation (g) [60]
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In order to develop biomedical materials and devices it is necessary to understand nature’s material-construction principles. Natural materials such as wood, bone and shells serve as models. They are composed of an only limited number of basic components and gain the diversity in properties by being hierarchically organised at the nano- and micro-structural level, and the main attraction of biological materials are multi-functionality and tailored graded properties. This allows them to fulfil a variety of functions such as self-healing, mechanical stability, high toughness, etc. [1]. Knowledge has been gained from nature’s processes of assembling materials to the technical manufacturing side as well as to give guidelines on how to efficiently produce biomedical materials and devices. As visual roadmap through the paper, please see Fig. 1.
Besides the existence of promising transfers from biology to applied technology, they cannot be fully considered is this short overview so that. Only a list of some of them is given, and some specific examples are treated a little more. Though photonic applications in the biomedical field, bio-mineralization inspired materials and systems, autonomous functional materials and systems as well as artificial intelligence and bioinspired materials are promising tasks, they are not considered in this paper.
List of Some Biomedical Materials Being Developed and Applied Successfully for Medical Devices Already
Self-cleaning Lotus Effect
The lotus effect refers to self-cleaning properties that are a result of ultra-hydro-phobicity by the leaves of the lotus flower. Dirt particles are picked up by water droplets due to the micro- and nanoscopic architecture of the leave surface, which minimizes the droplet adhesion to that surface. Various biomaterials have been widely used [2] for biomedical applications, including bio-organs, artificial kidneys and hearts, even in contact with blood, medical and clinical devices like vessel- and blood pumps. The blood compatibility has been studied intensively to prevent negative effects such as thrombosis due to the implanted devices. Recently, the non-wettable character of these surfaces has been shown to be useful for biomedical applications ranging from blood-vessel replacement to antibacterial surface treatment.
Spider silk is the strongest known natural fibre. It has biocompatible and biodegradable properties and is a protein-based material that does not have any allergens or cause immune or inflammatory reactions. Therefore, it is used widely as biomedical material and spiders are breeded and stimulated producing vast amounts of fibres. Silk fibres are even artificially created as replacement for the cellular matrix that human cells create. They are often wolved to scaffolds [3] which are mainly used to ease healing and connecting damaged skin. In addition, spider silk has antiseptic properties and is effective in clotting blood because of its high vitamin K-content. Such scaffolds are developed for a variety of medical devices and eventually antibiotics are added [4], accelerating the growth of new cells while slowly releasing them. Spider silk devices are used for the treatment of slow-healing wounds such as diabetic ulcers, burn injuries or deeper wounds. In addition, they are especially helpful for the fixation of coronary arteries, where they are eventually aimed to be dissolving by themselves after a defined period.
Several biomaterials have been investigated by [5] as possible “nano-plumbers” to fight cardiovascular diseases.
Development of Bio-inspired Medical Materials and Devices for Use as Surgery, Dental and Orthopaedic Implants
Stents are used for several decades for different purposes [6]. They have been improved in various directions, namely in size, construction, temperature-adaption, introduction of chemical substances in order to improve their stability and also to influence the body to withstand negative influences of the surrounding liquids leading to the risk of clogging.
Coronary stents are placed during a coronary angioplasty. They usually consist of a net with the possibility to extent or contract by for example, temperature or forces acting from outside. The most common use for coronary stents is in the coronary arteries, into which a bare-metal stent, a drug-eluting stent, a bio-absorbable stent, a dual-therapy stent (combination of both drug and bioengineered stent), or occasionally a covered stent is inserted [7,8,9]. Vascular stents made of metals can lead to thrombosis at the site of treatment or to inflammation scarring. Drug-eluting stents with pharmacologic agents or as drug delivery [10] vehicles have been developed as an alternative to decrease the chances of restenosis.
Vascular stents are a common treatment for advanced peripheral and cerebrovascular disease. Common sites treated with vascular stents include the carotid, iliac and femoral arteries. Because of the external compression and mechanical forces subjected to these locations, flexible stent materials such as nitinol (Ni–Ti-alloy) are used in a majority of peripheral stents. Also, bio-resorbable Mg-alloys are used if an only temporary presence of the stent is needed [6,7,8].
Soft and hard tissue response in osteo-synthesis implant systems was studied by [9].
Glaucoma drainage stents are recent developments and are awaiting approval in some countries. They are used to reduce intraocular pressure by providing a drainage channel [10].
Materials for use as dental, orthopaedic, and surgery implants have to fulfil especially mechanical properties besides bio-compatibility and avoiding inflammation.
Nacre
High strength and stiffness can be reached by material having nacre structure which is composed of mineralized layers [11,12,13]. The layers are shifted against each other thus hindering crack propagation. Even higher strengths can be attained if organic interfaces are introduced between the layers enabling ultrahigh cross-linking. The increase in strength benefits thus first from the hybrid effect of cross-linking of the organic (collagen) nano-sheets acting as glue. Second, the hierarchical assembly of inorganic and organic layers allows another increase of the crack growth resistance. “Bridges” form at the interface of second-order structure. Such a hybrid shows a high fracture strength, and there are obvious “steps” caused by the growth of channel cracks along the interface of the lamellas. Both, the strength and the Young’s modulus increase dramatically and the plasticity decreases, which is due to the fact that interfacial interactions of the fine plate-like structure of mollusc shells increase and relative sliding of the nano-sheets is hindered during the deformation process [12, 14].
The mechanical properties of a mineralized tissue do not only depend on structural and organizational interactions of mineral and collagen at many hierarchical levels, but other important contributions have to be considered such as the morphological arrangement of phases and micro-porosity in mineralized tissues [15]. Differences in the tissue organization in these zones concerning their stiffness were assessed experimentally. Mean field models of mineralized fibres in the two zones provide a possible explanation of the stiffness differences, especially the mineral distribution between the collagen fibrils and extrafibrillar matrix. In addition, porous structure and fibrils facilitate ingrowth of osteoplasts and thus may support healing of damaged areas which is used especially in tooth surgery.
Bio-mineralisation inspired materials and systems: Nacre, or mother-of-pearl, is formed by calcium carbonate bio-mineralization and has a highly organized internal structure, chemical complexity, and possesses special mechanical properties and optical effects [12, 14, 15]. It is an organo-mineral composite at micro- and nano-scale. The bio-crystal itself is a composite. It has not only the mineral structure of aragonite but possesses intra-crystalline organic material. The primary structural component is a pseudo-hexagonal tablet, about 0.5 µm thick and about 5 to 10 µm in width, consisting primarily (97%) of aragonite, a polymorph of CaCO3, and of organics (3%). It is composed of aragonite crystal tablets covered by an organic matrix. The water-soluble organic matrix dictates which calcium carbonate crystal structure is formed. Nacre can be worked as pieces and powder.
A particular attention was paid to the water-soluble matrix (WSM) molecules of nacre. Rousseau supposed that this part contains molecules, which are released when the nacre implants are placed in living systems. Molecules extracted from nacre of the pearl oyster P. maxima with water have been shown to have a biological activity on various mammalian pre-osteogenic cell types [13]. Nacre, a natural biomaterial of low molecular weight molecules, stimulates the early stages of bone differentiation and the formation of an extracellular matrix able to initiate hydroxyapatite nucleation.
Biomedical devices used as bone implants are based on the above mentioned stimulation of bone differentiation and on the fact that, a complete sequence of osteogenesis results from direct contact between newly formed bone and nacre, anchoring the nacre implant. Another benefit of nacre implants in bone is the formation of new cartilage and bone without any inflammatory reaction and fibrous formation. Experiments on animals with implants in the subchondral bone area in a sheep knee corroborated that, acre can attract and activate osteoblasts and does not cause inflammatory reactions. Nacre was used also as resorbable powder thus inducing the formation of normal bone in vivo. Nacreous trochlea sites were covered with new non fibrous cartilage after implantation in sheep [13].
For intra-medular implants often titanium-alloys [16] are used, whereas for outside fixtures high-strength steels [17] are needed.
Plate failure following plate osteo-synthesis in peri-prosthetic femoral fractures with intramedullary and also outside-bone and tissue implants was studied by [17]. Figure 2 shows in Fig. 2a to c radiographic images viewed from the front and laterally. Figure 2a characterizes double plate fixation following plate failure; b Refracture of the double plate osteo-synthesis; c Revision surgery following double plate failure with another double plate fixation, bone graft, and bone biologics.
Radiographic images seen from the front (a, c, e) and laterally (b, d, f): a Initial bone; b Plate osteo-synthesis; c Bending of the plate; d Revision surgery following plate bending with double plate fixation; e Double plate fracture; f Revision surgery with a long-stem prosthesis, bone graft, bone biologics and cerclage
Incredibly tough composite materials can be made by mimicking the fine plate-like structure of mollusc shells. The hybrid material matches the strength and toughness of the best aluminium alloys and is even more resistant to fracture than nacre. Construction of biomedical devices often consist of a layered double hydroxide-based hierarchical structural hybrid material with two orders of interfaces (sandwich structure). The highest strength of this hybrid material with an optimal combination of these two orders of interfacial interactions is about 230 MPa, and the Young’s modulus is about 131 GPa. These values are higher than most common clay-based hybrids, making the strengthening strategy (strengthening materials by adjusting interfacial interactions of these two orders of interfaces) a new and effective strengthening method. Another potential improvement of the impact strength of sandwich structures is obtained using graded layered woven composite core and elastomer skin additions [3]. Using poly-urea (PU) elastomer additions to the face-sheets, and a functionally graded plain weave architecture that is graded along the through-thickness as a substitute for the unidirectional laminated core, the impact resistance of the structure can be greatly increased. Using graded woven composite core can improve the through-thickness stiffness of the sandwich. Another technique uses glass fibre-reinforced polymers. There, a crack is deflected at the interface between the fibres by a delamination process which absorbs more energy than that of unidirectional carbon fibre-reinforced polymers.
Use of strategies to obtain ultra-high strengthening effects refer for example, to devices in human dentistry [18]. The strength of the ultrahigh strength material is similar to the extremely hard enamel covering of the tooth below. This tough shell is the hardest tissue in the human body and protects the inner more fragile areas of the tooth, known as dentin and pulp. It is the first and most important line of defending tooth decay. Enamel is comprised the mineral calcium phosphate, arranged in a crystal structure known as hydroxyapatite. Likewise sea shells and pearls are made from calcium carbonate. Both, teeth and sea shells are more complex than they might first appear. Dentistry has developed and still regularly improves devices similar to the mentioned organizational structure. For this, the patient’s age, condition of jaw, number of teeth that should be arranged, etc. must be identified. But ultra-high strengthening material, such as special steels is also used for devices which help reinforcing the hoofs of horses which are extremely stressed during racings or on granite streets.
Another application of ultra-high strength material is for human children to help shaping their bones during growth.
Deformity Correction, Intra-medular Bone Nails and External Fixation, Bone Repair
Either intra-medular bone nails, including fixation with scews, are used or plates are placed outside of the leg, and connected with the bone using screws eventually with an outside fixation (osteo-synthesis). For intra-medular nails titanium or titanium alloys are used as material, whereas high-strength steels are used for external devices [17, 19, 20].
In order to correct misalignments of limbs osteotomy is eventually applied severing the bone with a chisel or a saw. The consecutive use of external fixation and guided growth to treat congenital deformities of the lower limbs is a promising direction for paediatric orthopaedics. The repeated use of guided growth, because of its minimal invasiveness, is the most effective solution for the recurrence of deformity in a growing child. The present results indicate that consistent use of methods of trans-osseous osteo-synthesis and controlled growth in the treatment of congenital deformities of the lower extremities is a promising approach in paediatric orthopaedics. Repeated use of controlled growth owing to its low morbidity provides an effective solution for the problem of recurrent deformity in a growing child [19, 20].
Not only corrections of congenital deformities are performed today but, also growth of bones that cannot grow as a result of, for example, accidents or other dramatic external influences is made possible. For this, sophisticated devices are and have to be developed considering the individual bone geometry, the driving muscle system, which is determined by physical parameters as velocity, acceleration, force, energy, and power, etc. The replacement of complete lower legs are constructed and permanently adapted to the individual men who have lost their legs. As demonstrated in several competitions they achieve incredible velocities [21].
Bio-degradable Material for Temporary Orthopaedic Implants and Development of Devices
Metallic biomaterials such as stainless steels, titanium and cobalt-chromium-based alloys must be resistant against corrosion, if it is aimed to keep their structural stability for long enough periods. But their mechanical properties are different from the biological tissue. Especially the elastic modulus is much higher than the bone and thus stress shielding is resulting [22]. For only temporary usage of the implant, however, a painful operation for its removing is necessary.
Another aspect has likewise to be considered [23]: tissue morphology and function are undividable for a correct understanding diseases in childhood and adolescence [24]. When bone diseases are closely associated with muscular function—because mechanical stimuli are essential elements in the feedback loop regulating bone development—the pediatric osteologist must consider the muscular function. This leads to an understanding key principles in the clinical analysis of the muscular function by the use of physical parameters as velocity, acceleration, force, energy, and power. Mechano-graphic analyses of jumping and chair-rising were introduced in the diagnostic repertoire of the pediatric osteologist [25]. There are many open questions in paediatric biomechanics and how to make use for medical devices, for example: are maximal forces the essential parameter modeling bone and what is the optimal frequency at which these forces should be applied to support the increase in bone mass. A precise answer to these open questions will be necessary for a sufficient understanding of the physiology and treatment of bone diseases.
In order to avoid a second operation for removal of the implant, material for temporary usage has been chosen. Especially magnesium and Mg-alloys proved as most successful metallic biodegradable materials for defined temporary usage. Their advantages are manifold [3, 26,27,28,29,30,31,32,33]:
-
1.
They can degrade owing to their corrosion susceptibility in aqueous and especially in physiological environment.
-
2.
Mg is biocompatible [33], Mg ions (Mg2+) are used in the regular metabolism and no toxic effects appear.
-
3.
The elastic modulus of Mg is 40–45 GPa and thus matches the stiffness of bone (3–20 GPa) much better than the above mentioned stainless steel (ca. 200 GPa), cobalt-based alloys (ca. 230 GPa) and titanium alloys 115 GPa).
-
4.
Mg alloys are light-weight metals, and their density is similar to that of bone (1.8–2.1 g/cm3), which in addition makes implants lighter.
-
5.
Excess Mg as produced during implantation and implant degradation in the body is excreted via the kidneys [34].
-
6.
Mg forms corrosion products on the implant surfaces. These are Ca-phosphates, very similar to the inorganic constituent of human bone, hydroxyapatite [34]
-
7.
The most advantageous property of Mg implant material is that, they need not be removed by a second operation, but are resolved by the body—exact timing is most crucial (see later)—which is especially advantageous in children’s surgery (see later).
One problem, however, was the formation of hydrogen bubbles which may grow and form thicker layers as a result of local Mg corrosion. This caused surgeons about 40 years ago to abandon usage of Mg implants due to pains of their patients. Today [35] however, this problem has been mainly solved by scientific research and understanding of the responsible mechanisms [36].
The use and development of bio-degradable material for temporary orthopaedic implants and devices is especially helpful for healing bones of children. Numerous partly new research methods are applied in order to obtain optimum results. From a clinical point of view, biodegradable materials suitable for osteo-synthetic applications must exhibit several fundamental properties:
-
(i)
biocompatibility with the living organism;
-
(ii)
sufficient mechanical flexural rigidity to ensure fracture stabilization during the inflammatory and reparative phases of fracture healing (4–16 weeks) [35]; and
-
(iii)
degradation performance in balance with fracture consolidation. The implant has to allow strengthening of a damaged bone during the initial period after its damage and has to become weaker later when the bone has attained some stiffness already.
Material and research methods were developed in a consortium group in Graz (Medical University), Vienna (Technical University and University of Natural Resources (all in Austria) and ETH Zürich (Switzerland) [34, 35]. Development of material was done in Switzerland with parallel and subsequent mechanical and analytical testing. Material selection was concentrated on polymers, some metal alloys, partly equipped with polymer covers. Following results were obtained: Mg-alloys were found to meet best the requirements [26,27,28,29, 31,32,33, 37,38,39,40,41,42,43,44,45,46,47,48,49].
Animal tests, experimental procedures, main results and consequences obtained by the above mentioned researchers and some companies are reported in the following chapters.
Animal tests were performed with male Spragley rats of different age and after different specified times. All animals were fed and taken care in exactly the same way. For the implantation cylindrical pins with a diameter of 1–1.2 mm were implanted trans-cortically in the rats’ femur of their hind legs and were measured ex-vivo after 1, 3, 6, 9 and 12 months after implantation. This procedure allowed to observe the development of healing in different ways. Mainly following tests were performed [34] with:
-
1.
Push-out tests, the adhesive strength of the bone-implant-interfaces and thus the difference of titanium and Mg-alloy was determined.
-
2.
SEM (scanning electron microscopy) and EDX (energy dispersive X-ray spectroscopy), reasons for different shear strengths and energies were investigated and surface structures of explanted implants made visible.
-
3.
Inductively Coupled Plasma-Mass Spectrometry (ICP-MS), allowed determining the content of alloying elements like Mn, Y, Zn was quantitatively analysed and by
-
4.
Additional use of laser ablation of the bone surface, the distribution of elements in the bone surrounding the implant was detected. The push-out tests demonstrated that the push-out energy of the Mg–Zn–Ca—bone interface is higher after all implantation times than for the Titanium interface. The SEM images showed metal oxides and a significant amount of phosphorus, suggesting that organic components have been deposited.
Kraus et al. [35] studied: thirty-two male Sprague–Dawley rats with a body weight of 140–160 g and 5 weeks of age were used in this study. The rats were divided in two groups: 16 rats belonged to the ‘‘continuous-μCT’’ group and 16 to the ‘‘histological’’ group. Each rat in each group got two identical pins (either WZ21 or ZX50) implanted into its femoral bones. The rodents in the ‘‘continuous μCT’’ group underwent μCT evaluation at the seventh day after operation followed by further μCT examinations every four weeks up to the 24th week. Histological examinations were performed at weeks 4, 12, 24, and 36 post operation in which two rats of each group (ZX50 and WZ21) were sacrificed.
Following analysing methods were utilized by [35]:
-
5.
Histologic methods to obtain information on bone and tissue development during specified times after the implantation were applied whereby the two Mg-alloys WZ21 (chem., mech.) and ZX50 chem., mech.) were compared.
-
6.
Micro-focus computed tomography (μCT) combining both non-invasive, tissue-preserving imaging and quantitative morphometry of bone structure in three dimensions [35, 42, 50, 51], was used. For the 3-D reconstruction of a specimen from a set of 2-D projections the back projection of a filtered projection was used after the animals were generally anaesthetized by volatile isoflurane. For histological sample processing explanted bones were dissected from soft tissues and fixed in neutral buffered 4% formalin solution (Fig. 3).
Push-out energy and displacement measured in push-out tests of the Mg–Zn–Ca-alloy (WZ21) and Titanium (F-dx) plot (a), by integration of the F-dx plot the push-out energy is derived (b) which characterizes the quality of the incorporation of different implants. Use of the LA-ICP-MS technique (c): reveales very high Mn-concentrations up to 0.8 mm distance from the implant and a constant level in bone as well as lower Mn-accumulation in the bone marrow
The main results of [34], and [35] are summarized in the following paragraphs and figures:
-
1.
Most advantages in comparison to Ti-alloys or polymers were obtained with Mg-alloys (typically ZX50 and WZ21). It could be demonstrated that, the mechanical properties of the interface between bone and implant WZ 21 are superior to that of a Ti-implant. Use of the LA-ICP-MS technique revealed very high Mn-concentrations up to 0.8 mm distance from the implant and a constant level in bone and lower Mn-accumulation in the bone marrow. Similar studies were performed on Y, Zn, Mg and Ca. Degradation elements were stored in the body, including rare earths such as Y. Therefore material without these elements was suggested to not be used in the future.
-
2.
In paediatric surgery, the elastic stable intramedullary technique is the rising osteo-synthetic technique for long bone fractures of children which allows to prevent a second operation for removing the implant. This was and still is the greatest advantage of using bio-resorbable implants for stabilizing children’s bone fractures. [35] obtained with sophisticated techniques (see above: histologic methods and micro-focus computed tomography (μCT)) following results on the degradation behavior of ZX50 material and a WZ21 alloy:
The volume and surface changes and the amount of hydrogen gas formation of the ZX50 and WZ21 pins were followed by in vivo CT analysis throughout the entire study period of 24 weeks (see Fig. 4a–d). The alloys exhibited very different degradation rates, as indicated by the change in volume shown in Fig 4a. The ZX50 pins did start to corrode immediately after implantation and exhibited surface pits already within the first week, as illustrated in the 3-D reconstructions shown in Fig. 4d. The measured pin surface slightly increased initially as a result of the degradation (Fig. 4b). Besides disturbing the bone implant connectivity, the hydrogen gas was particularly pronounced in the case of the ZX50 pins.
Degradation of WZ21 and ZX50 alloys during the 24 week study period detected by CT measurements. a Loss of volume; b change of surface area; c volume of hydrogen-gas bubbles; d 3-D reconstruction of the pins. WZ21 alloy shows a moderate loss of pin volume and a degradation process in a rather linear manner after the eighth week. During degradation only few gas bubbles were visible. In ZX50, corrosion is rather fast and grooving is visible after the first week and no more pin material is seen at week 16. The rapid corrosion of ZX50 formed great amounts of gas within a short period
Tissue healing appeared quite undisturbed in WZ21 and newly formed bone entered tight connection to the implant surface (Fig. 5e and f). The degradation was delayed within the first 4 weeks probably owing to the formation of a fairly stable corrosion- product layer on the implant surface, which protected the material against further degradation [27, 40, 52].
Histological thin slides of ZX50 (a–d) and WZ21 (e–h) pins in a Levai–Laczko staining. WZ21 shows the positive properties of Mg alloys by enhancing new bone formation around the implant. Even in the case of massive callus formation and release of high amounts of gas (e–h) the bone does not show any permanent damage
It may be summarized that, the main results on material and devices providing repair of children’s bone fractures have been obtained by careful selection of optimum biomedical materials and extensive development of devices. Mg-alloy WZ21 proved to be the optimum biomedical material. Among other properties, the connection with muscles surrounding the bone, has to be considered. As each child is different from another one, the developments referring material and devices were and still are adapted in an individually driven manner and hopefully render further improvements in the future.
Adverse properties, namely low weight devices with optimized strength, stiffness, and elasticity can be reached by mimicking the structure of bamboo. Its fast reproduction is additionally advantageous. These features are realized nowadays by men already.
Biomedical Devices Derived from Giraffes’ Blood Circulation
A rather new finding on nature’s mechanisms is promising to improve the operation technique by stabilising the blood circulation of patients with narrowed blood vessels [53]. Giraffes have solved a problem that kills millions of people every year by high blood pressure. The solutions, only partly understood by scientists so far, involve pressurized organs, altered heart rhythms, blood storage and the biological equivalent of support stockings. Investigations on giraffes with their extremely long neck showed that, the blood pumping during moving periodically changes and regulates it. This is based on a giraffe's large neck veins containing a complex system of valves that stop the blood from flowing backward and pooling when it dips its head. Researchers hope revealing the secrets of the circulatory system of giraffes in order to help humans with the tools to combat high blood pressure and cardiovascular disease which is responsible for millions of dead people in the civilized world.
The blood vessels of giraffes have an extremely high diameter, namely up to about 25 mm. In fact, an adult giraffe's blood pressure can reach 300/180 mm. This value is roughly twice of that of an adult human and would cause heart and kidney failure, swollen ankles and legs for people. But the value of 300/180 mm is only near the giraffe’s heart. In their heads, a giraffe's blood pressure remains about equal to that of humans. This is accomplished with a tight wrapping of natural compression socks in the tight skin of necks and legs.
Blood pools develop in the big veins of the neck when the head is down. This stores more than a litre of blood, temporarily reducing the amount of blood returning to the heart. With less blood available, the heart generates less pressure with each beat while the head is down. As the head is raised again, the stored blood rushes suddenly back to the heart, which responds with a strong high-pressure stroke that helps pumping blood up to the brain.
It was also found that the left ventricles of heart did get thicker with the giraffes’ age, but without the stiffening, or fibrosis, that would occur in people. The researchers also found that giraffes have mutations in five genes related to fibrosis. In keeping with that find, other researchers who examined the giraffe genome found several giraffe-specific gene variants related to cardiovascular development and maintenance of blood pressure and circulation. All these properties are based on several specific biomedical materials’ of the giraffe body.
And the giraffe has another trick to avoid heart failure: The electrical rhythm of its heart differs from that of other mammals so that the ventricular-filling phase of the heartbeat is extended. This allows the heart to pump more blood with each stroke, allowing a giraffe to run hard despite its thicker heart muscle. “All you have to do is look at a picture of a fleeing giraffe” Natterson-Horowitz said, “and you realize that the giraffe has solved the problem” [53].
Owing to non-certified information, NASA is developing biomedical devices for astronauts losing their perfect body response due to vacuum environment. The involved researchers in addition aim to help human beings suffering from different diseases such as cardiovascular problems after strokes. by implementing the above mentioned strategies of nature.
Discussion
Based on trying to understand nature’s material-construction principles, several materials and developments of medical devices are discussed in more or less detail. The main advantage of biological materials are their multi-functionality and tailored graded properties. Thus a variety of functions such as self-healing, mechanical stability, high toughness, and strength are obtained. Studies of the hierarchical structure of natural materials [54,55,56] allow additional understanding of their properties. Guidelines on how to efficiently produce biomedical materials and devices have become possible [57]. Future emphasis should be laid on an even better understanding of material-construction properties and it may be expected that the rapid development of new technological procedures contributes essentially. An example refers to replace legs by artificial prosthetic devices [58]. New bio-medical devices probably will help human beings to become older and remain healthier during their higher age. Nonetheless we must not ignore that serious problems will arise and may lead to bad social conflicts.
Change history
31 October 2022
A Correction to this paper has been published: https://doi.org/10.1007/s44174-022-00043-y
Abbreviations
- WSM:
-
Water-soluble matrix
- MPa:
-
Mega Pascals
- GPa:
-
Giga Pascals
- PU:
-
Poly-urea
- Mg:
-
Magnesium
- ETH:
-
Eidgenössische Technische Hochschule
- SEM:
-
Scanning electron microscopy
- EDG:
-
Energy dispersive X-ray spectroscopy
- ICP-MS:
-
Inductively Coupled Plasma-Mass Spectrometry
- Mn:
-
Mangan
- Y:
-
Yttrium
- Zn:
-
Zinc
- Mg–Zn–Ca:
-
Magnesium–zinc–calcium
- ICT:
-
In-circuit-test
- g:
-
Grams
- 3-D:
-
Three dimensional
- 2-D:
-
Two dimensional
- Ti-alloys:
-
Titanium alloys
- Ti-implant:
-
Titanium implant
- μCT:
-
Micro-computer tomography
- mm:
-
Milli metre
- F-dx:
-
Force versus distance
References
I. Milne, U.K. Matlock, R.O. Ritchie, B. Karihaloo, Y.W. Mai (eds.) Structural integrity management. Comprehensive Structural Integrity, vol 9 Teoh SH. Failure in biomaterials (2007), pp. 1–30
J.C.Y. Kenry, C.T. Lim, Emerging flexible and wearable physical sensing platforms for healthcare and biomedical applications. Nature 2, 1–19 (2016)
J. Williams, Potential improvement of the impact strength of hybrid fiber reinforced polymers using graded layered woven composite core and elastomeric. Acad. Lett. (2021). https://doi.org/10.20935/AL1953
A. Liu, M. Sun, H. Shao, X. Yang, Outstanding mechanical response and bone regeneration capacity of Robocast dilute magnesium-doped Wollastonite scaffolds in critical size bone defect. J. Mater. Chem. 4, 3945–3958 (2016)
F. Dormont, M. Varna, P. Couvreur, Nanoplumber: biomaterials to fight cardiovascular diseases. Mater. Today 21, 122–143 (2018)
A.C. Hänzi, Development of biodegradable magnesium alloys for cardiovascular stent applications (2009) Diss. ETH No. 18745
M. Moravej, D. Mantovani, Biodegradable metals for cardiovascular stent application: interests and new opportunities. Int. J. Mol. Sci. 12, 4250–4270 (2011)
M. Weidling, S. Besdo, T. Schilling, M. Bauer, T. Hassel, F.W. Bach, H.J. Maier, J. Lamon, A. Haverich, P. Wriggers, Development of Magnesium Alloy Scaffolds to Support Biological Myocardial Grafts, A Finite Element Investigation. Lecture Notes in “Biomedical Technology,” in Lecture Notes in Applied and Computational Mechanics. ed. by T. Lenarz, P. Wriggers (Springer, Berlin, 2015)
B. Schaller, In vivo degradation of magnesium plate/screw osteo-synthesis implant systems: soft and hard tissue response in a calvarial model in miniature pigs. J. Cranio-Maxillo-Facial Surg. J. 44(3), 309–317 (2016)
T. Demetrios, D.T. Manasses, L. Au, The new era of glaucoma micro-stent surgery. Ophthalmol. Therapy 5, 135–146 (2016)
S.C. Cao, J. Liu, L. Zhu, L. Li, M. Dao, J. Lu, R.O. Ritchie, Optimization of the strain rate to achieve exceptional mechanical properties. Nature 8(1), 10 (2018)
R. Metzler, A. Mike, O. Ronke, A. Daniel, J. Christopher, F. Bradley, S. Coppersmith, G. Pupa, Architecture of columnar nacre, and implications for its formation mechanism. Phys. Rev. Lett. (2007). https://doi.org/10.1103/PhysRevLett.98.268102
M. Rousseau, Nacre, a natural biomaterial. Biomater. Appl. Nanomed. 14, 281–298 (2008)
P. Ghosh, D.R. Katti, S. Kalpana, Mineral proximity influences mechanical response of proteins in biological mineral−protein hybrid systems. Biomacromolecules 8, 851–856 (2007)
M. Rousseau, The water-soluble matrix fraction extracted from the nacre of Pinctada maxima produces earlier mineralization of MC3T3-E1 mouse osteoplasts. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 10, 1–7 (2003). https://doi.org/10.1016/S1096-4959(03)00032-0
R. Zhou, Y. Han, J. Cao, M. Li, G. Jin, H. Luo, L. Zhang, B. Su, Electrically bioactive coating on Ti with bi-layered SnO2-TiO2 hetero-structure for improving osteointegration. J. Mater. Chem. B 6, 3989–3998 (2018)
S. Boesmueller, S.F. Baumbach, M. Hofbauer, G.E. Wozasek, Plate failure following plate osteosynthesis in periprosthetic femoral fractures. Wien Klin Wochenschr 127, 770–778 (2015)
M. Mardall, H. Salimijazi, F. Karimzadeh, B.J.C. Luthringer-Feyerabend, C. Balwert, S. Labbaf, Fabrication and characterization of nanostructured hydroxyapatite coating on Mg-based alloy by high-velocity oxygen fuel spraying. Ceram. Int. 44, 4667-4676.18 (2018)
A. Popkov, P. Foster, A. Gubin, D. Borzunov, D. Popkov, The use of flexible intramedullary nails in limb lengthening. Expert Rev. Med. Devices 14(9), 741–753 (2017)
V.A. Vilensky, E.A. Zakharjan, A.A. Pozdeev, T.F. Zubairov, A.P. Pozdeev, Treatment of congenital long-bone deformities in children using the consequtive application of guided growth and external fixation: preliminary report. Turner Sci. Res. Inst. Child. Orthop. 6(3), 12–24 (2018)
R. Keerthana, M.C. Jochan, S. Dharshini, S.S. Sunilkumar, S. Kalidindi, S. Vidhya, A review on prosthetics and orthotics for amputees and disabled. J. Crit. Rev. 7(15), 2175–2189 (2020)
O. Bostman, H. Pihlajamaki, Clinical biocompatibility of biodegradable orthopaedic implants for internal fixation: a review. Biomaterials 21, 2615–2621 (2000)
E. Schoenau, O. Fricke, Mechanical influences on bone development in children. Eur. J. Endocrinol. 159, 27–31 (2008)
J. Wolff, Morphogenesis of bone. J. Am. Med. Assoc. 213, 2260 (1970)
O. Fricke, J. Weidler, B. Tutlewski, E. Schoenau, Mechanography – a new device for the assessment of muscle function in pediatrics. Pediatr. Res. 59, 46–49 (2006)
S. Kamrani, C. Fleck, Biodegradable magnesium alloys as temporary orthopaedic implants: a review. Biometals 32, 185–193 (2019)
H.R. Tiyyagura, T. Mohan, S. Pal, Surface modification of magnesium and its alloy as orthopaedic biomaterials with biopolymers. in Fundamental Biomaterials; Metals (2018)
M.I. Rahim, S. Ullah, P.P. Mueller, Advances and challenges of biodegradable implant materials with a focus on magnesium-alloys and bacterial infections. Metals 8(7), 532 (2018)
Bionano-Materials, de Gruyter, Abstracts. Jahrestagung der Deutschen Gesellschaft für Biomaterialien, vol. 14, no.1, eds. by S. Barzilowsky, K. Jockenhövel, K. Sternberg, M. Stiesch (2013)
Y. Su, J. Lin, Y. Su, W. Zai, G. Li, C.U. Wen, Investigation on composition, mechanical properties, and corrosion resistance of Mg-0.5Ca-X (Sr, Zr, Sn) biological alloy. Hindawi Scanning 10, 1–11 (2018). https://doi.org/10.1155/2018/6519310
B. Schaller, N. Saulacic, T. Imwinkelried, S. Beck, E.W. Liu, J. Gralla, K. Nakahara, W. Hofstetter, Iizura T In vivo degradation of magnesium plate/screw osteosythesis implant systems: soft and hard tissue response in a calvarial model in miniature pigs. Craniomaxillofac. Surg. 44(3), 309–317 (2016)
N.T. Kirkland, M.P. Staiger, D. Nisbet, C.H.J. Davies, N. Birbilis, Performance-driven design of biocompatible Mg alloys. JOM (2011). https://doi.org/10.1007/S11837-011-0089-z
L. Li, J. Gao, Y. Wang, Evaluation of cyto-toxicity and corrosion behavior of alkali-heat-treated magnesium in simulated body fluid. Surf. Coat. Technol. 185(1), 92–98 (2004)
F. Amerstorfer, S.F. Fischerauer, L. Fischer, J. Eichler, J. Draxler, A. Zitek, M. Meischel, E. Martinelli, T. Kraus, S. Hann, S.E. Stanzl-Tschegg, P.J. Uggowitzer, J.F. Löffler, A.M. Weinberg, T. Prohaska, Long-term in vivo degradation behavior and near-implant distribution of resorbed elements for magnesium alloys WZ21 and ZX50. Acta Biomater. 42, 440–450 (2016)
T. Kraus, S. Fischerauer, A.C. Hänzi, P.J. Uggowitzer, J.F. Löffler, A.M. Weinberg, Magnesium alloys for temporary implants in osteosynthesis: In vivo studies of their degradation and interaction with bone. Acta Biomater. 8, 1230–1238 (2012)
B. Zberg, P.J. Uggowitzer, J.F. Löffler, MgZnCa glasses without clinically observable hydrogen evolution for biodegradable implants. Nat. Mater. Lett. 8, 887 (2009)
M. Meischel, D. Hörmann, J. Draxler, E.K. Tschegg, J. Eichler, T. Prohaska, S.E. Stanzl-Tschegg, Bone-implant degradation and mechanical response of bone surrounding Mg-alloy implants. J. Mech. Behav. Biomed. Mater. 71, 307–313 (2017)
X. Song, L. Chang, J. Wang, S. Zhu, L. Wang, K. Feng, Y. Luo, S. Guan, Investigation on the in vitro cytocompatibility of Mg-Zn-Y-Zr alloys as degradable orthopaedic implant materials. J. Mater. Sci. 29, 44 (2018)
Y. Su, J. Lin, Y. Su, W. Zai, G. Li, C. Wen, Investigation on composition, mechanical properties, and corrosion resistance of Mg-0.5Ca-X (Sr, Zr, Sn) biological alloy. Scanning (2018). https://doi.org/10.1155/2018/6519310
G. Jia, C. Chen, J. Zhang, Y.Y. Wang, R. Yue, B.J.C. Luthringer-Feyerabend, R. Willumeit-Roemer, H. Zhang, M. Xiong, H. Huang, G. Yuan, F. Feyerabend, In vitro degradation behavior of Mg scaffolds with three-dimensional interconnected porous structures for bone tissue engineering. Corrosion Sci. 144, 301–312 (2018)
E.L. Silva, S.V. Lamaka, D. Mei, M.L. Zheludkevich, The reduction of dissolved oxygen during magnesium corrosion. Chem. Open Commun. 7, 664–668 (2018)
N. Stephanopoulos, J.H. Ortony, S.I. Stupp, Self-assembly for the synthesis of functional biomaterials. Acta Mater. 61, 912–930 (2013)
J.R. Mosley, L.E. Lanyon, Strain rate as a controlling influence on adaptive modeling in response to dynamic loading of the ulna in growing male rats. Bone 23, 313–318 (1998)
K.M. Reich, C.D. Huber, P. Heimel, C. Ulm, H. Redl, S. Tangl, A quantification of regenerated bone tissue in human sinus biopsies: influences of anatomical region, age and sex. Clin. Oral Impl. Res. 27, 1–8 (2015)
P. Fratzl, H. Gupta, E. Paschalis, P. Roschger, Structure and mechanical quality of the collagen-mineral nano-composite in bone. J. Mater. Chem. 14, 2115–2123 (2004)
A.G. Reisinger, D.H. Pahr, P.K. Zysset, Sensitivity analysis and parametric study of elastic properties of an unidirectional mineralized bone fibril-array using mean field methods. Biomech. Model Mechanobiol. 9, 499–510 (2010)
A. Atkinsa, M.N. Deanb, M.L. Habegger, P.J. Mottac, L. Ofera, F. Repp, A. Shipova, S. Weinerd, J.D. Curreye, R. Shahara, Remodeling in bone without osteocytes: Billfish challenge bone structure–function paradigms. Proc. Natl. Acad. Sci. 111, 16047–16052 (2014)
R.V. Marrey, R. Burgermeister, R. Grishaber, R.O. Ritchie, Mechanical properties of bone and other biological materials. Medical Device Materials, Proceedings of the 3rd ASM Materials & Processes for Medical Devices Conference, ASM Intl (2006)
P. Li, M.R. Ryder, J.P. Stoddard, Hydrogen-bonded organic frameworks: a ricing class of porous molecular materials. Accounts Mater. Res. 1, 77–87 (2020)
L.A. Feldkamp, S.A. Goldstein, A.M. Parfitt, G. Jesion, M. Kleerekoper, The direct examination of three-dimensional bone architecture in vitro by computed tomography. J. Bone Miner. Res. 4, 3–11 (1989)
M.H. Bünger, M. Foss, K. Erlacher, H. Li, X. Zou, B.L. Langdahl, C. Bünger, H. Birkedal, F. Besenbacher, J.S. Pedersen, Bone nanostructure near Titanium and porous Tantalum implants studied by scanning small angle X-ray scattering. Eur. Cells Mater. 12, 81–91 (2006)
F. Witte, V. Kaese, H. Haferkamp, E. Switzer, A. Meyer-Lindenberg, C.J. Wirth, In vivo corrosion of four magnesium alloys and the associated bone response. Biomaterials 26, 3557–3563 (2005)
B. Holmes, The cardiovascular secrets of giraffes. Because of their height, giraffes require scarily high blood pressures—yet they escape the massive health problems that plague humans with hypertension. Smithonian mag.com/science-nature May 21, 2021.
P. Fratzl, R. Weinkammer, Nature’s hierarchical materials. Progress Mater. Sci. 52, 1263–1334 (2007)
P. Fratzl, R. Weinkammer, Hierarchical Structure and Repair of Bone: Deformation, Remodelling, Healing (Springer, Berlin, 2007)
P. Fratzl, O. Kolednik, F.D. Fischer, M.D. Deana, The mechanics of tessellations – bioinspired strategies for fracture resistance. Chem. Soc. Rev. 45, 252 (2015)
U.G.K. Wegst, H. Bai, H. Saiz, A.P. Tomsia, R.O. Ritchie, Bioinspired structural materials. Nat. Mater. 14, 23–36 (2014)
R. Keerthana, M.C. Jochan, S. Dharshini, S.S. Sunilkumar, S. Kalidindi, S. Vidhya, A review on prosthetics and orzholtics for amputees and disabled. J. Crit. Rev. 7, 2175 (2020)
B. Medical, Medical gallery of Blausen medical 2014. WikiJ. Med. 1(2), 1–79 (2014). https://doi.org/10.15347/wjm/2014.010.ISSN2002-4436
Abspires 40. Best of Kruger 2014 Giraffe drinking. https://www.flickr.com/photos/76506919@N04/13312427465. Licensed under CC BY 2.0 https://creativecommons.org/licenses/by/2.0/
Funding
Open access funding provided by University of Natural Resources and Life Sciences Vienna (BOKU). The author declares that no funding was provided.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that she has no conflict of interest.
Additional information
The original online version of this article was revised: In author affiliation footnote 2: IMP, Department of Material Sciences and Process Engineering – MAP, Cambridge, 1130 Vienna, Austria. The word “Cambridge” should be deleted.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stanzl-Tschegg, S.E. Biomedical Materials and Devices with Focus on Orthopaedic and Cardio-vascular Problems. Biomedical Materials & Devices 1, 179–190 (2023). https://doi.org/10.1007/s44174-022-00025-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44174-022-00025-0