Abstract
The reports over the years on chemotherapeutic regimen involving cyclophosphamide (CYP), a bifunctional alkylating agent, demonstrated hepatotoxic side effect. Eulophia gracilis (EG) is a medicinal plant with folkloric utility in the treatment of liver damage and blood related diseases. However, there is a knowledge gap on the impact of E. gracilis effectiveness on CYP-associated hepatic toxicity in the literature. We investigated on potency of aqueous methanolic extract of E. gracilis (AMEG) and CYP-mediated hepatic toxicity in rats. Experimental rats were administered with CYP (2 mg/kg) or co-treated with AMEG (200 or 400 mg/kg) for 7 days consecutively. The result showed that co-treatment with AMEG significantly reduces alanine aminotransferase, aspartate aminotransferase, gamma glutamyl transferase and lactate dehydrogenase activities compared to the CYP group. Moreover, AMEG abated CYP-induced decreases in superoxide dismutase, catalase and glutathione peroxidase enzymes in the liver homogenate. AMEG alleviated CYP-facilitated surges of hepatic concentration of advanced oxidized protein product (AOPPs) and lipid peroxidation in rats. Additionally, AMEG reduced pathological lesions in the liver of co-treated rats and elicited anti-genotoxic effect by mitigating CYP-mediated increases of frequency of formation of polychromatic erythrocyte in the bone marrow and hepatic percentage DNA fragmentation in CYP-exposed rats. Overall, AMEG protective effect improved liver dysfunction occasioned by CYP-mediated toxicities in rats by abating oxidative stress and alleviating genotoxic responses.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The treatment of ever-increasing cases of cancer with recommended conventional chemotherapy has generated lots of conflicting concerns as treatment presents myriads of miserable side effects alongside their chemotherapeutic curative roles. Many of the active anticancer drugs commonly used caused infertility, immuno-suppression, hepatic injury and blood clots fatigue [1]. In the course of their metabolism, majority of chemotherapeutic agents such as Doxorubicin, Cisplatin, Methotrexate and Cyclophosphamide induced oxidative stress and thereby caused injury to the tissues [2, 3].
Cyclophosphamide is one of the old and common chemotherapies used in treating cancer, autoimmune disease, blood disease and marrow transplantation [4]. It was introduced as cancer therapy in 1959 [5] and remains an important therapy of hematological malignancies such as lymphoma, leukemia and epithelial tumors like ovarian, breast carcinomas and small-cell lung carcinomas [6]. The drug was approved in Germany as treatment for acute lymphoblastic leukemia, non-Hodgkin lymphoma, chronic lymphoblastic leukemia and Hodgkin, ovarian carcinoma, plasmacytoma, breast cancer and rhabdomyosarcoma and neuroblastoma [7]. Though, it is used as an anticancer drug, it has however been identified as human and animal carcinogen by International Agency for Research Centre [8]. It causes injuries to liver and other vital organs in which such side effects are linked to the reactive oxygen species [9] such as acrolein and phosphoramide mustard formed during its metabolism in the liver by cytochrome P450 mixed functional oxidase system [10, 11]. Its active metabolite phosphoramide mustard was shown to form reactive cyclic aziridinium cation that reacts with N7 of guanine and also with cytidine from DNA and making it genotoxic [12].
However, the input on medicinal plant research has increased on the basis of less toxic and little or no adverse effects and possession of bioactive compounds with proven chemopreventive potentials [13, 14]. This has therefore led to recent screening for novel therapeutic compounds in plants that have healthy interactions with biological molecules. Eulophia gracilis belongs to family of Orchidaceae which is the largest family in plant kingdom that comprises 25,000–35,000 species [15]. In addition to ornamental values of Orchidaceae family, they possess medicinal value due to their high content of alkaloids, phenols, glycosides and useful phytochemicals [16]. Eulophia gracilis in particular has been shown to contain phytochemicals of therapeutic importance and are used in traditional medicine for treatment of myriads of ailment including cancer, tumors, as aphrodisiac and liver disorders in Nigeria [17].
Therefore, this study aimed to determine protective effect of E. gracilis extract on cyclophosphamide-induced liver injury in rats employing the antioxidant markers and biomarkers of liver damage. This study may improve the cyclophosphamide regimen in enhancing its tolerance with minimal adverse effects.
2 Materials and methods
2.1 Plant collection, identification and preparation
Fresh E. gracilis plants were collected from rocky area in Oyo, Nigeria. The plant was identified and authenticated by plant taxonomist at Herbarium section of Department of Botany, University of Ibadan, Nigeria with University of Ibadan Herbarium (UIH) number 22528. The tubers were thoroughly washed with tap water, sliced into pieces and shed dried. The dried tubers were pulverized into powder form using electric grinder. 200 g of E. gracilis fine powdered sample was extracted with 800 mL aqueous methanol (20:80) for 48 h by cold maceration. The extract was evaporated under reduced pressure by using a rotary evaporator and further lyophilized using freeze-dryer machine.
2.2 Chemicals and reagents
Cyclophosphamide tablet is a product of West Coast Pharmaceutical Works Ltd, Gota, Ahmedabad, Glutathione (GSH), 1-chloro-2, 4-dinitrobenzene (CDNB), thiobarbituric acid (TBA), 5′,5′-dithio-bis-2-nitrobenzoic acid (DTNB), hydrogen peroxide and epinephrine were purchased from Sigma® Chemical Company (London, UK). Alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma glutamyl transferase (GGT) and lactate dehydrogenase (LDH) assay kits were obtained from RANDOX® Laboratories Ltd., Antrim, UK. All other chemicals and reagent were of analytical grade and were obtained from British Drug House Poole UK.
2.3 Experimental animals
Male Wistar rats weighing between 160 and 180 g were used in this study. The rats were acclimatized and housed in wire-meshed cages at room temperature and under controlled light cycle (12 h light:dark) in the research animal house. The commercial rat chow and clean tap water were supplied to the rats ad libitum. The experimental study was conducted without anesthesia and the protocol conformed to National Institute of Health for laboratory animal care and use guidelines (National Research Council, Washington, USA) [18].
2.4 Drug treatments and animal grouping
The simple randomized design was employed in this study. Thirty-six male Wistar strain of albino rats were randomized into six groups of 6 rats each in separate cages using a randomization table. The groupings were determined by the cyclophosphamide alkylating agent and E. gracilis extract used. Group A animals were used as control and received distilled water. Group B animals (AMEG 200) were orally and daily administered with 200 mg/kg.bw AMEG for 7 days. Group C animals (AMEG 400) were orally and daily administered with 400 mg/kgBW AMEG for 7 days. Group D animals (CYP alone) were treated with cyclophosphamide (CYP) only (the dosage equivalent to 2 mg/kg body weight of cyclophosphamide). Group E animals (CYP + AMEG 200) were co-treated with daily administration of AMEG 200 (equivalent to 200 mg/kg body weight of AMEG) and CYP (2 mg/kg b.w.) for 7 days. Group F animals (CYP + AMEG 400) were co-treated with daily administration of AMEG 400 (equivalent to 400 mg/kg body weight of AMEG) and CYP (2 mg/kg b.w.) for 7 days. The prepared extract and cyclophosphamide solution were administered to the animals orally and once daily for a period of 7 days (the normal duration of therapy in humans). The dose for CYP was arrived at based on recommended adult dose for leukemia treatment while the dose for extract was decided from the result of our pilot study. The animals were euthanized 24 h after the last treatment.
2.5 Collection of blood and liver samples
After 24 h of final administration and 2 h before sacrifice, the animals were administered intraperitoneally with 0.04% colchicine (1 mL/100 g body weight). The blood samples were collected from each animal through retro orbitals plexus into lithium heparinized tubes and then sacrificed. The bone marrows of femur bone were collected for the assessment of micronucleated polychromatic erythrocyte frequency and livers were also excised for determination of enzymatic and non-enzymatic antioxidants, lipid peroxidation estimated by quantitation of malondialdehyde and percentage DNA fragmentation.
2.6 Determination of plasma and liver protein content
The Biuret method of Gornall et al. [19] was used to determine protein concentration in the plasma and liver homogenate after which the protein concentration in the samples was extrapolated from the standard bovine serum albumin (BSA) curve.
2.7 Assay of biomarkers of hepatotoxicity
Biomarkers of hepatotoxicity such as activities of alanine aminotransferase (ALT) and aspartate aminotransferase (AST), gamma glutamyl transferase (GGT) and lactate dehydrogenase were assayed using Randox diagnostic kits based on the manufacturer’s procedure. Estimation of plasma ALT and AST activities were based on the principle described by Reltman and Frankel in 1957 [20]. GGT was determined by the principle described by Szasz [21]. LDH activity was determined based on the method of Cabaud and Wroblewski [22].
2.8 Assay for oxidative stress maker in the liver
Hepatic AOPP concentration was determined following the method highlighted by Witko et al. [23]. The degree of lipid peroxidation (LPO) in the post mitochondrial fraction of liver organ was determined as described by the method of Varshney and Kale [24]. The method entailed the reaction between malondialdehyde and thiobarbituric acid to give a stable pink chromophore with maximum absorption at 532 nm. Lipid peroxidation in nmole/mg protein was computed as:
where E532 is the molar extinction coefficient for MDA = 1.56 × 105 M−1 cm−1
2.9 Assay of hepatic antioxidant enzymes
The activity of superoxide dismutase (SOD) in liver tissue was determined following the procedure of Misra and Fridovich [25] by monitoring how auto-oxidation of epinephrine at alkaline medium pH 10.2 was being inhibited. The highlighted procedure of Singha [26] was followed to determine catalase activity in liver based on reduction of dichromate in acetic acid to chromic acetate when heated in the presence of hydrogen peroxide (H2O2). The chromic acetate produced was measured spectrophotometrically at 570 nm and the amount of H2O2 remaining was extrapolated from the standard curve for H2O2. Catalase activity in the sample was expressed as micromole of H2O2 consumed per min per mg protein. Glutathione peroxidase (GPx) activity was estimated by the procedure of Rotruck et al. [27]. To 0.5 mL of phosphate buffer (0.1 M, pH 7.4) in a test tube was added 0.1 mL of 10 mM NaN3, 0.2 mL of GSH (4 mM), 0.1 mL of 2.5 mM H2O2, and 0.5 mL of testicular fraction. The reaction mixture was incubated for at 37 °C for 3 min after which 0.5 mL of 10% trichloroacetic acid was added. The resulting mixture was centrifuged at 3000 rpm for 5 min. One milliliter of the supernatant obtained was reacted with 2 mL of 0.3 M K2HPO4 and 1 mL of 5′,5′-dithio-bis-2-nitrobenzoic acid and the absorbance read against a reagent blank at 412 nm. GPx activity is expressed as mg GSH/mg protein.
2.10 Percentage DNA fragmentation assay
The endonuclease cleavage of the end product of apoptosis was assessed. DNA was extracted from the homogenate and treated with diphenylamine (DPA). The chromophore formed was measured on the spectrophotometer at 620 nm. Briefly, the livers were homogenized in 10 volumes of Tris–EDTA buffer (TE) pH 8.0 containing Triton-X100. Homogenates were centrifuged at 27,000g for 20 min to separate the intact chromatin (pellet named A) from fragmented ones (supernatant named B). The pellet (A) was suspended in Tris–EDTA buffer (TE!) of pH 8.0 without Triton-X100. 0.5 mL aliquot of each sample (pellet and supernatant) was placed in separate test tubes and 1.5 mL of freshly prepared diphenylamine solution was added to each test tubes. Reaction mixture was incubated at 37 °C for 20 h. Absorbance of mixture was then measured at 620 nm.
Calculation Quantity of fragmented DNA was estimated by using the formula;
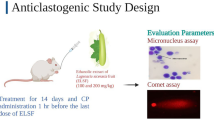
2.11 Micronucleus assay
Clastogenicity in cyclophosphamide treated rats was evaluated in the bone marrow of the rats employing the micronucleus assay techniques as described by Heddle and Salmone [28] with modification by Heddle et al. [29]. Briefly, bone marrow from femurs of rats was used for preparation of slides using the standard procedure by Matter and Schmid [30]. Bone marrow samples from both femur bones were processed to smear on slides. The slides were fixed in absolute methanol, allowed to air-dry and pre-treated with May-Grunewald stain solution and then stained in 5% Giemsa solution. The slides were blindly scored for micronucleated polychromatic erythrocytes under a light microscope.
2.12 Statistical analysis
Data are presented as the mean ± standard deviation (SD) of six replicates. Statistical significance was determined by one-way analysis of variance (ANOVA) followed by Duncan’s multiple comparison between control and treated rats in all groups using SigmaPlot® statistical package (Systat Software Inc., San Jose, CA, USA). P values less than 0.05 (P < 0.05) were considered statistically significant.
3 Results
3.1 Influence of Eulophia gracilis extract on cyclophosphamide-induced hepatic function
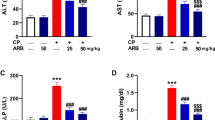
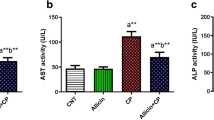
Protective effects of E. gracilis on Cyclophosphamide-induced changes in the activities of plasma alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma glutamyl transferase (GGT) and lactate dehydrogenase (LDH) in rats were shown in Fig. 1. The activities of ALT, AST and GGT in the plasma of cyclophosphamide-treated rats increased significantly by 45.34%, 76% and 94.14% respectively while the activity of LDH decreased significantly by 41.45% when compared to the values of control. However, AMEG co-treatment significantly ameliorated the cyclophosphamide-induced changes in the plasma activities of ALT, AST, GGT and LDH.
3.2 Protective effects of Eulophia gracilis extract on cyclophosphamide-induced changes in the activities of hepatic enzymatic antioxidants
The protection by E. gracilis on cyclophosphamide-induced toxicity on superoxide dismutase (SOD), catalase and glutathione peroxidase (GPx) activities in rat liver were represented in Fig. 2. Hepatic SOD, catalase and GPx activity were significantly reduced by 39.49%, 34.87% and 18.96% in group treated with cyclophosphamide as compared with control. However, treating with AMEG significantly alleviated the reduced activities of these enzymatic antioxidants in liver.
3.3 Protective effects of Eulophia gracilis on cyclophosphamide-induced changes in the levels of oxidative stress biomarkers
Figure 3 shows the effect of E. gracilis on Cyclophosphamide-induced toxicity on lipid peroxidation and protein oxidation in rats. Malondialdehyde level which is one of the lipid peroxidation products was significantly increased by 61.05% while there was also an elevation in the concentration of AOPPs in liver homogenate by 32.03% in the group exposed to cyclophosphamide when compared with the control (p < 0.05). However, treating animals with AMEG attenuated this increase in MDA and AOPPs level when compared to cyclophosphamide alone group.
Protective effects of Eulophia gracilis on cyclophosphamide induced changes in the levels of hepatic malondialdehyde (MDA) and advanced oxidation protein products (AOPPs) in rats. Data are presented as mean SD for six rats in group. *Significantly different from control, #significantly different from CYP at p < 0.05
3.4 Effect of Eulophia gracilis extract on cyclophosphamide-induced genotoxicity in rats
Figure 4 shows the effects of co-treating animals with AMEG on induction of percentage DNA fragmentation and induction of bone marrow micronucleus on rats treated with cyclophosphamide. The reduction in liver redox status induced by cyclophosphamide intoxication was followed by elevated level of percentage DNA fragmentation in liver tissue by 32.97% relative to control as well increased frequency of formation of micronucleated polychromatic erythrocytes in rats administered with cyclophosphamide. Treatment with AMEG attenuated the increase in hepatic percentage DNA fragmentation and inhibited the mPCEs induction by cyclophosphamide when compared with cyclophosphamide group.
Effect of Eulophia gracilis extract on formation of micronucleus in polychromatic erythrocytes in bone marrow and hepatic percentage DNA fragmentation in Cyclophosphamide treated rats. Data are presented as mean SD for six rats in group. *Significantly different from control, #significantly different from CYP at p < 0.05
4 Discussion
It has been documented that most synthetic chemotherapeutic agents, especially cyclophosphamide, mediated their toxic effects by generating reactive oxygen species [31]. It was recently shown that administration of cyclophosphamide could damage liver and kidney cells [3, 32]. Reports from several studies reveal the protective potential of medicinal plants on toxicity of several synthetic anticancer agents [32]. This may be due to antioxidant potential of bioactive compounds in medicinal plants. Thus, this study focuses on protective effect of E. gracilis against cyclophosphamide-induced oxidative damage to liver and kidney organ in rats.
In order to ascertain the protective effect of E. gracilis in cyclophosphamide-induced renal injury, markers of nephrotoxicity were evaluated. Plasma creatinine and urea values are established markers of kidney function in human being and animal subjects [33, 34]. Both of them are metabolic products filtered freely during glomerular filtration of kidney from circulation and their increased levels in plasma indicate altered renal function [35]. Data generated in this study suggested that cyclophosphamide altered renal function resulting to accumulation of these biomarkers in the rat plasma. The increased urea and creatinine contents observed in this study is consistent with a previous report on cyclophosphamide in rats [3]. However, the results of this study showed that the methanolic extract of E. gracilis offered protection to kidneys against injury caused by cyclophosphamide causing a reversal in levels of urea and creatinine raised by cyclophosphamide.
Chemotherapy-related liver dysfunction may be a result of different mechanisms such as generation of reactive oxygen species during drug metabolism that form adduct with biomolecule, inhibition of mitochondrial activity with accumulated lipid peroxidation and reactive oxygen species, disruption of biological membrane, generation of immunologic response and inflammation. Administration of cyclophosphamide causes different types aberration in liver ranging from destruction of membrane architecture, alteration in biochemical indices to perturbation of antioxidant status. In general, oxidative stress resulted from an imbalance between oxidant and antioxidant status is known to be a crucial factor responsible for disruption of the health of biological systems [36]. Therefore, to treat liver diseases, numerous researches have emphasized the importance of compounds with antioxidant potential that can inhibit liver injuries by scavenging free radicals [37, 38].
Typically, the activities of ALT and AST are commonly used to assess hepatic function where AST is a proven sensitive indicator of mitochondrial problem especially in centrilobular area of the liver and ALT activity is more precise in monitoring hepatocellular status. The extent of the damage in liver is assessed by the level of these cytoplasmic transaminases released into circulation [39] and increase in the activities of these cytoplasmic enzymes are therefore considered as indicators of hepatic disorder [36]. Plasma levels of GGT constitute a reliable marker of hepatotoxicity that is specifically associated with the cell membrane [40]. It is increased in pathological state associated with GGT leakage or overproduction and or hepatobiliary damage [41]. In this study, the activities of AST, ALT with GGT in plasma were increased following the administration of cyclophosphamide which may be due to hepatic damage leading to increased rate of synthesis and or release of functional enzymes from biological membranes [42]. Elevation in the activities of these enzymes is an indication of hepatic lesion or histoarchitecture disruption as observed in the liver of rats exclusively administered with cyclophosphamide in Fig. 5D. Moreover, Lactate dehydrogenase (LDH) is a tetrameric oxidoreductase that anaerobically catalyzed the conversion of pyruvic acid to lactic acid and vice versa [43]. The estimation of LDH in addition to basic liver function tests like activities of ALT, AST and ALP increase the accuracy of diagnosis of liver diseases. The ALT-LDH index predicts the prognosis of acute liver injury at their early stages and differentiate typhoid hepatitis from acute viral hepatitis [44]. Estimation of the amount of LDH production in hepatocytes based on biopsy examination was used to detect acute liver failure [45]. Therefore, the increase in LDH activity following the administration of cyclophosphamide confirmed the hepatotoxic effect of cyclophosphamide in this present work. However, these increases in LDH activity and transaminases were significantly reduced by treatment with E. gracilis. It has been shown that activities of serum aminotransferases return to normalcy as hepatocytes regenerate and as hepatic parenchyma heal up which corresponded to histopathological result obtained in Fig. 5 [46]. Therefore, these results suggest that E. gracilis prevented cyclophosphamide-induced hepatotoxicity.
Photomicrograph (× 400) showing Protective role of Eulophia gracilis extract on Cyclophosphamide-induced Damages on Architecture of Rats liver (Eosin & HX Stained). A Distilled water only. B 200 mg/kg AMEG only. C 400 mg/kg AMEG only. D CYP alone. E 200 mg/kg AMEG + CYP. F 400 mg/kg AMEG + CYP. A No visible lesion. B No visible lesion. C There is a mild diffuse vacuolar degeneration of hepatocytes. D There is a severe portal congestion, with moderate periportal cellular infiltration (black arrow). E No visible lesion is observed. F No visible lesion observed
Reports have established an association between oxidative stress and hepatic injuries [47, 48]. Most alkylating agents especially Cyclophosphamide possessed pro-oxidant character and therefore generated reactive oxygen species (ROS) that depleted cellular thiols and antioxidant enzymes [49, 50]. The result showed that cyclophosphamide caused significant reduction in Glutathione peroxidase (GPx), catalase (CAT) and superoxide dismutase (SOD) with a concomitant lipid peroxidation increase. SOD enzyme catalyzed dismutation process that converted superoxide ion to hydrogen peroxide (H2O2) that was removed by CAT through conversion to water and oxygen [51]. A reduced SOD and CAT activities in this study after cyclophosphamide administration might be due to oxidant activity of cyclophosphamide metabolites. In this study, co-treatment of cyclophosphamide-dosed animals with extract of E. gracilis improved the overall CAT and SOD activities in the liver of rat. The observation may be supported by the presence phenolics and flavonoids content in the plant that contributed to antioxidant activities of some Eulophia species [52].
AOPPs are dityrosine-containing cross-linked protein products used for evaluation of the degree of protein damage mediated by oxidant [23]. AOPPs are early marker of oxidized proteins that has prolong circulation periods in the blood as it takes hours and days to be degraded by cells. It is a considered marker for monitoring oxidative stress in drug-induced hepatotoxicity when it is determined in the hepatic tissue [53]. This is because generated intracellularly oxidized proteins is associated with disrupted balance in pro-oxidant and antioxidant ratio [54]. Sulfhydryl was sensitive group to oxidative damage and its diminution was associated with pathological states like ischemia insult [55]. Reduced levels of protein thiols in earlier reports correlated negatively with the levels of AOPPs [56]. The significantly increased hepatic AOPP level observed in cyclophosphamide-exposed animal as compared to control present study suggested induction of oxidative stress in the liver that subsequently damaged the tissue proteins. However, coadministration of E. gracilis extract reduced AOPP content significantly in extract co-treated group when compared to cyclophosphamide alone treated group. This shows that the extract may improve hepatic antioxidant activity by preserving its surfydryl protein content.
The involvement of lipid peroxidation in DNA damage has been reported asperoxidation products regularly interact with DNA causing oxidative DNA damage [57]. This therefore suggested a link between genotoxicity and generation of free radicals [58]. The potential of extract of Eulophia to prevent cyclophosphamide genotoxicity using induction of micronucleated polychromatic erythrocytes and percentage DNA fragments in liver as biomarkers was investigated. The genotoxicity and antigenotoxicity of substances are commonly assessed using Micronucleus assay. In the current study, exposure with cyclophosphamide increased the liver percentage DNA fragmentation of rats and their frequency of occurrence of polychromatic micronucleated cells in bone marrow, which suggested induction of chromosomal damage and DNA strand breaks. This corroborated earlier finding that Cyclophosphamide induced chromosomal aberrations and also DNA damage through the active carbonium ions generation [59]. More specifically it was earlier reported that percentage DNA breakage in the cells increased with increasing dose of cyclophosphamide [60]. More importantly, this study indicated that the plant extract was potent against cyclophosphamide–induced genotoxicity as depicted by the reduction in percentage DNA fragmentation with lowered mPCEs frequency in group co-treated with E. gracilis extract. The genoprotection offered by the plant extract may due to its antioxidant capability.
In conclusion, pseudobulb extract of E. gracilis offered protective potential against oxidative stress-mediated toxic side effect of cyclophosphamide on rat liver.
Data availability
All the data are included in the manuscript.
References
Toale KM, Johnson TN, Ma MQ (2016) Chemotherapy-induced toxicities. In: Todd K, Thomas Jr C (eds) Oncologic emergency medicine. Springer, Cham, pp 381–406. https://doi.org/10.1007/978-3-319-26387-8_33
Clavo B, Rodríguez-Esparragón F, Rodríguez-Abreu D, Martínez-Sánchez G, Llontop P, Aguiar-Bujanda D, Fernández-Pérez L, Santana-Rodríguez N (2019) Modulation of oxidative stress by ozone therapy in the prevention and treatment of chemotherapy-induced toxicity: review and prospects. Antioxidants (Basel) 8(12):588. https://doi.org/10.3390/antiox8120588. (PMID:31779159;PMCID:PMC6943601)
Olayinka ET, Ore A, Ola OS, Adeyemo OA (2015) Ameliorative effect of gallic acid on cyclophosphamide-induced oxidative injury and hepatic dysfunction in rats. Med Sci 3:78–92. https://doi.org/10.3390/medsci3030078
Ahlmann M, Hempel G (2016) The effect of cyclophosphamide on the immune system: implications for clinical cancer therapy. Cancer Chemother Pharmacol 78:661–671. https://doi.org/10.1007/s00280-016-3152-1
Brock N, Wilmanns H (1958) Effect of a cyclic nitrogen mustard-phosphamidester on experimentally induced tumors in rats, chemotherapeutic effect and pharmacological properties of B518 ASTA [German]. Dtsch Med Wochenschr 83:453–458
Emadi A, Jones RJ, Brodsky RA (2009) Cyclophosphamide and cancer: golden anniversary. Nat Rev Clin Oncol 6:638–647
Baxter Oncology. German Product Summary Endoxan®. http://www.fachinfo.de/suche/fi/000728. Accessed Jan 2015
IARC (1987) Benzene-IARC monographs on the evaluation of carcinogenic risks to humans, vol 1-42. International Agency for Research on Cancer, Lyon
Patel JM (1987) Stimulation of cyclophosphamide-induced pulmonary microsomal lipid peroxidation by oxygen. Toxicology 45:79–91
Hales BF (1982) Comparison of the mutagenicity and teratogenicity of cyclophosphamide and its active metabolites, 4-hydroxycyclophosphamide, phosphoramide mustard, and acrolein. Cancer Res 42:3016–3021
Dhamija E, Meena P, Ramalingam V, Sahoo R, Rastogi S, Thulkar S (2020) Chemotherapy-induced pulmonary complications in cancer: significance of clinicoradiological correlation. Indian J Radiol Imaging 30(1):20–26
Wang JY, Prorok G, Vaughan WP (1993) Cytotoxicity, DNA cross-linking, and DNA single-strand breaks induced by cyclophosphamide in a rat leukemia in vivo. Cancer Chemother Pharmacol 31:381–386
Johnson IT (2007) Phytochemicals and cancer. Proc Nutr Soc 66:207–215
Sawadogo WR, Schumacher M, Teiten MH, Dicato M, Diederich M (2012) Traditional West African pharmacopeia, plants and derived compounds for cancer therapy. Biochem Pharmacol 84:1225–1240
Hossain MM (2011) Therapeutic orchids: traditional uses and recent advances: an overview. Fitoterapia 82:102–140
Besra RC, Majhee L, Sharma J (2011) Evaluation of phytochemical, antioxidant and hepatoprotective activity of tuber of Geodorum laxiflorum Griff. J Pharmacol Toxicol 6:610–623
Ola SO (2017) Preliminary proximate analysis, chemical composition and phytoconstituents of Eulophia gracilis orchid. Int J Sci Basic Appl Res 36(8):215–222
Research Council N (2011) Guide for the care and use of laboratory animals, 8th edn. National Research; The National Academies Press, Washington, DC
Gornall AG, Bardawill CJ, David MM (1949) Determination of serum proteins by means of biuret reaction. J Biol Chem 177:751–766
Reltman S, Frankel SA (1957) Colorimetric method for the determination of serum ALT and AST. Am J Clin Pathol 28:56–63
Szasz GA (1969) Kinetic photometric method for serum gamma-glutamyl transpeptidase. Clin Chem 15:124–136
Cabaud PG, Wroblewski F (1958) Colorimetric measurement of lactic dehydrogenase activity of body fluids. Am J Clin Pathol 30:234–236
Witko-Sarsat V, Friedlander M, Capeillere-Blandin C (1996) Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int 49(5):1304–1313
Varshney R, Kale RK (1990) Effect of calmodulin antagonist on radiation induced lipid peroxidation in microsomes. Int J Radiat Biol 58:733–743
Misra HP, Fridovich I (1972) The role of superoxide anion in the auto-oxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247:3170–3175
Singha AK (1972) Colorimetric assay of catalase. Anal Biochem 47:389–394
Rotruck JT, Pope AL, Ganther HE (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179:588–590
Heddle JA, Salmone MF (1981) The micronucleus assay I: In vivo. In: Stich HF, San RHC (eds) Topics in environmental physiology and medicine. Short term tests for chemical carcinogens. Springer, New York, pp 243–249
Heddle JA, Sudharsan RA, Krepinsky AB (1981) The micronucleus assay II: in vitro. In: Stich HF, San RHC (eds) Topics in environmental physiology and medicine. Short term tests for chemical carcinogens. Springer, New York, pp 250–254
Matter B, Schmid W (1971) Bone marrow toxicity. Mutat Res 12:417–425
Sangeetha P, Das UN, Koratkar R, Suryaprabha P (1990) Increase in free radical generation and lipid peroxidation following chemotherapy in patients with cancer. Free Radic Biol Med 8:15–19
Devi HP, Mazumder PB (2016) Methanolic extract of Curcuma caesia Roxb. prevents the toxicity caused by cyclophosphamide to bone marrow cells, liver and kidney of mice. Pharmacogn Res 8(1):43–49
Ferguson MA, Waikar SS (2012) Established and emerging markers of kidney function. Clin Chem 58(4):680–689
Simsek A, Tugcu V, Tasci AI (2013) New biomarkers for the quick detection of acute kidney injury. ISRN Nephrol. https://doi.org/10.5402/2013/394582
George GS, Wakasi ME, Egoro E (2014) Creatinine and urea levels as critical markers in end-stage renal failure. Res Rev J Med Health Sci 3:41–44
Wang Y, Tang C, Zhang H (2015) Hepatoprotective effects of kaempferol 3-O-rutinoside and kaempferol 3-O-glucoside from Carthamus tinctorius L. on CCl4-induced oxidative liver injury in mice. J Food Drug Anal 23(2):310e7
Casas-Grajales S, Muriel P (2015) Antioxidants in liver health. World J Gastrointest Pharmacol Ther 6(3):59–72
Ola OS, Sofolahan TA (2021) A monoterpene antioxidant, linalool, mitigates benzene-induced oxidative toxicities on hematology and liver of male rats. Egypt J Basic Appl Sci 8(1):39–53
Janbaz KH, Saeed SA, Gilani AH (2002) Protective effect of rutin on paracetamol and CCl4 induced hepatotoxicity in rodents. Fitoterapia 73:557–563
Boone L, Meyer D, Cusick P, Ennulat D, Bolliger AP, Everds N, Meador V, Elliott G, Honor D, Bounous D (2005) Selection and interpretation of clinical pathology indicators of hepatic injury in preclinical studies. Vet Clin Pathol 34:182–188
Ramaiah SK (2007) A toxicologist guide to the diagnostic interpretation of hepatic biochemical parameters. Food Chem Toxicol 45:1551–1557
Pari L, Prasath A (2008) Efficacy of caffeic acid in preventing nickel induced oxidative damage in liver of rats. Chem Biol Interact 173(2):77–83
Danner BC, Didlis VN, Wiemeyer S, Stojaovic T, Kitz J, Emmert A (2010) Long-term survival is linked to serum LDH and partly to tumor LDH-5 in NSCLC. Anticancer Res 30:1347–1351
Balasubramanian S, Kaarthigeyan K, Srinivas S, Rajeswari R (2010) Serum ALT: LDH ratio in typhoid fever and acute viral hepatitis. Indian Pediatr 47:339–341
Kotoh K, Kato M, Kohjima M, Tanaka M, Miyazaki M, Nakamura K (2011) Lactate dehydrogenase production in hepatocytes is increased at an early stage of acute liver failure. Exp Ther Med 2:195–199
Thabrew M, Joice P, Rajatissa W (1987) A comparative study of the efficacy of Pavetta indica and Osbeckia octandra in the treatment of liver dysfunction. Planta Med 53:239–241
Yamamoto T, Kikkawa R, Yamada H, Horii I (2005) Identification of oxidative stress related proteins for predictive screening of hepatotoxicity using a proteomic approach. J Toxicol Sci 30:213–227
Oh JM, Jung YS, Jeon BS, Yoon BII, Lee KS, Kim BH, Oh SJ, Kim SK (2012) Evaluation of hepatotoxicity and oxidative stress in rats treated with tert-butyl hydroperoxide. Food Chem Toxicol 50:1215–1221
King PD, Perry MC (2001) Hepatotoxicity of chemotherapy. Oncolgist 6:162–176
Yost GS, Horstman MG, Walily AF, Gordon WP, Nelson SD (1985) Procarbazine spermatogenesis toxicity: deuterium isotope effect point to regioselective metabolism in mice. Toxicol Appl Pharmacol 80:316–322
Valko M, Leibfritz D, Moncol J, Cronin MTD, Mazur M, Telser J (2007) Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol 39(1):44–84
Dawande V, Rajaram Gurav R (2017) Total phenolics, flavonoids content and antioxidant activities of some Eulophia species. J Med Plants Stud 5(2):106–111
Afolabi OK, Oyewo EB (2014) Effects of ciprofloxacin and levofloxacin administration on some oxidative stress markers in the rat. World Acad Sci Eng Technol Int J Biol Vet Agric Food Eng 8(1):38–42
Penna C, Mancardi D, Rastaldo R (2009) Cardioprotection: a radical view: free radicals in pre and postconditioning. Biochim Biophys Acta 1787:781–793
Soszynski M, Bartosz G (1997) Decrease in accessible thiols as an index of oxidative damage to membrane proteins. Free Rad Biol Med 23:463–469
Kolagal V, Karanam SA, Dharmavarapu PK, Souza RD, Upadhya S, Kumar V (2010) Determination of oxidative stress marker and their importance in early diagnosis of uremia-related complications. Indian J Nephrol 19(1):8–12
Agarwal S, Draper H (1992) Isolation of a malondialdehyde-deoxyguanosine adduct from rat liver DNA. Free Rad Biol Med 13:695–699
Mitchell L, Adzick N, Melchionne J (2004) Spina bifida. Lancet 364:1885–1895
Schneider EL, Sternberg M, Tice RR (1977) In vivo analysis of cellular replications. Proc Natl Acad Sci (USA) 74:2041–2044
Amit KD, Kalpana A, Anita M, Diptis S (1995) Inhibition by capsaicin against cyclophosphamide-induced clastogenicity and DNA damage in mice. Mutat Res 335:253–258
Funding
This research was carried out without any specific grant from a funding agency in the public, commercial, individual or not-for-profit sector.
Author information
Authors and Affiliations
Contributions
OSO (OSO) designed the study and OAO (OAO) supervised the study. Benchwork and data collection were carried out by OSO while all the authors performed data analysis. The first draft of the manuscript was written by OSO and was vetted, commented on and corrected by OAO. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There is no potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ola, O.S., Odunola, O.A. Eulophia gracilis pseudobulb extract mitigates cyclophosphamide-induced genotoxicity and oxidative stress on murine hepatic tissue. J.Umm Al-Qura Univ. Appll. Sci. 9, 426–435 (2023). https://doi.org/10.1007/s43994-023-00050-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43994-023-00050-9