Abstract
Aging mammalian results in impaired bio-functions and neurological disorders. The current study investigated whether whey protein (WP) syrup supplementation may improve age-related changes in diseased brain indicators like tau protein, β-amyloid and α-amylase. The study was carried out in conjunction with immunohistochemistry, histology, and flow cytometry of apoptosis. At the ages of 8 and 30 months, Wistar albino rats (Rattus novergicus) were divided into four groups (n = 8; G1; 8 months old rats; G2, 8 months old rats supplemented WP; G3, 30 months old rats; G4, 30 months old rats supplemented WP), with or without whey syrup administration. For 2 months, oral whey supplementation in 2 mL/kg doses is given twice a day every 12 h. Rats were sacrificed, and their brains were subjected to biochemical, histological, immunohistochemistry, and flow cytometric investigations. Aged rats had lower levels of superoxide dismutase (SOD), adenosine triphosphate (ATP), serotonin (5-HT), and dopamine (DA). These observations were parallel with increased inflammatory markers [tumor necrosis factor α- and 5-lipoxygenase (5-LO)], lipid peroxidation products (MDA), as well as apoptotic marker caspase-3, annexin-v, tau protein, β-amyloid, and α-amylase. Whey administration to aged rats reduced inflammatory and oxidative stress markers as well as improved neurotransmitters, tau protein, β-amyloid, and α-amylase. The advantages of supplementation were validated by improved histology and immunohistochemistry in aged rats’ cerebrum, cerebellum, and hippocampus. In addition, apoptosis was reduced, according to flow cytometry analysis of annexin-v. In conclusion, WP contains amino acids and bioactive compounds that could decrease brain oxidative stress and restore normal metabolic function. Furthermore, increased antioxidant defense and DA and 5-HT neurotransmitters, while decreasing brain tau protein and β-amyloid, were associated with better histology in aged rats’ cerebrum, cerebellum, and hippocampus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Aging is characterized by arterial stiffness, abnormal autonomic dysregulation, and damage to the blood–brain barriers. Cortical and subcortical micro-infarcts, scattered white matter disorders have all been connected to these processes. These injuries resulted in demyelination and axonal damage [1,2,3,4]. In addition, the ischemic damaging of the neuronal cells and the development of cerebral micro-hemorrhages of venous origin facilitate neurodegenerative disorders and dementia [5, 6]. Aging also increased transactive response DNA-binding protein (TDP-43) [7], which increases glial and neuronal cell inflammation. The inflammation resulted in the accumulation of pro-inflammatory microRNA cytokines targeting genes involved in neuronal apoptosis [8]. In addition, old age showed irregular glycolytic enzyme activity that impede synaptic function and trigger neuronal cell loss [9]. Besides, old age might lead to heme degradation because of heme-oxygenase-1 regulation, which causes injury to the mitochondrial membrane in neuronal cell [10]. Parkinson's disease, Alzheimer's disease, and glaucoma are common disorders connected with aging. Glaucoma manifests oxidative stress assessed by mitochondrial and endoplasmic reticulum dysfunction and endothelial cell damage. The glaucomatous patient exhibited an abnormal aggregation of β-amyloid or tau protein in the retinal ganglion cell [11].
Whey protein (WP) is a bio-waste from cheese processing. Whey is a rich source of amino acids and plays a vital role in treating type 2 diabetes and glucose homeostasis [12,13,14,15]. Mainly bovine serum albumin and lactoferrin in WP are the main components of milk [16, 17]. These nutrients are critical constituents of the human diet [18] and found commonly in infant formula [19]. It was shown that animals fed on a diet containing milk fat globule, lactoferrin, and a polydextrose/galactooligosaccharide probiotic led to a high increase in dendritic spine density in hippocampal dentate gyrus neurons [20].
Supplementation (1 g/kg b.w./day) of WP improved antioxidant potential and decreased free radicals and protein carbonyl [21]. Administration of WP to animals fed a diet containing a high phytoestrogen for ten weeks improved estradiol levels, T4, and glucose homeostasis [22]. Female mice (C57BL/6 J) fed (100 g WP/L water) with WP for 12 weeks showed activated brain function with increased levels of cytochromes [23].
Diabetes mellitus (DM) could damage the brain through oxidative stress [24]. The WP treatment alleviated DM progression by increasing blood flow, insulin secretion, cerebral oxygen, and decreased blood sugar levels [25]. The consumption of fermented dairy products alleviated cognitive function and enhanced symptoms of Alzheimer’s in mouse models. Ano et al. [26] reported that tryptophan–tyrosine (Trp–Tyr)-dipeptide (WY)-containing peptides increased dopamine level, while decreasing monoamine oxidase-β activities in brain tissues. The oxidative stress implicated in injured neurons was reduced in CD1 mice administered Immunocal® (WP). The reduction alleviated axonal demyelination and brain-derived neurotrophic factor [27]. Patients with Parkinson’s disease who used WP supplements had lower plasma homocysteine and higher plasma glutathione level, upregulation of branched and essential amino acids [28]. Sarcopenic older adults given WP, essential amino acids (containing leucine), and vitamin D exhibited increased muscle strength with a handgrip dynamometer, as well as enhanced health conditions based on blood biochemical indicators [29].
The goal of the current research was to investigate the aid of whey supplementation effects on neurodegenerative markers and brain redox state, apoptosis and increase the antioxidant defense parallel with improving histology and immunohistochemistry of brain regions during the aging process.
2 Materials and methods
2.1 Characterization of whey syrup
Fresh bovine whey was obtained from the Dairy Product Laboratory (Faculty of Agriculture, Mansoura University, Egypt). WP was analyzed according to Parris and Baginskla [30]. The method used reversed-phase HPLC for denaturation of WP (Whey from bovine milk, Sigma-Aldrich Chemie, Taufkirchen, Germany), which is precipitated at pH 4.6. Un-denatured WP's absorbance and nitrogen concentration were compared to known WP standard to quantify WP nitrogen. Lactose was quantified according to Essig and Kleyn [31]. Lactose is hydrolyzed to β-galactose and glucose in the presence of β-galactosidase. In the presence of β-galactose dehydrogenase, β-galactose is oxidized by NAD to galactonic acid. Reduced nicotinamide-adenine dinucleotide resulted in stoichiometric with lactose amount determined at 340 nm.
The antioxidant content was determined according to Lim and Quah [32]. One mL of a methanol solution (100 µM) of 2,2-diphenyl-1-picrylhydrazyl (DPPH·) and the different concentrations of WP samples (0–30 mg/mL) were mixed. The mixtures were incubated for 20 min, and the absorbance was detected at 517 nm. The data expressed as radical scavenging activity (%).
According to Karami et al. [33], the number of lactobacilli colonies in WP was estimated. This was performed by incubating 2 mL of whey in a flask containing MRS broth media containing 100 mL distilled water at 37 °C. After 24 h, 100 μL enriched sample was spread over MRS agar and cultured in an anaerobic environment for 48 h at 37 °C. The number of bacterial colonies was counted.
2.2 Animal grouping and investigation
Male adult Wistar albino rats (Rattus novergicus) (8 months old, n = 16, weight 200 ± 10 g) and senile rats (30 months old, n = 16, weight 400 ± 10 g) were obtained from the Breeding Lab (Ministry of Health and Population, Egypt). Adult (8 months old) and old rats (30 months old) were divided into four groups (n = 8; G1; 8 months old rats; G2, 8 months old rats supplemented whey; G3, 30 months old rats; G4, 30 months old rats supplemented whey), with or without whey syrup administration. For 2 months, oral whey supplementation was administered twice a day in 2 mL/kg of whey syrup (every 12 h) [34, 35]. The animals were housed in an aerated environment with a 12 h light/dark cycle and 180–200 lx light intensity [34, 35]. Free access to diet and water are ad libitum. At the end of treatment, the animals were starved overnight and euthanized with halothane (2-bromo-2-chloro-1,1,1-trifluoroethane), followed by cervical dislocation and dissection. The blood was collected from the heart, coagulated, centrifuged, and serum separated and stored in the refrigerator. The brain was dissected and divided into two halves, one of which was preserved in the refrigerator, and the other was fixed in phosphate-buffered formalin (10%, pH 7.4) for immunohistochemical and histological studies.
The Experimental Animal Ethical Committee (Faculty of Science, Mansoura University, Egypt) approved the experiment (Approved Statement No. RZ19004).
2.3 Body and brain weight
The whole absolute body and brain weights (g) were recorded. The relative weights are calculated by dividing the brain weight on the body weight and multiplying by 100, followed by statistical analysis. With or without whey supplementation, absolute and relative brain weights were determined in the animals.
2.4 Superoxide dismutase and lipid peroxidation product (MDA)
Superoxide dismutase (SOD) activity was determined by incubating 100 μL brain supernatant samples in 100 μL nitroblue tetrazolium, 100 μL xanthine oxidase, and 3100 μL phosphate buffer solution (PBS) at 30 °C for 30 min. The generated color was spectrophotometrically recorded at 500–600 nm [36]. The reaction of malondialdehyde with thiobarbituric acid-producing a thiobarbituric acid reactive substance (TBARS) was detected at 532 nm [37].
2.5 Brain dopamine (DA) and serotonin (5-HT)
According to the manufacturer’s instructions, the assayed neurotransmitters were determined using the ELISA Kit of CUSABIO TECHNOLOGY (Houston, USA). The ELISA Kit was used to assay dopamine (DA). Serotonin (5-HT) was determined using Kit-Cat Nu. E-El-0033.
2.6 TNF-α, 5-lipoxygenase and caspase 3
Caspase-3 (Catalog No. CSB-E08857r) and TNF-α (Catalog No. CSB-e11987r) were measured according to the instruction of the ELISA kit from CUSABIO TECHNOLOGY (Houston, USA). In addition, rat My BioSource assayed 5-lipoxygenase (5-LO) using ELSA kit (Catalog. No. MBS722629). The competitive inhibitory response method involved labeling biotin and either TNF-α or 5-LO, then incubating with horseradish peroxidase-conjugated with avidin. The absorbance was calculated at 540 nm with the aid of the standard curve.
2.7 Neurodegenerative markers
ELISA Kit (CUSABIO TECHNOLOGY) was used for the determination of brain tau protein (Catalog No. CSB-E13729r), α-amylase (CSB-EL001689RA), acetylcholinesterase (AChe) (CSB-E11304r), β-amyloid peptide (Aβ) (CSB-E-10786r), brain natriuretic peptide (CSB-E07972r) and nerve growth factor (CSB-E04685r). Adenosine triphosphate (ATP) was determined using ELISA Kit (My Biosource Comp., MBS723034). Brain xanthine oxidase activity (Catalog No. K710-100) and creatine kinase (Catalog No. K777-100) were determined by Bio-vision incorporated (Milpitas boulevard, GA, 5USA).
2.8 Histopathological investigation
Brain specimens were fixed in phosphate-buffered formalin (10%, pH 7.4), dehydrated in ascending grades of ethanol, cleared in toluene, and mounted in molten pararplast (58–62 °C). The serial 5-μm thick histological sections were cut and stained with hematoxylin and eosin (H&E). To visualize the cerebellum, cerebrum, and hippocampus changes, sections were examined under light microscopy.
2.9 Immunohistochemistry of caspase-3 and synaptophysin
Serial 5-μm thick histological paraffin sections were cut and mounted onto super frost t plus glass slides (Fisher Thermo Scientific, Nepean, Canada). Tissue sections were processed for antigen retrieval by digestion in trypsin (0.05%, pH 7.8) at 37 °C for 15 min, then incubated against either mouse anti-synaptophysin (Thermo Fisher Scientific, Catalog MA5-14532) or caspase-3 (Catalog MA5-11516) overnight at 4 °C. These followed by treatment with a horseradish peroxidase streptavidin, then DAB plus Chromagen to detect the immunoactivity, and counterstained with Mayer hematoxylin. The negative control sections were incubated with non-immune rabbit serum (1%) instead of the caspase-3 and synaptophysin antibody. The brain regions were examined and photographed with a Leica BM5000 microscope (Leica Microsystems, Wetzlar, Germany). Image processing was carried out at 40 X objective and an Olympus digital camera fixed on an Olympus microscope with 1/2 X frame adaptor. Video Test Morphology Software analyzed the resulting images, and the percentage area was calculated and reported.
2.10 Flow cytometry assessments of annexin-v
The flow cytometric analysis of annexin-v was performed using V‐FITC/PI double staining method. First, brain tissue was lysed with tris–EDTA solution (pH 7.4) and fixed in 70% ethyl alcohol. After that, cells were washed with PBS, suspended at 0.1–0.3 × 106/mL, and stained with fluorescein isothiocyanate‐conjugated annexin‐v (annexin V‐FITC). Specimens were incubated at room temperature for 15 min and determined by Becton Dickinson Fac Scan Fluorescence-Activated Cell Analyzer (Becton Dickinson, CA, USA).
2.11 Statistical analysis
The results were presented as means ± standard deviations (SD). Statistical analysis was conducted using SPSS (Version 13) one-way ANOVA post hoc analysis for windows, comparing the adult and aged groups and WP supplementation. The significance is at p < 0.05.
3 Results
3.1 Whey characterization
The nutritional content of WP, including total protein, lactose content, total antioxidant activity, and lactobacilli content, is given in Table 1.
3.2 Absolute and relative brain weights
When compared to the non-supplemented group, whey syrup supplementation resulted in a non-significant increase in absolute brain weight and decreased relative brain weight in the 8 months old animals. Thus, whey syrup supplementation decreased the absolute brain weight of old age while the nonsignificant increase was in the relative brain weight (Fig. 1A–C).
Mean body weight (A), absolute brain weight (B), and relative brain weight (C) of aging rats with or without whey supplementation. Data represent the mean ± SD (n = 5). *Significant at p < 0.05. G1 8 months old rats, G2 8 months old rats supplemented whey, G3 30 months old rats, G4 30 months old rats supplemented whey
3.3 Serum neurodegenerative markers
The serum amounts of xanthine oxidoreductase (XOR), creatine kinase (CK), and acetylcholinesterase increased significantly in aged rats, with a percent of + 47.01, + 30.93 + 29.79, respectively. Brain natriuretic peptide (BNP) and nerve growth factor (NGF) decreased significantly with 19.4% and 20.6%, respectively. The assayed serum levels of aging rats were improved by whey supplementation. Levels of NGF, XOR, CK, BNP and AChE were + 14.3, − 18.4, − 15.3, + 14.7 and − 14.4%, respectively, following comparison of G3 and G4 (Table 2).
3.4 Brain superoxide dismutase and lipid peroxidation product (malondialdehyde)
The brain SOD of the adult group (G1) was 15.4, while it was decreased in the aged group (G3), which was 9.81 with a percentage of reduction of 36.5%. When compared to G1, G2 supplemented whey revealed a non-significant increase in SOD activity. However, it increased significantly in G4, reaching 12.7 to the percentage increase of + 30.2 compared to G3. There were no variations in brain MDA concentrations between G1 and G2 on the other side. Compared to G1 (4.66), a significant increase of MDA was detected in G3 (6.97) with a percentage increase of + 49.5%. On the other hand, whey supplementation decreased MDA content (5.78) in G4 with a percent reduction of − 17.0%. When comparing G4 to G1, the percent of increased MDA during treatment reached + 24.0%, which is half-folded of the G3 value (Table 3).
3.5 Brain neurotransmitters dopamine and serotonin
In G1 and G2, there were no differences in serotonin (5-HT), dopamine (DA), and adenosine triphosphate (ATP). However, DA, 5-HT, and ATP levels in the brain were considerably decreased in G3 during aging, reaching 6.78, 103.5, and 82.6, respectively, compared to 13.05, 147.3, and 117.6 in G1. The decrease percentages were − 48.2, − 29.5, and − 29.6%, respectively. Whey administration to the aged group (G4) enhanced the assayed neurotransmitters and ATP levels, and their percentages of reduction were still higher in DA at − 37.0. Meanwhile, 5-HT and ATP exhibited less decreased percent at − 10.3 and − 18.8%, respectively, compared to G1 (Table 3).
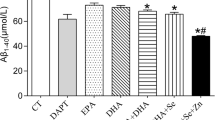
3.6 Brain tau protein, β-amyloid and α-amylase
Aged rats (G3) exhibited a significant increase of tau protein, β-amyloid, and α-amylase (218.8, 11.2, and 92.9, respectively) compared to 162.7, 6.83, and 9.75 in G1. The increased percentages were + 34.4, + 64.4, and + 853.6%, respectively. Whey syrup administration moderately enhanced the assayed parameters, but their levels were still above the normal ranges in G1. Their increase percentages reached + 13.4, + 21.3, and + 549.2%, respectively (Table 3).
3.7 Brain biomarkers of cell death and inflammation
From Table 3, 5-lipooxygenase, tumor necrosis-α, and caspase-3 were significantly higher in G2 at 8.68, 127.3, and 100.6 than in G1 (6.21, 67.4, and 6.28, respectively). The increased percentages were + 88.7, + 1502.5, and + 39.7%, respectively. Whey syrup supplementation caused minor alterations in G1. G4 exhibited moderate improvement in the brain levels of both caspase-3 and TNF-α, but 5-lipooxygenase levels were remained considerably higher than in G1. Their percentages of improvement were + 40.0, + 1141.5, and + 13.3%, respectively.
3.8 Histopathological observations
In adult groups with or without supplementation (G1 & G2), the cerebral external granular (OGL) outer ganglionic layer showed a dense distribution of pyramidal and stellate cells with centrally located nuclei (Fig. 2A, A1, B, B1). In the aged rats (G3), many neuroglial cells exhibited either chromatolysis (karyolysis) or clumping nuclear chromatin (pyknosis). Angiogenesis of the blood vessels seems to be widely spread throughout the cerebral tissue clarifying the diseased pathological feature. The glial cells seem to be grouped, manifesting inflammation of the brain tissues. Necrotic, edematous, and spongiform degenerated foci were comparatively increased (Fig. 2C, C1). Whey syrup supplementation enhanced these aged-related alterations (G4) (Fig. 2D, D1).
Photomicrographs of a sagittal histological section of the cerebrum (A–D, A1–D1), cerebellum (A2–D2), and hippocampus (A3–D3). A, A1, 8 months old cerebrum. B, B1, whey supplemented 8 months old. Note normal cerebral neurons. C, C1, cerebrum of old rat (G3) showing edematous lesions and fragile white matter (star) and dense aggregation of glial cells (arrowhead) infiltrated in between degenerated neuron. D, D1, whey supplemented old group showing less damaged neurons with vesicular nuclei. A2, Cerebellum of adult rats. B2, Whey supplemented cerebellum of the adult rat. Note normal molecular, Purkinje, and granular cells. C2, The cerebellum of old rat showing pyknotic of Purkinje cells with pyknotic nuclei and reduction of granular cells. D2, The cerebellum of old rats supplemented whey showing partial improvement of Purkinje cells and regenerated granular cells. A3, The hippocampus of the adult rat. B3, The hippocampus of adult rats, supplemented whey. Note the normal pattern of pyramidal cells [(PyL) and outer polymorphic layer (PL)]. C3, The hippocampus of old rat showing vacuolar degeneration of pyramidal layer (PyL). D3, Old rats supplemented whey showing moderate improvement in pyramidal cells
The cerebellar cortex of 8 months old and whey supplemented groups (G1 & G2) exhibited normal structures of the molecular (MCL), Purkinje cell layer (PCL), and the granular cell layer (GCL) (Fig. 2A2, B2). In the aged rat (G3), the Purkinje cells had either pyknotic or karyolysed nuclei embedded in necrotic spaces. In addition, the damaged granular cells were invaded by a large glomerular space (Fig. 2C2). Whey syrup supplementation to the aged group (G4) enhanced the cerebellar structure, particularly Purkinje cells (Fig. 2D2).
Histological investigations of the hippocampus of 8 months old rats with or without whey syrup supplementation (G1 & G2) revealed a well-defined pyramidal, polymorphic, and molecular layer. The pyramidal layer comprises small pyramidal cells that are densely packed together. Each cell had a large polygonal shape with rounded nuclei, prominent nucleoli, and scanty cytoplasm. The dentate gyrus makes up of small granule cells. The molecular layer has regular neuronal axons and dendrites distribution (Fig. 2A3, B3). The aged group (G3) had chromatolysis or grouping nuclear chromatin that resulted from apoptosis in pyramidal cells (Fig. 2C3). Whey syrup treatment impaired the progress of angiogenesis in cerebral tissues of aging rats and vanishing most of them. However, there were just a few spots of primitive angiogenesis (Fig. 2D3).
3.9 Immunohistochemistry of caspase-3 and synaptophysin
Cerebral neurons, cysteine-aspartic acid protease 3 (caspase-3), cerebellar Purkinje and granular cells, and pyramidal hippocampus cells displayed overexpression of the immunohistochemical reactions in aged rats (G3) with increased apoptotic cells (Fig. 3A2, B2, C2). Whey syrup supplementation decreased the immunohistochemical reaction in the G4 group (Fig. 3A3, B3, C3) compared to the adult group with or without whey supplementation compared to adult supplemented whey (Fig. 3A, B, C, A1, B1, C1). Image analysis revealed the increased intensity of the caspase-3 reaction in the aged group (G3) compared to that of the aged animals supplemented whey syrup (G4) or adult rats with or without whey syrup supplementation (G1 & G2) (Fig. 3B).
A Photomicrographs of sagittal histological sections of formalin-fixed immunostained with caspase-3 of the cerebrum (A–A3), cerebellum (B–B3), and hippocampus (C–C3). A, adult rat cerebrum. A1, Whey supplemented adult rat. Note negative immune reaction. A3, Aged rat showing increased immune reaction in neuronal cells. A3, Whey supplemented old rats showing a decreased immune reaction. B, B1, cerebellum of adult and whey supplemented group showing a negative immune reaction. B2, The cerebellum of aged rats showing a dense immune reaction. B3, Old cerebellum supplemented whey showing a decreased immune reaction. C, C1, Adult hippocampus, and whey supplemented group. C2, The hippocampus of aged rats showing a dense immune reaction. C3, The hippocampus of old rats supplemented whey, showing a decreased immune reaction. B Chart illustrating Image analysis of caspase 4 showing overexpression of caspase-3 in an aging group compared to adult and aging group supplemented whey syrup. Each result represent the mean ± SD (n = 5); asterisk means significant at p < 0.05. G1; 8 months old rats; G2, 8 months old rats supplemented whey; G3, 30 months old rats; G4, 30 months old rats supplemented whey
Imnunohistochemistry with synaptophysin revealed a decrease of the immune reaction in the synaptic axons of the cerebellum, cerebrum, and hippocampus of the aged group (G3) (Fig. 4A2, B2, C2) and increased in aged groups supplemented whey (Fig. 4A3, B3, C3), but less than that of the adult group with or without whey supplementation (Fig. 4A–C, A1–C1). In addition, the image analysis revealed the decreased intensity of the synaptophysin immune reaction in the aged group (G3) compared to that of G4 and an adult with or without whey syrup supplementation (Fig. 4A–B).
A Photomicrographs of sagittal histological sections of formalin-fixed immunostained with synaptophysin of the cerebrum (A–A3), cerebellum (B–B3), and hippocampus (C-C3) of different ages of rats with or without whey supplementation. A, A1, Cerebrum of adult and whey supplemented group. A3, Old cerebrum showing a decreased immune reaction. B, B1, Adult, and whey supplemented cerebellum. C2, Old cerebellum showing a decreased immune reaction. C, C1, Adult and whey supplemented hippocampus. C2, Aged hippocampus showing a decreased immune reaction. C3, Aged hippocampus supplemented whey showing improvement. B Chart illustrating Image analysis of synaptophysin showing decreased expression in an aging group compared to adult and aging group supplemented whey syrup. Each result represent the mean ± SD (n = 5); asterisk means significant at p < 0.05. G1 8 months old rats, G2 8 months old rats supplemented whey, G3 30 months old rats, G4 30 months old rats supplemented whey
3.10 Flow cytometry of annexin-v
The average number of apoptotic cells significantly increased in the aging brain compared to the aged group (G4) that fed whey. The percentages of apoptotic neuronal cells reached 54.1% compared to 92.4% in the experimental aged group (G4). Adult animals with or without whey syrup supplementation showed 14.3% and 17.3%, respectively (Fig. 5A, B).
A Flow cytometry analysis of annexin‐v of brain cells of aging rats with or without whey supplementation. From the chart, UL & LL show negative annexin‐v and negative propidium iodide (PI), indicating viable cells. UR showed positive annexin‐v and positive propidium iodide (PI), indicating early apoptosis. LR showed negative annexin‐v and positive propidium iodide (PI), indicating necrotic cells. UR + LR illustrating apoptosis. B Chart illustrating the apoptosis in the brain of aged rats and percent of improvement post whey supplementation. G1 8 months old rats, G2 8 months old rats supplemented whey, G3 30 months old rats, G4 30 months old rats supplemented whey. Data represent as the mean ± SD (n = 5); UR + LR in G2 is significant at p < 0.05
4 Discussion
Rats with advanced age had a significant increase in body weight, an increase in absolute brain weight, and a decrease in relative brain weight. The increase in body weights and decreased relative brain weight reflected the decline of metabolic function of body organs [38, 39] and the loss in body fat oxidation [40]. Thus, WP supplementation improved body weight gain and brain weight. In addition, whey supplements promoted protein synthesis and improved brain function during aging [41]. These resulted from its high content of amino acids [27] and its antioxidant activity to eliminate free radicals [42].
In elderly rats, the activity of SOD was significantly depleted, resulting in increasing MDA. The generation of free radicals caused damage to neuronal cells in the cerebrum, cerebellar cortex, and hippocampus. Damaged cerebral neurons led to the development of cerebral ischemia assessed by increased cerebral vasculogenesis. The hippocampus’s pyramidal layer becomes atrophied, and nearly all of its neurons become pleomorphic. Increased brain caspase-3, UR + LR of annexin-v flow cytometry, and caspase-3 immunohistochemistry confirmed apoptotic cell death.
The current findings supported those of Li et al. [43], who reported that aged rats had higher levels of β-galactosidase, and MDA as well as decreased SOD activity. In addition, aging decreased catalase activity, glutathione/oxidized glutathione ratio, and MDA increase in the animal brain cortex [44, 45]. These facilitated the release of an active form of oxygen, a cell death promoter [46, 47].
Whey supplementation also improved the histologic structure by increasing brain SOD activity and decreasing MDA levels, reducing caspase-3 immune reaction, and synaptophysin proliferation in neurons. These seemed to be connected to an increase in cysteine, an amino acid that serves as a precursor to glutathione, the machine of antioxidative activity [48].
The increase of inflammatory markers TNF-α and 5-lipoxygenase during the aging process activated the angiogenesis of blood capillaries and the dense grouping of glia cells in the cerebrum. It also caused a comparative increase of Purkinje and granular cells and the widespread occurrence of pleomorphic neuronal cells in the necrotic zones of the hippocampus.
These results were consistent with studies carried out by Garg et al. [41], who highlighted increased inflammatory markers (TNF-α, IL-1β, and IL-6) and reduced acetylcholinesterase activity in aged rats. These resulted in impaired learning and memory function, particularly in the subgranular region of the dentate gyrus, considering the hippocampus more susceptible to aging-related damage [49].
TNF-α and 5-LO were downregulated in aged rats therapeutically treated with whey. These findings matched Hashemilar et al. [50], who supplemented WP in critically ill patients with ischemic stroke. In addition, Banerjee and Poddar [51] mentioned that neurodegeneration enhanced 5-LO coincides with increased synthesis of leukotriene and inflammatory eicosanoids.
The observed neuronal damage of the aged brain reflected the significant depletion of DA, 5-HT, and ATP. In addition, the depletion of ATP content expressed the loss of mitochondrial function and failure of neuronal cellular energy [52] and a reduction of energy requirements for brain function [53].
The data revealed that the impaired brain function of elderly rats resulted in a significant increase in brain β-amyloid, tau protein, and α-amylase levels. Tau is a highly regulated microtubule-associated protein in neurons. The abnormal aggregation of insoluble tau has been linked to neuronal cell loss and synapse degeneration in pathological conditions [54]. Tau phosphorylation is a secondary effect of β-amyloid accumulation related to aging [55]. Disrupted brain metabolism has been associated with extracellular β-amyloid plaques and intracellular tau phosphorylated protein [56]. The hippocampus has been linked to memory development [57]. In aged rats, decreasing synaptophysin expression in neuronal axons predicted neurodegeneration and the onset of cognitive failure [58].
In addition, when compared to the adult rat, the aged brain had a higher level of α-amylase. Periodic acid-Schiff showed positive polyglucosan bodies in the brain of AD patients, correlated with the increased α-amylase activity [59]. Furthermore, aged rats exhibited a significant increase in serum levels of xanthine oxidoreductase (XOR), creatine kinase (CK), and acetylcholinesterase while having a lower decrease in NGF levels and BNP. In vitro antidiabetic effect exhibited inhibition of α-amylase activity by 60%, and in vivo activities tested in albino rats showed that the final product helped regulate the blood glucose [60].
The elevated serum acetylcholinesterase seen in the elderly brain reflected the neurodegenerative condition described by García-Ayllón et al. [61] in Alzheimer’s patients. Additionally, an XOR-generated ROS enhanced catabolism of purine bases [62] by liberating electrons to oxygen and generating O2− and H2O2 within the cell [63], results in an imbalance in the antioxidant systems. Furthermore, increased serum creatine kinase reflected damage in the aged rats that reflected mitochondrial damage [64]. Finally, reducing the serum nerve growth factor [65] and natriuretic peptides mirrored the neurodegenerative condition [66].
According to the current findings, orally supplemented whey improves MDA, caspase-3, inflammatory markers, β-amyloid, tau protein, and α-amylase is, reflecting increased SOD activity and ATP contents of the brain in aged rats. These changes increased brain function and improved histology and immunohistochemistry in the cerebrum, hippocampus, and cerebellum.
The current findings are inconsistent with previous brain research. WP has been shown to improve rats with aging-related galactosaemic disease associated with SOD depletion and MDA. Adult (8 months) and old Wistar rats (30 months) consumed WP (300 mg/kg b.w.) for 28 days showed a reduction in inflammatory markers (TNF-α, interleukin (IL)-1β, IL-6) associated with oxidative stress in aged animals [43]. Whey treatment also reduced MDA and improved brain coordination in diabetic animals [67, 68], increased mitochondrial activity [19], and managed brain structure and function [69, 70]. Sixteen weeks of leucine-enriched WP supplementation and combined with resistance-based exercise enhanced cardiometabolic health markers in old adults [16]. The inclusion of WP increased levels of liver antioxidant enzymes and decreased MDA and alanine aminotransferase (ALT) activities. Thus, WP could be recommended to enhance the growth performance and liver antioxidant enzymes in broiler chickens challenged with ethyl alcohol [12]. Reversal of the WP/Cas ratio in milk enhanced the insulin response, an effect possibly mediated by amino acids and/or incretins [15].
5 Conclusion
The current work investigated whether WP supplementation may improve age-related changes in diseased brain indicators like tau protein, β-amyloid and α-amylase. In conclusion, WP contains bioactive compounds and amino acids, which decrease brain oxidative stress and restore normal cognitive function. The increased antioxidant defense and DA and 5-HT neurotransmitters, while decreased brain tau and β-amyloid, were associated with better histology in aged rats' cerebrum, cerebellum, and hippocampus.
Availability of data and materials
Not applicable. Our manuscript has no associated data
Change history
02 November 2022
A Correction to this paper has been published: https://doi.org/10.1007/s43994-022-00010-9
References
Chu W, Wang P, Ma Z, Peng L, Wang Z, Chen Z (2022) Ultrasonic treatment of Dendrobium officinale polysaccharide enhances antioxidant and anti-inflammatory activity in a mouse D-galactose-induced aging model. Food Sci Nutr 00:1–11. https://doi.org/10.1002/fsn3.2867
Kalaria RN, Hase Y (2019) Neurovascular ageing and age-related diseases. Subcell Biochem 91:477–499
Lin B, Xu D, Wu S, Qi S, Xu Y, Liu X, Zhang X, Chen C (2021) Antioxidant effects of Sophora davidi (Franch.) Skeels on d–galactose–induced aging model in mice via activating the SIRT1/p53 pathway. Front Pharmacol 12:754554. https://doi.org/10.3389/fphar.2021.754554
Wan S, Liu Y, Shi J, Fan D, Li B (2021) Anti-photoaging and anti-inflammatory effects of ginsenoside Rk3 during exposure to UV irradiation. Front Pharmacol 12:716248. https://doi.org/10.3389/fphar.2021.716248
Fulop GA, Tarantini S, Yabluchanskiy A, Molnar A, Prodan CI, Kiss T et al (2019) Role of age-related alterations of the cerebral venous circulation in the pathogenesis of vascular cognitive impairment. Am J Physiol Heart Circ Physiol 6(5):H1124–H1140
Metzler-Baddeley C, Mole JP, Sims R, Fasano F, Evans J, Jones DK et al (2019) Fornix white matter glia damage causes gray matter damage during age-dependent limbic decline. Sci Rep 9(1):1060
Thammisetty SS, Pedragosa J, Weng YC, Calon F, Planas A, Kriz J (2018) Age-related deregulation of TDP-43 after stroke enhances NF-κB-mediated inflammation and neuronal damage. J Neuroinflammation 15(1):312
Sessa F, Maglietta F, Bertozzi G, Salerno M, Di Mizio G, Messina G et al (2019) Human brain injury and miRNAs: an experimental study. Int J Mol Sci 20(7):1546
Butterfield DA, Halliwell B (2019) Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Nat Rev Neurosci 20(3):148–160
Schipper HM, Song W, Tavitian A, Cressatti M (2019) The sinister face of heme oxygenase-1 in brain aging and disease. Prog Neurobiol 172:40–70
Saccà SC, Cutolo Rossi T (2019) Visual defects and ageing. Subcell Biochem 91:393–434
Afkhami M, Kermanshahi H, Majidzadeh Heravi R (2020) Evaluation of whey protein sources on performance, liver antioxidants and immune responses of broiler chickens challenged with ethanol. J Anim Physiol Anim Nutr 104(3):898–908. https://doi.org/10.1111/jpn.13327
Mignone LE, Wu T, Horowitz M, Rayner CK (2015) Whey protein: the “whey” forward for treatment of type 2 diabetes? World J Diabetes 6(14):1274–1284
Rodrigues G, Moraes T, Elisei L, Malta I, dos Santos R, Novaes R, Lollo P, Galdino G (2021) Resistance exercise and whey protein supplementation reduce mechanical allodynia and spinal microglia activation after acute muscle trauma in rats. Front Pharmacol 12:726423. https://doi.org/10.3389/fphar.2021.726423
Toffolon A, Rocco-Ponce M, Vettore M, Iori E, Lante A, Tessari P (2021) Effect of reversal of whey-protein to casein ratio of cow milk, on insulin, incretin and amino acid responses in humans. Mol Nutr Food Res. https://doi.org/10.1002/mnfr.202100069
Kirk B, Mooney K, Vogrin S, Jackson M, Duque G, Khaiyat O, Amirabdollahian F (2021) Leucine-enriched whey protein supplementation, resistance-based exercise, and cardiometabolic health in older adults: a randomized controlled trial. J Cachexia Sarcopenia Muscle. https://doi.org/10.1002/jcsm.12805
Waworuntu RV, Hanania T, Boikess SR, Rex CS, Berg BM (2016) Early life diet containing prebiotics and bioactive whey protein fractions increased dendritic spine density of rat hippocampal neurons. Int J Dev Neurosci 55:28–33
Krissansen GW (2007) Emerging health properties of whey proteins and their clinical implications. J Am Coll Nutr 26:713S-723S
Warinner C, Hendy J, Speller C, Cappellini E, Fischer R, Trachsel C et al (2014) Direct evidence of milk consumption from ancient human dental calculus. Sci Rep 4:7104
Gu F, Chauhan V, Chauhan A (2015) Glutathione redox imbalance in brain disorders. Curr Opin Clin Nutr Metab Care 18(1):89–95
Kerasioti E, Stagos D, Tsatsakis AM, Spandidos DA, Taitzoglou I, Kouretas D (2018) Effects of sheep/goat whey protein dietary supplementation on the redox status of rats. Mol Med Rep 17(4):5774–5781
Andreoli MF, Stoker C, Lazzarino GP, Canesini G, Luque EH, Ramos JG et al (2016) Dietary whey reduces energy intake and alters hypothalamic gene expression in obese phyto-oestrogen-deprived male rats. Brit J Nutr 116(6):1125–1133
Shertzer HG, Krishan M, Genter MB (2013) Dietary whey protein stimulates mitochondrial and decreases oxidative stress in mouse female brain. Neurosci Lett 548:159–164
Ajarem J, Allam AA, Ebaid H, Maodaa SN, Al-Sobeai SM, Rady AM et al (2013) Neurochemical, structural and neurobehavioral evidence of neuronal protection by whey proteins in diabetic albino mice. Behav Brain Function 11:7
Ho ST, Hsieh YT, Wang SY, Chen MJ (2019) Improving effect of a probiotic mixture on memory and learning abilities in d-galactose-treated aging mice. J Dairy Sci 102(3):1901–1909
Ano Y, Kutsukake T, Sasaki T, Uchida S, Yamada K, Kondo K (2019) Identification of a novel peptide from β-casein that enhances spatial and object recognition memory in mice. J Agri Food Chem 67(29):8160–8167
Ignowski E, Winter AN, Duval N, Fleming H, Wallace T, Manning E, Koza L et al (2018) The cysteine-rich whey protein supplement, Immunocal®, preserves brain glutathione and improves cognitive, motor, and histopathological indices of traumatic brain injury in a mouse model of controlled cortical impact. Free Rad Biol Med 24:328–341
Tosukhowong P, Boonla C, Dissayabutra T, Kaewwilai L, Muensri S, Chotipanich C et al (2016) Biochemical and clinical effects of whey protein supplementation in Parkinson’s disease: A pilot study. J Neurol Sci 2(367):162–170
Rondanelli M, Klersy C, Terracol G, Talluri J, Maugeri R, Guido D et al (2016) Whey protein, amino acids, and vitamin D supplementation with physical activity increases fat-free mass and strength, functionality, and quality of life and decreases inflammation in sarcopenic elderly. Am J Clin Nutr 103(3):830–840
Parris S, Baginskla MA (1991) Rapid method for the determination of whey protein denaturation. J Dairy Sci 74:58–64
Essig AM, Kleyn DH (1983) Determination of lactose in milk: comparison of methods. J Assoc Off Anal Chem 66(6):1514–1516
Lim YY, Quah EPL (2007) Antioxidant properties of different cultivars of Portulaca Oleracea. Food Chem 103(3):734–740
Karami S, Roayaei M, Hamzavi H, Bahmani M, Hassanzad-Azar H, Leila M et al (2017) Isolation and identification of probiotic Lactobacillus from local dairy and evaluating their antagonistic effect on pathogens. Int J Pharm Invest 7(3):137–141
Al-Gebaly AS (2019) Ameliorating role of whey syrup against ageing-related damage of myocardial muscle of Wistar Albino rats. Saudi J Biol Sci 26(5):950–956. https://doi.org/10.1016/j.sjbs.2018.03.014
Al-Gebaly AS (2019) Ameliorating role of whey syrup against ageing-related damage of myocardial muscle of Wistar Albino rats. Saudi J Biol Sci 26:950–956. https://doi.org/10.1016/j.sjbs.2018.03.014
Fairbanks V, Klee G (1996) Biochemistry aspects of haematology. Tietz fundamental of clinical chemistry 4th edn. WB Saunders Co., Philadelphia, 704, 30
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95(2):351–358
Heilbronn LK, Smith SR, Martin CK, Anton SD, Ravussin E (2005) Resting metabolic rate and respiratory quotient in human longevity. J Clin Endocrinol Metab 90:409–413
Solomon TP, Sistrun SN, Krishnan RK, Del Aguila LF, Marchetti CM, O’Carroll SM et al (2008) Exercise and diet enhance fat oxidation and reduce insulin resistance in older obese adults. J Appl Physiol (1985) 104(5):1313–1319
Jura M, Kozak LP (2016) Obesity and related consequences to ageing. Age (Dordr) 38(1):23
Garg G, Singh S, Singh AK, Rizvi SI (2018) Whey protein concentrate supplementation protects rat brain against aging-induced oxidative stress and neurodegeneration. Appl Physiol Nutr Metabol 43(5):437–444
Gholamian-Dehkordi N, Luther T, Asadi-Samani M, Mahmoudian MR (2017) An overview on natural antioxidants for oxidative stress reduction in cancers; a systematic review. Immunopathol Persa 3(2):e12
Li X, Chen Y, Shao S, Tang Q, Chen W, Chen Y et al (2016) Oxidative stress induces the decline of brain EPO expression in aging rats. Exp Gerontol 83:89–93
Halliwell B, Gutteridge JMC (1989) Free radicals in biology and medicine, second reprint. Clarendon Press, Oxford, p 543
Sousa MSB, Holanda IMS, Monteiro HMC, Amâncio-Dos-Santos  (2018) Antioxidant extract counteracts the effects of aging on cortical spreading depression and oxidative stress in the brain cortex. Acta Cir Bras 6:472
Amin AH, Bughdadi FA, Abo-Zaid MA, Ismail AH, El-Agamy SA, Alqahtani A et al (2019) Immunomodulatory effect of papaya (Carica papaya) pulp and seed extracts as a potential natural treatment for bacterial stress. J Food Biochem 43(12):e13050
Stefanatos R, Sanz A (2018) The role of mitochondrial ROS in the aging brain. FEBS Lett 592(5):743–758
Parodi PW (2007) A role for milk proteins and their peptides in cancer prevention. Curr Pharm Design 13(8):813–828
Lazarov O, Hollands C (2018) Hippocampal neurogenesis: learning to remember. Prog Neurobiol 138–140:1–18
Hashemilar M, Khalili M, Rezaeimanesh N, Sadeghi Hokmabadi E, Rasulzade S, Shamshirgaran SM et al (2020) Effect of whey protein supplementation on inflammatory and antioxidant markers, and clinical prognosis in acute ischemic stroke (TNS Trial): A randomized, double blind, controlled, clinical trial. Adv Pharm Bull 10(1):135–140
Banerjee S, Poddar MK (2016) Aging-induced changes in brain regional serotonin receptor binding: effect of arnosine. Neuroscience 319:79–91
Ferrari F, Viscardi P, Gorini A, Villa RF (2019) Synaptic ATPases system of rat frontal cerebral cortex during aging. Neurosci Lett 694:74–79
Kim WS, Bhatia S, Elliott DA, Agholme L, Kågedal K, McCann H et al (2010) Increased ATP-binding cassette transporter A1 expression in Alzheimer’s disease hippocampal neurons. J Alzheimers Dis 21(1):193–205
Hefti MM, Kim S, Bell AJ, Betters RK, Fiock KL, Iida MA, Smalley ME et al (2019) Tau Phosphorylation and aggregation in the developing human brain. J Neuropathol Exp Neurol 78(10):930–938
Shen LL, Li WW, Xu YL, Gao SH, Xu MY, Bu XL, Liu YH et al (2019) Neurotrophin receptor p75 mediates amyloid β-induced tau pathology. Neurobiol Dis 132:104567
Forny-Germano L, Lyra e Silva NM, Batista AF, Brito-Moreira J, Gralle M, Boehnke SE et al (2014) Alzheimer’s disease-like pathology induced by amyloid-β oligomers in nonhuman primates. J Neurosci 34(41):13629–13643
Hara Y, Rapp PR, Morrison JH (2012) Neuronal and morphological bases of cognitive decline in aged rhesus monkeys. Age 34(5):1051–1073
Byman E, Schultz N (2018) Netherlands Brain Bank, Fex M, Wennström M, Brain alpha-amylase: a noval energy regulator important in Alzheimer disease? Brain Pathol 28(6):920–932
Brown AM, Ransom BR (2007) Astrocyte glycogen and brain energy metabolism. Glia 55(12):1263–1271
Jan Y, Malik M, Sharma V, Yaseen M, Bora J, Haq A (2021) Utilization of whey for formulation of whey jamun juice ice pops with antidiabetic potential. Food Frontiers. https://doi.org/10.1002/fft2.90
García-Ayllón MS, Riba-Llena I, Serra-Basante C, Alom J, Boopathy R, Sáez-Valero J (2010) Altered levels of acetylcholinesterase in Alzheimer plasma. PLoS ONE 5(1):e8701
Battelli MG, Polito L, Bortolotti M, Bolognesi A (2016) Xanthine oxidoreductase-derived reactive species: physiological and pathological effects. Oxid Med Cell Long 2016:3527579
Harrison R (2002) Structure and function of xanthine oxido-reductase: where are we now? Free Rad Biol Med 33(6):774–797
Bürklen TS, Schlattner U, Homayouni R, Gough K, Rak M, Szeghalmi A, Wallimann T (2008) The creatine kinase/creatine connection to Alzheimer’s disease: CK-inactivation, APP-CK complexes and focal creatine deposits. J Biomed Biotechnol 3:35936
Budni J, Bellettini-Santos T, Mina F, Garcez ML, Zugno AI (2015) The involvement of BDNF, NGF and GDNF in aging and Alzheimer’s disease. Aging Dis 6(5):331–341
Mahinrad S, Bulk M, van der Velpen I, Mahfouz A, van Roon-Mom W, Fedarko N et al (2018) Natriuretic peptides in post-mortem brain tissue and cerebrospinal fluid of non-demented humans and Alzheimer’s disease patients. Front Neurosci 12:864
Ochoa TJ, Sizonenko SV (2017) Lactoferrin and prematurity: a promising milk protein? Biochem Cell Biol 95(1):22–30
Qian Y, Zhang J, Zhou X, Yi R, Mu J, Long X et al (2018) Lactobacillus plantarum CQPC11 Isolated from sichuan pickled cabbages antagonizes d-galactose-induced oxidation and aging in mice. Molecules 23(11):3026
Ano Y, Ayabe T, Kutsukake T, Ohya R, Takaichi Y, Uchida S et al (2018) Novel lactopeptides in fermented dairy products improve memory function and cognitive decline. Neurobiol Aging 72:23–31
Wurtman RJ, Cansev M, Sakamoto T, Ulus IH (2009) Use of phosphatide precursors to promote synaptogenesis. Ann Rev Nutr 29:59–87
Acknowledgements
The authors thank the Deanship of Scientific Research at Umm Al-Qura University for supporting this work by Grant Code: 19-SCI-1-01- 0037.
Funding
Deanship of Scientific Research at Umm Al-Qura University (Grant Code: 19-SCI-1-01-0037).
Author information
Authors and Affiliations
Contributions
MEE and HIHE collected and analyzed the samples. HIHE, MEE, AAE, AHA, SHQ, and WMF carried out experimental analyses. MFR reviewed and edited the article. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
The Experimental Animal Ethical Committee (Faculty of Science, Mansoura University, Egypt) approved the experiment (Approved Statement No. RZ19004).
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised to correct the affiliations of authors Ali H. Amin and Sameer H. Qari.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El-Beeh, M.E., El-Badawi, A.A., Amin, A.H. et al. Anti-aging trait of whey protein against brain damage of senile rats. J.Umm Al-Qura Univ. Appll. Sci. 8, 8–20 (2022). https://doi.org/10.1007/s43994-022-00001-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43994-022-00001-w