Abstract
Colorimetric protein quantitation assays such as the Bradford method are susceptible to foaming when using manual or automated equipment, especially during mixing operations. The resulting bubbles cause unintended light scattering and, because they often persist for longer than the duration of the assay, can interfere with spectrometric sample analysis, for example in a 96-well plate reader at 585 nm. Here, we tested the ability of antifoam agents commonly used in biotechnology to reduce bubble formation when mixing Bradford reagent with protein samples from bioprocess development and production. We also assessed the impact of these agents on optical density at the assay-relevant wavelength to verify the fidelity of the readouts. We used a design of experiments approach to identify synergies between different antifoam agents, aiming to reduce the concentration needed to avoid foam formation and thus minimize any distortion of the assay results. Finally, we confirmed that antifoam-containing Bradford reagent retained its performance even after prolonged storage. We found that 0.15 g L−1 of Struktol J673A was sufficient to prevent foaming even when air was deliberately introduced into the samples. Furthermore, the modified Bradford reagent was stable for 4 weeks and did not distort the readout at 585 nm but only shifted it by ~ 0.1 dimensionless absorbance units. Therefore, our modified Bradford reagent will benefit biologists, biotechnologists and bioprocess engineers, especially those using automated workstations, because it improves the robustness of the assay.
Highlights
-
Adding 0.15 g L−1 of Struktol J673A to Bradford reagent completely prevents foam formation.
-
The addition of antifoam has a minimal effect on spectrometric assay readouts.
-
An antifoam-containing Bradford reagent can be stored for at least 4 weeks.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The original description of the Bradford method is among the most cited publications in biology and biotechnology [1]. This reliable method for protein quantitation is widely used in molecular biology laboratories and is also a workhorse in bioprocess development, for example to determine the purity of protein products in samples as a fraction of the total soluble protein content [2]. The assay is based on the dye Coomassie Brilliant Blue, which primarily binds to amino acids with amino and carboxyl side chains, whereupon it changes color from brown to blue with an absorption maximum at 585 nm [1]. This allows the straightforward quantification of proteins by spectrophotometry, for example in a 96-well plate format. The measurement of protein standards over a known concentration range allows the construction of a standard curve of absorbance readings from which the protein concentration in any sample can be deduced.
Conveniently, standardized staining solutions are commercially available along with detailed protocols, even for microliter-scale variants of the assay [3]. However, physical implementation, for example using single/multi-channel pipettes or liquid handling stations, can be challenging because substantial amounts of foam can form when mixing the reagent with a protein-containing sample. The foam can result from the detergent-like properties of some proteins [4], or because the dye has a similar effect in the solution. Minimizing the introduction of air can address this issue, but it can be challenging even for skilled experimenters to achieve consistently foam-free samples, and extra effort is needed to manually eliminate any bubbles. Bubbles may even form when using automated liquid handling stations if liquid levels differ slightly between wells in 96-well plates. In this case, identifying and eliminating bubbles is especially cumbersome because the assay would typically be operated without manual intervention. Destroying the bubbles is necessary because they would otherwise cause unintended light scattering during spectrophotometry, thus distorting the reported protein concentration.
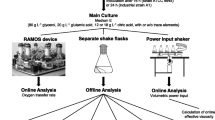
Here, we tested seven different antifoam agents developed for fermentation processes in terms of their ability to prevent foam formation in a 200-µL Bradford assay in a 96-well format. We fine-tuned the antifoam concentrations to ensure a minimal impact on the absolute absorbance measured in protein samples, thus maintaining the assay fidelity. Furthermore, we tested combinations of different antifoam agents to achieve this goal in a design of experiments (DoE) approach and ultimately confirmed that Bradford reagent containing antifoam agents can perform consistently after storage (Fig. 1).
2 Materials and methods
2.1 Screening of foam-suppressing and/or foam-destroying activity in Bradford reagent
The foam-suppressing and/or foam-destroying substances listed in Table 1 were prepared as 100.0 g L−1 stock solutions and then individually added to 5 mL Bradford reagent (Pierce Coomassie Protein Assay Kit; Thermo Fisher Scientific) to achieve a final concentration of 1.00 g L−1. Based on the foam-reducing ability of each reagent, this concentration was either reduced stepwise to 0.01 g L−1 (Struktol J673A, Medical Antifoam C and Xiameter AFE 100), kept constant (Bisomer G30) and or even increased to 10.00 g L−1 (Pluronic L61 and Dow Corning 365) in a second set of samples. All 5-mL samples were protein-free and were mixed in 15-mL reaction tubes by rigid manual shaking for 20 s. The time until complete foam disintegration was measured and the extinction at 585 nm was quantified using a BioPhotometer (Eppendorf) immediately after the addition of antifoam and again after incubation for 60 min at 22 °C in a light-protected environment. We selected a shift in absorbance of less than 0.10 as a selection criterion because it was ~ 10 time the average standard deviation of the Bradford assay as performed in our hands. A 5-mL sample of untreated Bradford reagent was used as a control.
Steps in the testing of antifoam agents for foam reduction in a Bradford assay setting. After foam-reducing activity was screened (first row), concentrations of antifoam agents were optimized on an individual (second row) and combined basis (third row), the latter using a design of experiments (DoE) approach. Finally, the performance of the optimized Bradford reagent containing antifoam was tested after storage (fourth row)
2.2 Assessment of antifoam activity in a Bradford assay setting
Different concentrations of antifoam agents that successfully disintegrated bubbles in Bradford reagent without protein were added to mixtures of Bradford reagent and 0, 125, 250, 500, 100, 1500 or 2000 mg L−1 bovine serum albumin (BSA; Thermo Fisher Scientific) in a 96-well plate format. Bubbles were intentionally induced by pipetting 200 µL of air into each sample. Bradford reagent containing chicken albumin (Sigma Aldrich), lysozyme (Sigma Aldrich), ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO; purified from tobacco leaves) and the human immunoglobulin biological reference preparations (BRP) (European Directorate for the Quality of Medicines & HealthCare) at the same concentrations as BSA were tested in the same manner. The number of bubbles (Figure S2), was quantified after incubation for 10 min at 22 °C (the standard condition of the assay) by counting bubbles ≥ 1 mm diameter individually and by including bubbles < 1 mm, e.g., in agglomerates, as a single count. The sample absorption at 585 nm was measured in an Enspire plate reader (PerkinElmer). Readouts for the individual antifoam agents in the presence of Bradford reagent and different concentrations of BSA were used to fit cubic regression models (Eq. 1). The resulting coefficients (kn) were compared to corresponding values in a sample without antifoam agent.
where y is the absorbance at 585 nm, kn are model coefficients and x is the protein concentration.
2.3 Determination of synergistic effects among antifoam agents
Antifoam agent mixtures containing different concentrations of Pluronic L61 (poloxamer), Struktol J673A (alkoxylated fatty acid ester) and XIAMETER AFE-0100 (silicon) (Tables S1 and S2) were tested in a 32-run i-optimal (minimizing the integral of the prediction variance across the factor space) mixture DoE for the absorbance and number of bubbles. The latter was an integer response, so each run was replicated 12 times to obtain a pseudo-continuous response [5, 6]. For sample preparation, stock solutions of Bradford reagent containing 0.50 g L−1 Pluronic L61, 0.15 g L−1 Struktol J673A or 0.50 g L−1 Xiameter AFE 0100 were mixed to yield the antifoam concentrations as defined by the DoE. Plain Bradford reagent was added as required. Then, 195 µL of the mixture was added to 5 µL of 2.00 g L−1 BSA in a 96-well plate format and mixed. Again, bubbles were artificially created by pipetting 200 µL of air to each well. Finally, the number of bubbles was counted after 10 min incubation and the absorption at 585 nm was measured in an Enspire plate reader.
2.4 Analysis of antifoam Bradford reagent storage stability
Bradford reagent containing 0.15 g L−1 Struktol J673A was stored for 4 weeks at 4 °C (storage temperature recommended by the manufacturer) or 37 °C. The solution was then used in a triplicate 96-well plate format to quantify BSA, human immunoglobulin BRP, RuBisCO and a mixture of tobacco host cell proteins using the same protein concentrations as before. Untreated Bradford reagent was stored under the same conditions and used as a control.
2.5 Statistical data analysis
Design Expert v10 was used to set up and evaluate all statistical designs as previously described [7]. Other data were assessed for normality first using a battery of tests in Origin 2020b (Shapiro–Wilk, Lilliefors, Kolmogorov–Smirnov, Anderson–Darling, D’Agostino-K squared, and Chen-Shapiro). If all tests indicated normality for two comparative samples, we used Welch’s t-test with Bonferroni correction for analysis. If the data distribution was non-normal, we used a Wilcoxon-Mann–Whitney test (U-test) [8, 9]. The two-sided alpha level was 0.05 and the p-value threshold to indicate significance was 0.05.
3 Results and discussion
3.1 Foam reduction in Bradford reagent
We screened seven antifoam agents at different concentrations (Table 1) for their ability to reduce the number of bubbles after vigorous mixing with 5 mL of Bradford reagent in 15-mL reaction tubes. Struktol J647 was insoluble in the reagent and was therefore excluded from further testing, whereas Dow Corning 365 and Pluronic L61 resulted in mixtures containing more than 20 bubbles, which was similar to the untreated control. Pluronic L61 was only able to reduce the number of bubbles when present at a concentration of 10 g L−1. We deemed this impractical because it substantially increased the absorbance of the solution at 585 nm from ~ 0.07 (blank control) to > 0.22, which potentially would interfere with the assay readout. A similar shift in absorbance was observed for Bisomer G30 (~ 0.16), but this occurred even at a concentration of only 0.50 g L−1. In contrast, less than one bubble on average was observed when Dow Corning Medical Antifoam C, Struktol J673A or XIAMETER AFE-0100 was added to the Bradford reagent. In addition, these antifoam agents increased the absorbance at 585 nm by less than 0.10. Therefore, we continued with these three antifoam agents and identified the minimal concentration required to suppress foam formation in Bradford reagent. These concentrations were 0.50 g L−1, 0.15 g L−1 and 0.50 g L−1 for Dow Corning Medical Antifoam C, Struktol J673A and XIAMETER AFE-0100, respectively (Table 1).
3.2 Assessment of antifoam agents in a Bradford assay setting
Next, we used Dow Croning Medical Antifoam C, Struktol J673A and XIAMETER AFE-0100 in an authentic Bradford assay setting to assess their antifoam properties in the presence of a protein sample and to evaluate any potential distortion of the assay readout at 585 nm in more detail. The antifoam agents fully prevented foam formation, however, in case of XIAMETER AFE-0100, the concentration stated in Table 1 had to be increased to 0.50 g L−1.
The shift in absorbance increased with increasing antifoam and protein concentration (Fig. 2, Table 2), but was less than 0.10 dimensionless absorbance units at the antifoam agent concentrations required to prevent bubbles. Importantly, the absorbance shift relative to the protein concentration was close to constant with slopes between -0.09 and 0.02, indicating minimal interference with the Bradford assay readout. The only two exceptions were Dow Corning Medical Antifoam C combined with RuBisCO at protein concentrations of 0.25–1.00 g L−1, where the maximal shift was up to 0.11 and protein concentrations below 0.25 g L−1 where the absorbance shift relative to the protein concentrations was higher compared to the rest of the protein concentration range. When analyzing the absorbance shift induced by antifoam agents in more detail by comparing sample spectra in the 280–900 nm range (Fig. 2D), we found that, for example in the case of Struktol J 673 A, the shift was between − 0.01 and 0.10 at an antifoam concentration of 0.15 g L−1 and in the presence of 2 g L−1 BSA. Importantly, in the assay-relevant wavelength region of 550–650 nm, the average difference between samples with and without antifoam was less than 0.01.
Effect of antifoam agents on Bradford assay readouts (absorbance at 585 nm). Different concentrations of the antifoam agents Dow Corning Medical Antifoam C (A), Struktol J673A (B) and XIAMETER AFE-0100 (C) were tested in Bradford reagent mixed with defined concentrations of BSA in the concentration range 0.000–2.000 g L−1. Additional measurements using lysozyme (blue) or RuBisCO (orange) in the same concentration range are shown for XIAMETER AFE-0100. Sample spectra (D) were taken for Struktol J673A in Bradford reagent in the presence (orange) or absence (green) of 2.000 g L−1 BSA. No artificial bubbles were introduced in the control (0 g L−1 antifoam agent) to ensure readout comparability. The in-panel legend shows the antifoam concentration [g L−1]. All tests were carried out in a 96-well plate format
We used a BSA dilution series, the common standard to calibrate Bradford assay measurements [3], to establish a cubic regression model between protein concentration and absorbance. We compared the model coefficients derived from measurements containing Dow Corning Medical Antifoam C, Struktol J673A or XIAMETER AFE-0100 with those from controls without antifoam to identify any systematic deviations (Table 3). We found that all models were capable of describing the data with high fidelity (R2 and adjusted R2 > 0.99). The model coefficients k0 and k1 for Struktol J673A did not differ significantly from the antifoam-free control. Importantly, the average absorbance difference between Struktol J673A and the control was smallest among all the tested substances. The average difference between Struktol J673A and the control was negative because the control exhibited less absorbance at 585 nm if more than 0.5 g L−1 BSA was present. Importantly, the difference in the area under the curve was also smallest between Struktol J673A and the control, indicating that this antifoam agent had the least impact on absorbance.
3.3 Effect of antifoam agent mixtures on foaming in a Bradford assay setting
Having established that individual agents can be used to suppress the foaming of Bradford reagent samples without substantially distorting the assay readout, we investigated the potential for synergies between the antifoam agents that might allow us to reduce the concentration even further and thus minimize any impact on absorbance. For example, we speculated that antifoam agents with different chemistries may achieve bubble disintegration using slightly different mechanisms that complement each other, and mixing them may therefore augment the foam-reducing effect. The initial set of antifoam agents we tested (Table 1) covered diverse chemistries, specifically block copolymers, silicone derivatives and fatty acid esters, but only the latter two chemistries were represented among the antifoam candidates that achieved both foam prevention and a negligible impact on absorbance. We therefore included Pluronic L61 as a block-copolymer-based antifoam agent and selected XIAMETER AFE-0100 over Dow Corning Medical Antifoam C because the latter is about 10-times more expensive. We tested various mixtures of Pluronic L61, Struktol J673A and XIAMETER AFE-0100 in a DoE approach to assess the number of bubbles and absorbance (Fig. 3). The data were well described by reduced cubic models with adjusted R2 > 0.80 (Table S2). Interestingly, all combinations of Struktol J673A and XIAMETER AFE-0100 prevented the formation of bubbles down to concentrations as low as 0.025 g L−1 (Fig. 3), a quarter or even 5% of the minimum concentration necessary for pure antifoam agents. As expected based on the initial experiments, numerous bubbles were observed if pure Pluronic L61 was used or if its fraction in the mixture exceeded ~ 50%. Interestingly, the absorbance at 585 nm was highest in binary mixtures of Struktol J673A and XIAMETER AFE-0100. However, the absorbance was at most 0.04 higher than that of antifoam agent-free Bradford reagent, thus below the 0.10 threshold discussed above and hence not relevant according to our criteria.
Ternary plots of the effect of antifoam agent mixtures on the number of bubbles (top row) and absorbance (bottom row) when Bradford reagent was mixed with 2 g L−1 BSA. The total concentrations of antifoam agents were 0.002 g L−1 (A and D), 0.05 g L−1 (B and E) and 0.35 g L−1 (C and F). Vertices correspond to conditions using only the indicated single antifoam agent. P—Pluronic L61, S—Struktol J673A, X—XIAMETER AFE-0100. All measurements were conducted in 96-well plates and air bubbles were deliberately introduced by pipetting. Absorbance was measured 10 min after the Bradford reagent was mixed with the protein solution
We concluded that mixtures of antifoam agents can effectively remove bubbles at total concentrations even below those necessary for pure agents. This helps to further reduce the effect of the agents on Bradford assay readouts. However, we found that the 100 g L−1 XIAMETER AFE-0100 stock solutions started to mold after about 4 days at 22 °C. The reason for the microbial growth is currently unclear and was beyond the scope of this study. Therefore, we decided to continue our assessment with Struktol J673A alone because it effectively reduced the number of bubbles even at low concentrations, did not substantially shift absorbance values, and did not support visual microbial growth within 7 days.
3.4 Stability of Bradford reagent containing antifoam agents
We extended our preliminary stability assessment by incubating individual aliquots of antifoam-containing Bradford reagent and antifoam-free controls for 4 weeks at 4 or 37 °C. The Struktol J673A concentration was 0.15 g L−1 and no bubbles formed even after the augmented Bradford reagent was stored at 4 or 37 °C. After 4 weeks of storage, absorbance values determined using Bradford reagent containing Struktol J673A exhibited standard deviations that were on average 8.4 times lower (incubation at 4 °C) or 2.6-fold lower (incubation at 37 °C) than antifoam-free controls (Fig. 4). These differences in the non-normally distributed data were not significant according to a Wilcoxon-Mann–Whitney test (U-test). However, the corresponding cubic models for the antifoam-containing reagent aligned more closely with the Bradford reagent without antifoam or bubbles than the antifoam-free Bradford reagent in which bubbles were induced at 4 °C. The exception was RuBisCO, for which the Bradford reagent containing Struktol J673A showed much higher absorbance, especially in the 0.500–1.500 g L−1 concentration range (Fig. 4C). We speculate that this protein-specific effect may be linked to the oligomeric structure of RuBisCO and a resulting unusual interaction with the dye. Authentic standards may be necessary to prevent systematic deviations when quantifying such proteins, but this challenge is not exclusive to Bradford reagent containing antifoam having been described for other proteins under regular conditions (e.g., elastin-like polypeptides that bind poorly to Coomassie Brilliant Blue G-250 [10]). Interestingly, the use of Bradford reagent stored at 37 °C resulted in a color shift from brown to red-brown in the presence or absence of Struktol J673A (Figure S1), but the shift was more pronounced in the presence of the antifoam agent. Specifically, the absorbance recorded when incubating proteins with Bradford reagent containing Struktol J673A was 0.08 ± 0.02 (n = 32), which is higher than for the reagent without antifoam. Because storage at 37 °C is not recommended by the Bradford reagent manufacturer, this observation may not be critical. However, storage at high temperatures is often regarded as a tool to assess accelerated aging [11] and the results may therefore indicate that storage at 4 °C for more than 4 weeks may alter the properties of the Bradford solution containing antifoam. However, the accelerated aging approach is a matter of debate [12], and further data under authentic storage conditions should be collected in the future.
Impact of storage on the absorbance of Bradford reagent containing antifoam agents. Bradford reagent containing 0.15 g L−1 Struktol J673A and controls were stored at 4 or 37 °C for 28 days and then used with BSA (A), a monoclonal antibody (B), RuBisCO (C) and a plant homogenate containing a diverse set of proteins (D). The concentrations of all protein stocks were in the 0.000–2.000 g L−1 range. Bubbles were induced in Bradford reagent with and without the antifoam agent, and the latter was also used in the absence of bubbles as a control. Data represent averages of 12 measurements in 96-well plates and the error bars are the corresponding standard deviations
For now, we ascertained that the storage of Bradford reagent containing Struktol J673A was possible for at least 4 weeks without negatively affecting the assay performance and it increased resistance to bubble formation, especially if stored at the manufacturer’s recommended temperature of 4 °C.
4 Conclusion
The Bradford assay is widely used in biology and biotechnology but its performance can suffer due to the formation of foam during pipetting. Here we tested seven antifoam agents and found that adding 0.15 g L−1 Struktol J673A alone was sufficient to avoid foaming even after intensive mixing while creating air bubbles. That concentration can be reduced to ~ 0.03 g L−1 if Struktol J673A is combined with XIAMETER AFE-0100. Importantly, it was possible to add the antifoam directly to the Bradford reagent and store the mixture at 4 °C for 4 weeks, which simplifies applications in the laboratory. Furthermore, the addition of Struktol J673A had no significant effect on absorbance readings and instead improved the performance of the Bradford reagent by preventing bubble formation, compared to a control without antifoam, reflecting an almost tenfold reduction in readout standard deviation. Our modified Bradford reagent will be helpful to biologists, biotechnologists and bioprocess engineers seeking to determine the total protein contents of samples, especially in high-throughput assays.
Data availability
Data can be made available upon request to the corresponding author.
Code availability
Not applicable.
References
Simonian MH, Smith JA. Spectrophotometric and colorimetric determination of protein concentration. Curr Protoc Mol Biol. 2006;76:10.1.1-10.1A.9. https://doi.org/10.1002/0471142727.mb1001as76.
Gengenbach BB, Opdensteinen P, Buyel JF. Robot cookies—plant cell packs as an automated high-throughput screening platform based on transient expression. Front Bioeng Biotechnol. 2020;8:393. https://doi.org/10.3389/fbioe.2020.00393.
Buyel JF, Fischer R. Scale-down models to optimize a filter train for the downstream purification of recombinant pharmaceutical proteins produced in tobacco leaves. Biotechnol J. 2014;9:415–25. https://doi.org/10.1002/biot.201300369.
Zayas JF. Foaming properties of proteins. In: Zayas JF, editor. Functionality of proteins in food. Berlin: Springer; 1997. p. 260–309.
Ali Z, Bhaskar SB. Basic statistical tools in research and data analysis. Indian J Anaesth. 2016;60:662–9. https://doi.org/10.4103/0019-5049.190623.
Green JW, Springer TA, Holbech H, editors. Statistical analysis of ecotoxicity studies. Hoboken: Wiley; 2018.
Buyel JF, Fischer R. Characterization of complex systems using the design of experiments approach: transient protein expression in tobacco as a case study. J Vis Exp. 2014;1:e51216. https://doi.org/10.3791/51216.
Pek J, Wong O, Wong ACM. How to address non-normality: a taxonomy of approaches, reviewed, and illustrated. Front Psychol. 2018;9:2104. https://doi.org/10.3389/fpsyg.2018.02104.
Sainani KL. Dealing with non-normal data. PM R. 2012;4:1001–5. https://doi.org/10.1016/j.pmrj.2012.10.013.
Hassouneh W, Christensen T, Chilkoti A. Elastin-like polypeptides as a purification tag for recombinant proteins. Curr Protoc Protein Sci. 2010. https://doi.org/10.1002/0471140864.ps0611s61.
Cho C, Nam SL, Mata AP, Harynuk JJ, Elias AL, Chung H-J, Dolez PI. Investigation of the accelerated thermal aging behavior of polyetherimide and lifetime prediction at elevated temperature. J Appl Polym Sci. 2022;139:51955. https://doi.org/10.1002/app.51955.
Frigione M, Rodríguez-Prieto A. Can accelerated aging procedures predict the long term behavior of polymers exposed to different environments? Polymers. 2021. https://doi.org/10.3390/polym13162688.
Acknowledgements
We wish to thank Dr. Richard M Twyman for editorial assistance. We also thank Robin Kastilan for discussions about the selection of antifoam agents.
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was funded in part by the Fraunhofer-Gesellschaft Internal Programs under grant no. Attract 125-600164 and the state of North-Rhine-Westphalia under the Leistungszentrum Grant No. 423 “Networked, adaptive production”.
Author information
Authors and Affiliations
Contributions
CB and JB planned the study. CB conducted the experiments. CB and JB analyzed the data. CB prepared the figures. CB and JB wrote the manuscript. JB revised the manuscript. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Consent for publication
All authors have seen a draft version of the manuscript and concur with its submission to the journal.
Competing interests
The authors declare they are inventors on a patent application disclosing the use of antifoam agents in Bradford reagent. They are however not the owners of the patent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bernau, C.R., Buyel, J.F. The use of antifoam agents to eliminate bubbles during biotechnological sample analysis. Discov Chem Eng 3, 5 (2023). https://doi.org/10.1007/s43938-023-00021-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43938-023-00021-w