Abstract
This study aims to explore technological advances and prospects in phenol treatment by providing a detailed bibliometric survey of wastewater treatment applications, highlighting innovative advances in research. Using the Web of Science database, we identified 79,104 articles from 2003 to 2023, later refined to 1848. The keywords were used for the initial search: “phenol”, “wastewater”, “degradation”, “treatment” and “removal”. The bibliographic review details the occurrence of journals, authors, newspapers, countries, institutions, keywords, highly cited articles, and prominent predominant research fields. In particular, the field of “Engineering” was responsible for 32% of the published articles, followed by “Ecology of Environmental Sciences” (25%) and “Chemistry” (12%). In addition, a keyword analysis revealed five major groups of clusters that indicate where the research is progressing. This aspect is crucial for understanding the evolution and perspectives of research interests over time. Therefore, future research in the field should prioritize wastewater treatment and feedstock diversification. This focus is essential to address significant challenges, such as production costs, stability, and durability of treatment processes.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Phenolic compounds are present in wastewater from various industries, including coking, pharmaceutical, petrochemical, and petroleum refining [1]. Industrial wastewater contains multiple organic products, such as phenol, PCBs, cyanide, and heavy metals (including Pb and Cd) and their compounds, originating from different industrial processes [2,3,4]. Contamination of water bodies with phenols can adversely affect aquatic fauna and flora and threaten the safety of water supplies for human consumption. Phenol and its derivatives, as aromatic pollutants with high toxicity, are widespread and persistent in aquatic environments because of the stability of their benzene structure [5,6,7].
Eliminating these compounds (e.g., phenol) plays a crucial role, as their presence in industrial and municipal wastewater challenges water quality. Conventional water treatment methods often face difficulties in effectively removing these pollutants, leading to a growing interest in investigating alternative and sustainable approaches [8].
Several methods for the removal of phenolic compounds from wastewater have been documented, including membrane separation and biodegradation [9], electrochemical oxidation of phenol [10, 11], direct osmosis [12], coal-based electrodes [13], ion exchange [14], anaerobic membrane [15, 16], aerobic biodegradation [17], photocatalytic degradation [18], and adsorption [19, 20]. However, these methods are expensive and often best suited for small to medium-scale applications [21]. Out of these methods, adsorption is emerging as a notable and promising solution for phenol removal because of its remarkable efficiency, favourable cost–benefit ratio, and simplified operational and design features compared to other methods [22]. Adsorption is a separation process in which one or more components are attracted to the surface of a solid adsorbent upon contact [23].
The present study aims to explore technological advances and prospects in phenol treatment through a detailed bibliometric survey of wastewater treatment applications, highlighting innovative advances in research. It will consider the primary search parameters, such as authors, journals, research institutions, countries, and keywords. This analysis allows us to answer specific questions about conventional treatment advances, highlighting effectiveness, limitations, and costs. Technological advances, innovations in efficiency, adsorbent materials, and biological processes will also be considered. In the future, the field of phenol treatment should focus on promising directions, identifying research gaps and growth opportunities, environmental, regulatory, and financial feasibility aspects, sectoral applications, and specific challenges in different industries.
2 Methods
2.1 Data analysis
This review was conducted based on previous studies [24,25,26,27,28,29,30,31,32]. Data were accessed and transferred to a text file for in-depth analysis. This procedure was carried out through “CAFe Access” on the CAPES Journal Portal, accessed on December 1, 2023. The Web of Science (WoS) Core Collection is considered the most reliable citation database in the world because of its verification and extensive data curation [33, 34]. The database was imported into VOSviewer, RStudio V3, and CiteSpace (v.6.2.R6) software to perform a visual, comprehensive bibliometric analysis of all documents in the database [35, 36]. The CiteSpace software performs a structural and temporal analysis of information from documents obtained from WoS, allowing the construction of different networks, such as collaboration networks, author co-citation networks, and bibliographic co-citation networks [37].
The search started with the term “phenol”. Subsequently, a second filter was applied based on the range of publication years, covering 2003 to 2023. The choice of the period from 2003 to 2023 was based on the delimitation of the research scope and the availability of relevant data for analysis. The year 2003 was chosen as the starting point, as only 19 publications were recorded, representing a considerably low number within the period investigated. Furthermore, the period up to 2023 allows for a comprehensive analysis of trends and developments in wastewater treatment over two decades. The leading search terms used in the WoS search field were “Phenol”, “wastewater”, “degradation”, “treatment”, and “removal”, which were included to refine the results further. The search covered a wide range of studies related to phenol wastewater treatment using a search string consisting of these keywords. This selection addressed different aspects of the problem, from the contaminated wastewater’s nature to phenol's degradation and removal processes. Together, these keywords provided a comprehensive approach to identifying relevant articles contributing to advancing phenol wastewater treatment technologies. This specific focus on wastewater treatment culminated in the identification of 1848 articles for analysis. This analysis allows us to identify the major trends, contributions, and gaps in this rapidly evolving research field. Figure 1 shows the search criteria used to select the database for the literature search. The present study uses the refined bibliometric dataset to address questions related to bibliometric analysis and provide a comprehensive view of wastewater treatment.
3 Results and discussion
3.1 Advanced bibliometric analysis
3.1.1 Bibliometric analysis of wastewater treatment with phenol over the last 20 years
The WoS search yielded a total of 79,104 articles with the word phenol, which were refined into 1848 articles with the above keywords, of which 1700 articles (92%) were classified as full articles, 110 articles (6%) as proceedings and 38 articles as review articles (2%). The distribution of citations and articles over the 20 years can reflect a research field's leading trends and evolution. Figure 1 shows the total number of citations and publications from 2003 to 2023. Bibliometrics is recognized as an appropriate method for mapping scientific activity in a given field, allowing both qualitative and quantitative assessment of scientific productivity through statistical and mathematical techniques [38].
Figure 2 displays the distribution of publications and citations over 20 years. The first article was published in 2003, stating that fixed film bioreactor technology showed higher removal rates and chemical oxygen demand (COD) removal rates of 85–90% and degradation of almost 100% of phenol from industrial oil refinery effluent [39]. A steady increase in annual publications was observed, with significant growth in the following three years. However, 2007, 2010, 2011, 2016, 2017, 2022, and 2023 showed a slight decrease in publications. A possible reason for the decreasing trend in the years above could be attributed to fluctuations in research prioritization, changes in funding availability, or temporary saturation of the topic, leading to a reorientation of research efforts to other areas or a temporary decrease in the number of publications. The period from 2018 to 2021 was marked by a remarkable and qualitative leap in scientific production, reflected in a significant increase. In particular, 2021 was the peak of publications, with 188 annual publications and more than 10 thousand citations. This scenario indicates that research on wastewater treatment has emerged as a central focus, generating remarkable enthusiasm among scientists.
3.2 Study of authors, journals, countries, and institutions
Table 1 shows the ranking of the ten most cited authors, journals, countries, and institutions in descending order of citation. Data also include the number of publications. Out of 4080 authors identified in this study, statistical analysis shows that the ten most cited authors have accumulated 5120 citations across 44 scientific publications. These results highlight the concentration of citations in a select group of authors. Among them, the three most cited authors were Dionysios D. Dionysiou (1149 citations in 2 publications), K. Palanivelu (633 citations in 3 publications), and Nihar Biswas (464 citations in 2 publications). Notably, they had few publications. Figure 3B shows that the most productive authors, Minghua Zhou (424 citations in ten publications) and Hongjun Han (316 citations in ten publications), ranked eighth and ninth in Table 1.
Figure 3A shows the leading networks of collaboration clusters among authors who published at least two documents with at least two citations. Again, Minghua Zhou and Hongjun Han appear to be the most significant authors within their groupings or clusters, represented by the pink and red colours. Minghua Zhou (pink cluster) contributes heavily to authors Xiaoye Lu, Jie Yang, and Qi Wang, while Hongjun Han (red cluster) contributes heavily to authors Yuxing Han and Wercheng Ma. The cluster network's node size and link line thickness confirm the results. Although these ten authors are gaining prominence, statistical analysis shows they contribute only 2.38% of the total publications in the field studied. This low representation suggests that, despite the significant influence of these authors, research in the area is diverse, with a wide variety of contributions from different researchers. This can be explained by the fact that the academic community increasingly values the quality of published work over mere quantity since authors with relatively few articles receive many citations in our analysis.
To analyze the journals prepared by VOSviewer, we observed the ten most cited journals out of 368. the journal with the highest number of citations is the Dutch Journal of Hazardous Materials (4229 citations in 54 publications), reaching an average of 78.31 citations per publication. Figure 3C shows the citation dimension of the prominent journals in different clusters (in moss green, blue, purple, and green), in which the Journal of Hazardous Materials appears as central within its cluster (moss green). However, regarding the number of articles published (as seen in Fig. 3D), it is only second to the American journal Desalination and Water Treatment, which had the highest number of publications with 64 publications and 835 citations. Once again, we found that the level of citations is not necessarily related to the number of publications. This depends on several factors, including the journal's impact factor. The impact factor (IF) is an important metric used to measure the recognition of journals in the academic world. In this case, the Journal of Hazardous Materials has an impact factor of 13.6 years (2023), a high value compared to the low value of 1.273 for the impact factor of the journal Desalination and Water Treatment year (2023). However, considering only the impact factor is insufficient to explain a journal's recognition and impact [40, 41]. An example is the Dutch journal Applied Catalysis B-Environmental, which has an impact factor of 22.1 years (2023), the highest in the ranking, but does not appear among the first journals published the most. Factors such as area of research concentration, specific fields of investigation, and places of interest may contribute to the preference for particular journals.
The database analysis also provides information on the region/country of origin indicated by the corresponding authors of the published articles. In Fig. 4A, we can see a network of coloured clusters formed by different countries, grouped according to their degree of collaboration and connected by lines. Among all these countries, represented by “nodes”, one node stands out due to its size and the number of connections with other countries. This country is China, which obtained the highest number of publications (1,512, 81.8% of the total) and a significant number of citations (11,491). In Fig. 4B, we confirm these data, and we get India in second (2862 citations and 282 publications) and third place Iran (2781 citations and 280 publications). India had the highest average number of citations per publication among the top three countries. China also stands out in terms of recent publications in the field studied. In Fig. 4C, compared to other countries, the highlighted “node” of China shows, through warm and cold colours, the significant contribution of this country to the number of publications. We also note that the participation of developed countries, such as the US, Spain, and France, indicates that research is not limited to specific regions. Although Asia leads as the continent with the most published and cited titles, other continents also contribute, covering Europe and the Americas, suggesting a global approach to the challenges associated with phenol wastewater treatment. The prominent presence of Asian countries, including China, India, South Korea, and Malaysia, highlights the considerable attention paid to environmental issues in the region. Such issues may be influenced by local needs, the concentration of industries generating phenolic waste, stringent environmental policies, and significant investments in research and development [42, 43].
Country analysis. A Visualization of the network map of the countries that most published, composed of at least four published articles, the thickness of the lines in the visualization indicates the strength of the co-authorship links that connect two countries and the most intense nuclei, divided into clusters, illustrate the groups of countries that published more significantly among themselves. B Distribution of the 25 countries that published the most in the research area, and C Co-authorship analysis among the leading research countries over 20 years
Figure 5A illustrates the major institutions organized in coloured clusters, highlighting those that are significant in terms of publications and their collaborative relationships. In particular, the Chinese Academy of Sciences, represented by the moss-green “knot”, occupies a central position, confirming its leadership in research on phenolic water treatment. Furthermore, the presence of the Indian institution Anna University in the same cluster indicates a close collaboration with the Chinese institution. Based on the affiliation of the corresponding authors, the three most prominent institutions in terms of publications were Chinese, namely the Harbin Institute of Technology, Dalian University of Technology, and the Chinese Academy of Sciences. These institutions contributed 93 publications (5.03% of the total) and accumulated 3,527 citations. The Chinese Academy of Sciences stood out with the highest average number of citations per publication, reaching 86.16. Figure 5B shows the three institutions mentioned above but were not necessarily the first most cited.
3.3 Most cited articles in the research area
Table 2 presents a compilation of the ten most cited articles, arranged in descending order of citations. Analysis of these publications reveals the central themes of the research. Examining the most influential documents makes it possible to understand the authors' approach to specific content and its direct relevance to the topic under study [35]. The most cited article, “The use of zero-valent iron for groundwater remediation and wastewater treatment: A review, " was published in the Journal of Hazardous Materials in 2014, with 1,144 citations and an average of 104 citations per year to date. The article comprehensively reviews recent advances in zero-valent iron (ZVI), groundwater remediation, and wastewater treatment. The review concludes that there is growing interest in using ZVI to remove various contaminants from groundwater and wastewater, including chlorinated organic compounds, nitroaromatics, arsenic, heavy metals, nitrate, dyes, and phenol [44].
The second article, “Photocatalytic degradation of azo dye acid red 14 in water: investigation of the effect of operational parameters”, ranks second with 787 citations, an average of 35.77 citations per year. This paper, published in the Journal of Photochemistry and Photobiology A-Chemistry, is the oldest on the list, dating back to 2003. The authors investigated how the UV/TiO2 process can be used efficiently to degrade a dye in water, identifying the ideal conditions and factors that influence the effectiveness of the process. Although their main focus is degrading dyes, their findings have valuable implications for treating wastewater containing phenolic compounds. The study highlights the importance of optimising treatment conditions by analyzing operational parameters and the influence of supporting agents, such as hydrogen peroxide. These insights can be applied to the development of phenolic wastewater treatment technologies. The UV/TiO2 method was found to be effective in degrading acid red 14, with a significantly reduced photodegradation efficiency in the absence of TiO2 and virtually zero in the absence of UV light. It is also highlighted that the controlled addition of hydrogen peroxide improved the degradation rate. However, at high concentrations, H2O2 neutralizes hydroxyl radicals [45].
The third most cited article in 2020 was authored by Dongjie Chen and collaborators, published in 2020, has 685 citations and an annual average of 137 citations, titled “Photocatalytic degradation of organic pollutants using TiO2-based photocatalysts: A review”. It has provided fundamental information on removing organic pollutants from wastewater effluents. In addition, the study reviewed several critical parameters that affect TiO2 photocatalytic degradation, such as dopant/catalyst concentration, particle size, pH adjustment, and substrate selection. It highlighted the importance of these factors for the future commercialization of TiO2 photocatalysis [46]. This study contributes to the advancement of scientific knowledge. It offers information directly contributing to advancing and understanding wastewater treatment technologies containing phenols. It provides valuable insights for researching and developing more effective methods of treating contaminated water with phenols.
3.4 Research areas
Figure 6A outlines the essential research areas that have attracted the attention of researchers in the advancement of phenol-containing wastewater treatment from 2003 to 2023. In analysing selected articles from the WoS database, Engineering (32%) and Environmental Sciences Ecology (25%) stand out as the two predominant areas, accounting for 57% of the total with 1687 published papers. Chemistry follows with 12%, corresponding to 347 records, and water resources with 11%, corresponding to 321 publications. The field of biotechnology and applied microbiology contributes 6%, represented by 184 publications. The last five fields, Materials Science (4%), Energy Fuels (3%), Science Technology Other Topics (3%), Physics (2%), and Electrochemistry (2%), account for a total of 412 publications. This distribution highlights the multidisciplinary approach to address the challenges of efficiently treating phenol-contaminated waters [47,48,49].
In addition, Fig. 6B complements this analysis by clearly visualising the relative differences between the top 10 highlighted areas and the number of publications in ascending order. This information provides a comprehensive overview of the regions. It demonstrates the active involvement of the academic community in addressing the environmental challenges associated with the presence of phenol in wastewater. The diversity of disciplines not only underscores the complexity of the problem but also opens up promising opportunities for developing advanced oxidation technologies. Some methods include using photocatalysis, cavitation, hydrogen peroxide, and combinations of these techniques to optimize phenol degradation more efficiently and sustainably within the same project [50,51,52,53,54,55,56].
3.5 Advanced keyword co-occurrence analysis
Keyword co-occurrence analysis plays an essential role in research development. This procedure allows it to visualize knowledge networks and relationships between concepts and identify emerging scientific literature trends [57, 58].
Table 3 shows the 20 keywords in ranking order and the total link strength in the analyzed articles. “Phenol” (568 citations), “degradation” (529 citations), and “removal” (413 citations) are the most prominent keywords and are the same topics of this analysis. “Biodegradation” (212 citations), “oxidation” (210 citations), and “effluent treatment” (146 citations) follow in fourth, fifth, and sixth place, respectively. Among the 3946 keywords analyzed, 383 met VOSviewer’s accuracy criteria, considering a minimum of 5 occurrences per keyword in our search. This bibliometric analysis of the predominant connections and relationships between the most searched keywords in the investigated articles [59].
Figure 7A displays a map of the visualization network where the keywords are organized into three broad clusters, each identified by a different colour, highlighting those with the highest frequency of participation. The red cluster stands out notably, highlighting the keywords "phenol", "degradation", and "removal", interconnected with other areas, representing the most significant cluster related to the research topic. The second highlighted cluster, identified by green, encompasses waste treatment and the "archeous solution". Then, in the third cluster, represented by yellow, the keywords "electrochemical oxidation" and "organic pollutants" stand out. The blue colour is centred on "oxidation" and "adsorption" and is integrated with the red cluster. The "kinetic" is highlighted in purple and is in the centre of the red and green cluster, indicating that a more significant number of connections between words implies a more extensive or closer cluster. This visual representation provides a clear view of the associations and groupings of the analyzed keywords.
Figure 7B shows the frequency of keywords within the four years in clusters of different colours, indicating the year of publication. The research projections are from 2020 to 2023. The keywords “phenol”, “degradation”, and “removal” have the most significant clusters, emerging as a central focus in 2020. Furthermore, these keywords may be correlated with others within the same research area. Figure 7C shows the frequency of the top 10 most cited keywords from 2019 to 2023. Degradation stands out as the most prominent word, with a total of 2151 occurrences, with an average of 430.2 over these five years, indicating that the search for these keywords is predominantly associated with the field of environmental research, especially in studies related to the treatment of contaminated water and industrial waste. These keywords are often found in articles that explore methods and technologies for dealing with pollutants in aquatic environments.
Table 4 was created to present the three most significant clusters identified in Fig. 7A, containing the frequency of cited keywords based on 383 articles that meet the limit of five publications. Topics such as wastewater treatment, organic pollutant degradation processes, and removal methods are recurrent in this context, reflecting the predominant interest in environmental research.
Meanwhile, a graphical representation constructed from the analysis using CiteSpace software is presented in Fig. 8A, characterized as a comprehensive bibliometric analysis tool, where 458 keywords and 851 occurrences were identified among the articles in the database data. From January 2004 to January 2024, the top 43 keywords were highlighted. Figure 8A shows them. Therefore, the keyword “wastewater treatment” was the most explicit, being used in more than 57 articles, directly highlighting the greater incidence of studies on wastewater treatment. This may be due to the lack of adequate treatment of these industrial wastes or the current environmental urgency to minimize damage to the ecological system. Keywords such as “biological treatment” (17 occurrences), “phenol degradation” (26 occurrences), and “electrochemical oxidation” (35 occurrences) stand out in the scenario as exciting possibilities for solving this general problem. Biological treatment is one of the most accepted by environmentalists because it is based on using live bacteria that assimilate pollutants (such as organic matter) and thus purify wastewater. Nevertheless, a consolidated trend can be identified regarding future research for wastewater treatment, where some of the strategies above will be the most prominent on research platforms.
In addition, Fig. 8B displays a distribution graph on the timeline of the keywords of the ten main clusters in this research area. Among the main clusters, the term wastewater treatment is at the centre, which explains its centrality to work. The other clusters are directly related to the central term, juxtaposed with the tools for solving the central problem under analysis. The concepts of electrochemistry and biological treatment are the most obvious, confirming themselves once again as promising possibilities for adequate wastewater treatment. The formation of four clusters around the wastewater treatment process can also be seen. One of these four clusters directly relates to five essential concepts: catalysis, phenol degradation, biological treatment, coking wastewater, and olive oil mill wastewater. The promising possibilities for wastewater treatment can be seen once again. Thus, the emphasis on the other critical points of the research review is reaffirmed.
Next, Fig. 9 displays a timeline view of the articles studied from January 2013 to mid-January 2024, in which we observe the most important keywords for each main keyword, that is, words that indicate the lines to be studied. Therefore, the central keyword #0 is “wastewater treatment”, which is related to the issue of the efficiency of the process of removal/treatment of phenol from wastewater, as the world is going through an attempt to minimize the industrial impact on the planet. In Cluster #1 (Electrochemical Oxidation), aspects related to efficiency in wastewater treatment. The oxidation process via electrochemistry of pollutants can effectively oxidize many inorganic and organic contaminants. In 2013 and 2019, the concentration of work publications was highest. In Cluster #2 (Biological Treatment), research is conducted on using organic materials such as enzymes and bacteria to purify wastewater. In 2014 and 2017, the concentration of work publications was highest. In Cluster #3 (Wastewater Coking), the highest concentration of publications was in 2013 and 2018. The coking of wastewater presents itself as a promising and efficient alternative to wastewater treatment.
4 Specific research topics
4.1 Sources and impacts of phenol in wastewater
Phenol, an organic molecule, has one or more hydroxyl groups (OH) attached to one or more of its aromatic rings (Fig. 10). Phenol is a chemical used in many industrial processes, including the manufacture of resins, plastics, insecticides, and pharmaceuticals [60]. Any living organism, including humans, animals, and plants, can be at risk from phenol waste released into the environment [61].
The presence of phenol in wastewater occurs in two ways: naturally and as a result of human activity [62]. The decomposition of dead plants and animals indicates natural occurrence [63], whereas disposal by industry, agriculture, municipal solid waste, and household waste indicates anthropogenic activity. Phenol is also produced as a by-product of petroleum refining [64]. Due to its widespread use and improper disposal, the chemical is often detected in wastewater, making it critical to investigate the sources (Fig. 11) and assess the impact of the substance on the aquatic ecosystem and its interaction with the environment.
The material is classified as a priority pollutant by the National Pollutant Release Inventory (NPRI) and the US Environmental Protection Agency (USEP) [65]. This is because phenol is exceptionally hazardous and highly reactive, even at low doses[66]. Consequently, interacting with aquatic life disrupts organisms' metabolism, subsequently impacting their developmental processes [67]. Therefore, the release of phenol into the environment is restricted by international regulatory authorities. The USEP sets a strict water purity standard that limits the amount of phenol at the water’s surface to 1 ppb [68]. In addition, these species die when exposed to large amounts of it.
The long-term persistence of phenol in the environment increases its impact and potential to contaminate drinking water sources [69]. Since phenol risks human health when found in surface or groundwater, adequate treatment is required [70]. Exposure to phenol can cause respiratory problems, liver and kidney damage, eye and skin irritation, and other side effects [71].
Thus, measures and processes are needed to control and treat effluents in the industrial production chain [72]. For example, phenol removal systems can be installed to remove the substance before it is discharged into the environment, and alternative manufacturing techniques can be developed to reduce the amount of waste that is produced that may contain phenol. From a regulatory perspective, environmental regulations must be designed to ensure that phenol concentrations are within a safe range for public health and the environment.
4.2 Conventional phenol treatment methods
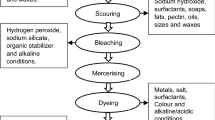
Table 5 summarizes the pros and cons of the phenol treatment method. This analysis is essential for selecting the most appropriate method for each situation, considering several factors, such as efficiency, availability, speed of action, ease of use and environmental impact. By understanding the pros and cons of each approach, professionals involved in chemical waste management can make more informed and effective decisions to ensure treatment effectiveness and protection of the environment. Phenols can have a significant environmental impact due to their high toxicity and solubility in aquatic environments [73]. Therefore, several treatment strategies have been developed recently, highlighted in Fig. 12, and their environmental and economic aspects have been evaluated.
4.2.1 Electrochemical oxidation
Electrochemical oxidation removes phenol by passing an electrical current through electrodes (made of platinum, graphite, or lead oxide) [74]. As a result, the electrical current plays an oxidizing role, causing the degradation of the organic molecule and releasing it to less dangerous components such as carbon dioxide and water [75]. Advantages include reduced production of unwanted byproducts and the ability to handle a wide range of phenol concentrations [76]. However, to guarantee the effectiveness and economic sustainability of the process, the electrodes must be selected appropriately, and the operating conditions must be analyzed [77]. Electrochemical techniques may present themselves as viable options for treating phenol as they do not require reagent costs and do not lead to the formation of new by-products. However, they do require the assembly of apparatus and electrochemical cells for the process.
4.2.2 Photocatalytic techniques
Photocatalytic techniques use natural or artificial light to degrade phenolic compounds. This process is efficient and environmentally friendly due to the low or zero consumption of chemical reagents [78]. A disadvantage from an economic point of view is that although the performance and cost of photocatalytic cells are favourable, they have a low reuse rate, i.e., the valuable life per cell is significantly reduced [79].
4.2.3 Enzymatic treatment
The use of biocatalysts in the degradation of polluting organic compounds is not new and has been explored by industry for many years [80]. This is because enzymes have high catalytic efficiency, are highly selective catalysts, and can operate in different temperature and pH ranges. The yield of these reactions is also of great interest, as enzymes, combined with immobilization techniques, can perform several reuse cycles without losing their catalytic efficiency [81]. An advantage of using enzymes is their solubility, which forms a homogeneous system in this catalysis, making removing phenolic compounds more efficient. Enzymatic treatment can also be used as one of the steps in other combined processes to optimize the residual phenol treatment route. A disadvantage is the need to isolate the enzymes and the low stability to high temperature and acid conditions [82].
4.2.4 Membrane method
Other very efficient techniques that offer several advantages for removing residual phenol are those that use membrane technologies. They are based on the difference in concentration, pressure, or temperature [83]. The membrane process has been studied for its efficiency and adaptability. This method uses semipermeable membranes that allow selective passage of water and other soluble components while retaining contaminants (e.g., phenol) [84]. The most common processes include “reverse osmosis”, which removes impurities from water by using pressure to force water through a semi-permeable membrane, leaving behind contaminants [85]. "Nanofiltration" uses membranes with nanopores to remove organic molecules and larger particles from water while allowing the passage of water and some minerals [86, 87]. “Ultrafiltration” uses a semipermeable membrane to remove particles, colloids, microorganisms and macromolecules [88]. These treatments can be used alone or in combination if specific criteria for phenol removal are met [89]. Advantages include low use of chemical reagents, high efficiency, and adaptability for operation in various environmental conditions. These methods are very reliable, reproducible and have low operating costs. One of their disadvantages is the encrustation of materials adhering to their surface, which reduces the activity of the membrane [90]. The primary membrane methods are listed in Table 6.
4.2.5 Extraction method
This technique is based on extracting residual phenol from water samples using eutectic solvents or specific types of surfactants. Extraction systems are set up to handle high volumes of phenol [91]. This technique can also be used as a preliminary step to other combined processes, usually in more concentrated samples, and has a high efficiency in recovering phenols [92]. A disadvantage of the method is the experimental time, which can be pretty long in some cases of high concentrations [93].
4.2.6 Adsorption processes
Using adsorption techniques to remove phenol from the aqueous medium also presents an excellent alternative for removing this residue [94]. When an adsorbent material is added to contaminated water samples, phenolic compounds adsorb to the material's surface. This can then be washed with methanol, producing a phenol mixture [95]. A significant advantage of this technique is its simplicity of operation, resulting in efficient and rapid removal of these products [96]. A disadvantage is that most adsorbents require thermal activation of the material and reactivation after use with chemical reagents [97].
4.2.7 Oxidation
Chemical oxidation efficiently removes Phenol from wastewater, using strong oxidants to degrade organic compounds into smaller or easily removed molecules [98]. The most common oxidants are ferrates, permanganates, peroxides and ozone, and their use varies depending on the pH range, temperature and system specifications [99]. A particularity of this method is phenol degradation, even at low concentrations [100]. This process can be combined with other treatment techniques to improve phenol removal efficiency [101]. The advantages involve the cost and efficiency of the method concerning other processes that require equipment assembly and constant maintenance. A disadvantage is using chemical reagents during the steps, which can lead to more significant waste generation and, consequently, a demand for treating these materials. Thus, concerns about the disposal and treatment of these wastes can make this technique more complex [102].
4.2.8 Ozonation
The ozonation process is based on heterogeneous catalysis using metals as catalysts. The degradation of phenols with ozonation leads to the formation of free radicals with the help of metal catalysts [103]. One of the advantages of the ozonation process is the operating time and catalytic efficiency. Although noble metal catalysts have cost disadvantages, they can be replaced by less expensive transition metals [104].
4.2.9 Distillation method
This method is based on differences in the boiling points of phenolic compounds and their relative volatilities [105]. The advantage of the technique is its efficiency on concentrated samples of phenolic compounds and large volumes of wastewater [106]. Disadvantages include low yield on samples with low phenol concentrations and higher operating costs [107].
4.2.10 Biological treatment
Based on aerobic and anaerobic processes of microorganisms, this technique presents itself as an excellent alternative for treating and degrading phenol in water. Advantages such as low operating costs and versatility in selecting different microorganisms make the technique viable [108]. Another positive aspect is the production of biodegradable materials that do not pose a pollution risk to the soil, making these processes an efficient and environmentally sound choice for treating pollutants [109]. A drawback is that these processes are only effective at low concentrations, as a high concentration of phenols interferes with the degradation activity of microorganisms. Thermal and acidic conditions must also be adapted and controlled for each process [110].
5 Recent technological advances
More effective and sustainable systems for phenol removal from wastewater are being developed. Diverse and improved methods are being developed to address the problems associated with phenol in municipal and industrial wastewater.
Nanotechnology provides new perspectives for wastewater treatment, with nanomaterials based on graphene (Gr) standing out for their excellent magnetic, electrical, optical, mechanical and catalytic properties, having a large surface area and high purity [111,112,113]. Graphene, a two-dimensional material known for its remarkable mechanical, electrical and thermal properties, has emerged as a promising choice for environmental applications, including the treatment of contaminated water [114]. Carbon nanotubes and nanocomposites exhibit greater surface area and chemical reactivity, making them highly effective at adsorbing and degrading organic and inorganic contaminants, including phenol [115]. Furthermore, a relatively small number of researchers have recently explored using green synthetic nanoparticles in wastewater purification, indicating a promising advancement in the search for more effective and sustainable solutions [116, 117]. This broad, integrated approach enables a deeper understanding of emerging trends and future opportunities in the field of wastewater treatment, helping to guide future research and engineering practices aimed at continually improving water quality and preserving the environment.
On the other hand, recent studies in the application of artificial intelligence (AI) have been contributing to phytobial remediation involving the use of machine learning algorithms and models to analyze and interpret data related to plant growth and the effectiveness of removing contaminants from plant bodies. 'water [118]. Furthermore, AI enables real-time analysis of large volumes of data, allowing for early detection of phenol concentration spikes and rapid adaptation of treatment strategies. The successful adoption of these technologies drives further research and innovation in structural models to overcome the limitations faced in the effective operation within water treatment industries [119]. Although AI models help treat wastewater with phenol, their disadvantages cannot be overlooked. Challenges such as the demand for large volumes of data, poor management of this data, low interpretability of the models, limited reproducibility, and lack of transparency pose significant obstacles to their effective use [120].
Ultrasound technology has attracted attention as a promising approach to improving the efficiency of wastewater treatment [121,122,123]. In this process, high-frequency ultrasound is applied to contaminated water, generating cavitation bubbles imploring under pressure [124,125,126]. It breaks these compounds into smaller fragments by generating explosions that create micro-liquid jets and shock waves, facilitating their treatment. [127, 128]. However, its high energy consumption represents a significant disadvantage, resulting in high operating costs [129].
Furthermore, in waters with a high concentration of suspended solids, the effectiveness of the technology may be compromised due to obstruction of cavitation bubbles [130,131,132]. Despite these limitations, Ultrasound Technology is widely applied in several industries, such as chemical products, to treat effluents contaminated with phenols [133, 134]. A real example of this application is found in chemical factories, where ultrasound fragments organic compounds, allowing them to be removed by subsequent treatment processes such as flocculation, filtration or sedimentation [135, 136].
Activated Carbon Technology is an established option for removing organic contaminants, including phenols, from water [137, 138]. Recent advances in technology have resulted in the development of more effective forms of activated carbon, such as granular activated carbon (CAG) and powdered activated carbon (PAC) [139,140,141]. These advanced forms have an even greater adsorption capacity and improved phenol removal efficiency, thus contributing to improved water quality in a variety of industrial and environmental applications [142, 143]. Regarding the advantages of Activated Charcoal, its effectiveness in removing flavors and undesirable odors, eliminating contaminants such as heavy metals, toxic gases, and reducing natural organic matter, which limits the formation of by-products, stands out [144,145,146]. However, Activated Charcoal also has disadvantages. Its large-scale use can be expensive [147, 148]. Activated Carbon's adsorption capacity is limited, meaning it can only trap a limited amount of pollutants before becoming ineffective [149, 150]. Therefore, Activated Carbon Technology plays a crucial role in mitigating climate change by demonstrating its value and effectiveness in real-world applications.
Supercritical water gasification is a new way to treat phenol in wastewater because it subjects the water to supercritical conditions (high pressures and temperatures above the critical point of water) to create a reactive medium [151]. The phenol then combines with water vapour and breaks into less harmful compounds [152]. Supercritical water gasification is still in the research and development stage. Although it offers benefits such as eliminating the need for additional chemical products, reducing costs, and minimizing the environmental impact of treating polluted wastewater, it is still a work in progress [153].
Finally, ozonation is a sophisticated approach to address phenol removal in wastewater due to its ability to oxidize organic molecules. In the ozonation process, ozone (O3) is introduced into the water to react with phenol, resulting in oxidation and the formation of less hazardous chemical compounds (such as phenic acid and carbon dioxide) [154, 155]. This process has several advantages, including adaptability, speed of reaction, and the absence of harmful by-products [156]. Ozone is an oxidant that can eliminate a wide range of organic contaminants in wastewater. Challenges to overcome include the equipment cost needed to produce and administer ozone [157, 158].
6 Future trends and opportunities
The methods presented in the work show a vital concern with chemical waste treatment that has gained momentum in recent research [159]. The high volume of work highlighting the use and concepts of removing phenol from wastewater confirms this pursuit of developing other innovative and low-cost techniques for handling organic substances [160, 161]. The environmental impacts of generating these materials show the need for this ecological responsibility, which must take even more excellent shape in industry, especially in the petrochemical and pharmaceutical areas [162]. Among the techniques and processes presented in this work, innovations in adsorption techniques and using enzymes or microorganisms stand out. In adsorption, innovations in the materials used and their association with nanotechnology stand out, which, in addition to being innovative and technological, has a high rate of efficiency and reproducibility [163, 164]. The economic factor also increases the spotlight in business sectors on this technique, which leads to a demand for improving its stages. In the case of enzymes, many current works tested have their use with immobilization supports, where different materials are tested to increase their thermal and pH stability, maintain their catalytic activity, and the need for low concentrations of enzymes [165, 166]. From this optimization that the support provides to the biocatalyst, properties such as high selectivity and yield under environmental conditions are added to the use of enzymes for handling residual phenols in water samples [167,168,169]. As a result, several research groups focus on improving, discovering, and optimising efficient adsorbents and ideal enzymatic solutions, both for processes combined with others and for their single technique.
7 Environmental, regulatory aspects, and financial viability
The primary sources of water pollution are agricultural activities, sewage systems, and commercial, domestic, and industrial waste discharge. The sources generate about two million tons of wastewater daily [170]. One of the most prevalent toxic substances is phenol, a critical component in producing pesticides, dyes, pharmaceuticals, and plastics [171]. Phenol production has reached an annual volume of 8.9 million tons, making it a priority pollutant due to its high toxicity even at minimal concentrations [172]. Exposure to phenol, which can occur through the skin, oesophagus, and respiratory tract, poses potential risks to the central nervous system, liver, and kidneys [173].
Phenol exists in the atmosphere, mainly in the gas phase [174]. The atmospheric half-life of phenol varies from 2.28 to 22.8 h, depending on conditions and reactions with the hydroxyl radical [175]. Highly soluble in water (88,360 mg/L) and most organic solvents, phenol acts as a weak acid (pKa = 9.99), and its toxicity is influenced by environmental pH [176,177,178]. The slower penetration of the phenolate ion through biological membranes than the neutral molecule explains the inverse relationship between water pH and phenol toxicity—higher water pH correlates with lower toxicity.
The US Environmental Protection Agency (US EPA) study ranked phenol eleventh among one hundred and twenty-six chemical pollutants [179, 180]. Concentrations above 50 ppm are associated with health problems such as neurological damage, kidney dysfunction, and decreased blood haemoglobin levels [181]. Phenolic derivatives, such as 2,4-dichlorophenol, exhibit mutagenic properties and cause liver damage at concentrations as low as 0.002 ppm [182]. Consequently, the US EPA has set the limit for phenol at 0.001 ppm in drinking water and 0.5 ppm in wastewater [183]. Because of its high toxicity, phenol has become a critical quality parameter closely monitored by regulatory agencies, especially concerning industrial effluents [184]. Various methods have been proposed for treating phenol in wastewater, including extraction, adsorption, chemical oxidation, biochemical oxidation, and electrochemical processes. Each technique has significant drawbacks regarding cost, by-product formation, and incomplete removal [185]. To ensure effectiveness and economic viability, selecting the appropriate effluent extraction and treatment technologies is critical [186, 187].
In 2015, the United Nations Sustainable Development Goal 6 (UN SDG6) highlighted the need to recycle wastewater to ensure water availability for individuals [188, 189]. Notably, the use of treated wastewater in agriculture is a promising application to address the scarcity of water resources, mainly caused by climate change and the increase in water consumption resulting from human activities [190].
However, the challenges, limitations, and opportunities in successful wastewater reuse have been widely discussed by various experts who are trying to achieve sustainable wastewater recycling [191]. In many regions of the world, environmental restrictions, aggravated by recurrent and prolonged droughts due to the effects of climate change, have led communities to explore the potential of treated waters as an alternative to meet their water needs, especially in applications that do not require the strict drinking water quality standards [192,193,194,195].
Yet, according to Chojnacka et al., anti-pollution legislation has significantly reduced discharges of harmful materials into coastal waterways over the past 40 years [196]. Accordingly, the popularity of wastewater reuse has increased across the planet, influencing several countries to adopt local legislation to regulate the quality of water for reuse to reduce risks to health and the environment [197]. In summary, regulatory laws for wastewater treatment are being updated and adapting to environmental and economic demands, seeking to mainly benefit the planet and safeguard the future population [198].
8 Sector applications and specific challenges
Phenol and its derivatives do not occur naturally in water. Their presence is invariably associated with human activities, particularly in industries (e.g., pharmaceuticals, mining, petrochemicals, and paper and pulp) [199]. The concentration of phenolic compounds in industrial wastewater typically ranges from 800 to 2000 ppm [200]. Out of the various treatment techniques, biodegradation has emerged as a notable method due to its environmental soundness and cost-effectiveness. Biodegradation is based on the concept that certain microorganisms can utilize phenol as their sole energy source and carbon. Studies indicate that indigenous bacterial species adapted to specific effluents excel in overcoming competition from other organisms, emphasizing the need to identify novel phenol-degrading bacterial species [201]. Two bacterial strains, Pseudomonas aeruginosa and Pseudomonas pseudomallei, were used to treat wastewater from the pharmaceutical industry. The P. aeruginosa strain showed a 26.16 mg L-1 h-1 phenol degradation rate. TP. pseudomallei strain showed a rate of 13.85 mg L−1 h−1, demonstrating high efficiency in degrading phenol from the specific effluent [202]. One hundred and eight rhizobial strains were isolated from wild legumes in a mining tailings area in Shaanxi Province, China. The strain CCNWTB701, isolated from Astragalus chrysopteru, showed remarkable efficiency in degrading phenol, achieving a 99.5% reduction from an initial concentration of 900 ppm within a 62-h interval [203].
Because of the low permissible limit for phenol as a pollutant, developing a sensitive and reliable sensor for monitoring this substance is essential. Colourimetric, chromatographic, or electrochemical methods can be used. In contrast, chromatographic methods have demonstrated the ability to achieve detection rates as low as four ppt when using single-drop microextraction and syringe derivatization [204]. A potential challenge is that much of the research on phenol removal in wastewater remains limited to laboratory conditions.
8.1 Innovations in materials and analytical methods
Innovations in materials and analytical methods for treating wastewater containing phenol have been fundamental in searching for more efficient and sustainable solutions [205]. In addition, materials functionalized with specific groups have excelled in the selectivity and effective removal of phenol in aqueous environments [206].
Continued research has led to promising new materials [207]. Some of these innovative materials include nanomaterials that have stood out. Mal oxide nanoparticles such as graphene, titanium dioxide, and zinc oxide have demonstrated significant efficacy [208,209,210,211]. Their adsorptive and photocatalytic properties effectively remove phenol, contributing to water purification [210].
Another promising category is metal–organic frameworks (MOFs), structures characterized by high porosity and surface area [212, 213]. These materials are effective in both the adsorption and degradation of phenolic compounds in wastewater [214]. The unique porosity of these MOFs makes them highly efficient in this treatment, providing an innovative approach to removing this contaminant [215].
Porous silicon carbide is characterized by its porous structure. Also, the remarkable chemical stability of porous silicon carbide contributes to the material's durability during the treatment process [216]. These innovative alternatives, coordination polymers, functional hydrogels, hierarchical porous carbons, and magnetic compounds, offer diverse options to meet specific needs in water treatment systems and provide more environmentally friendly solutions [217, 218]. The constant evolution of these materials reflects the continuous effort to develop new innovative, economical, and sustainable technologies for this research field.
Regarding analytical methods, new technologies have contributed to more accurate and real-time monitoring of phenol concentrations in wastewater [219]. Techniques such as high-performance liquid chromatography (HPLC), UV–Vis spectroscopy, and electrochemical methods have been improved to provide more sensitive and rapid analysis [220]. In addition, integrating advanced analytical methods, such as mass spectrometry and sensor-based techniques, allows for a more comprehensive assessment of phenol characteristics and transformations during treatment [221].
Innovative analytical methods are crucial in the treatment discussed here [222, 223]. Near-infrared spectroscopy (NIR) is a technique that offers rapid and non-destructive analysis [224]. In addition, gas chromatography coupled to mass spectrometry (GC–MS) offers high sensitivity and selectivity, allowing detailed analysis and precise identification of phenol, contributing to a deeper understanding of the components present [225].
In the field of sensors, notable advances include electrochemical sensors that, when specific for phenol, allow real-time monitoring [226]. In addition, environmental mass spectrometry (DART-MS) offers direct analysis of phenol in wastewater, eliminating the need for sample preparation and optimizing time and resources [227].
In addition, nanomaterial-based sensors using gold nanoparticles or carbon nanotubes show increased sensitivity in the detection of phenol [228,229,230,231]. Applying biosensory methods, such as enzymes or living organisms, or the selective detection of phenol represents a specific and environmentally friendly approach [232]. Finally, real-time UV–Vis spectroscopy can be used to monitor changes in phenol concentration during treatment, providing immediate insight into the effectiveness of ongoing processes.
These innovations in analytical methods improve the accuracy and sensitivity of phenol detection and facilitate the implementation of new strategies for treating wastewater contaminated with this compound [233]. Overall, the synergy between innovations in adsorbent materials and advances in analytical methods ultimately contributes significantly to environmental protection and public health.
8.2 Global and comparative perspectives between technologies
The choice of the most appropriate method for treating phenol-contaminated wastewater depends on the specific characteristics of the water. Out of the various options available, adsorption with activated carbon is an effective method due to its porous surface, which has an affinity for phenol [234]. Bioremediation, using microorganisms specialized for phenol degradation in aerobic or anaerobic processes, is an efficient approach [235]. Electrocoagulation, which uses electric fields to promote coagulation and phenol removal, is also recognized as an effective technique [236].
The technologies surrounding this issue are promising, and their results have the potential to change the current scenario significantly. In treating water contaminated with phenol, technological innovations have played a crucial role in the search for more effective and sustainable solutions. Various approaches, from traditional methods to state-of-the-art advances, have been explored to address the challenges posed by phenol in aquatic environments, offering several critical perspectives for progress in this field. The finding that the treatment method choice depends on the specific characteristics of the water, phenol concentrations, and environmental requirements highlights the need for a multidisciplinary approach. The diversity of techniques, from adsorption to bioremediation and electrocoagulation, highlights the importance of tailoring strategies to optimize treatment efficiency.
8.3 Practical applications of phenol wastewater treatment technologies
Several practical alternatives have been developed over the years for removing phenols, such as electrochemical oxidation, ozonation, UV/H2O2, Fenton reaction, and enzymatic treatment, among other processes [237,238,239,240].
In this sense, electrochemical oxidation (EO) uses electrical current or potential difference between two electrodes (anode and cathode), with which hydroxyl radicals or oxidizing species can be generated and used for the oxidation of pollutants [241]. The advancement of technology (EO) is no longer limited to treating simulated wastewater in a laboratory environment. It has currently been applied to the practical treatment of various sewage. However, its large-scale implementation still faces limitations due to high energy consumption [242, 243]. With the limitations of conventional treatment procedures, there is a pressing need to develop innovative and more environmentally acceptable methods for removing contaminants [244].
Still, in this field of alternative technologies, based on biological treatment principles, enzymatic treatments of an enzyme as a biocatalyst transform phenolic compounds, thus promoting their efficient removal from water [245]. Enzymes oxidize pollutants into simpler organic compounds [246]. Furthermore, enzymes are biological catalysts that accelerate biochemical reactions in living organisms and can be extracted from cells to facilitate various processes of great commercial importance [247]. The degradation of pollutants is rapid and selective [248]. Enzymes further increase their potential when immobilized on solid support[249].
As the most prominent example, peroxidases are enzymes widely used in phenol remediation due to heme cofactor or redox-active cysteine/selenocysteine residues in their active sites [250,251,252]. Several studies have investigated peroxidase and proven its efficiency in removing phenol from contaminated water. Kurnik et al. analyzed the application of peroxidases produced from potato pulp waste byproducts in removing phenol from synthetic and industrial wastewater. The phenol removal efficiency reached 95% with an initial phenol concentration of 94.11 mg/L, where enzymes maintained their activity over a pH range of 4 to 8 and remained stable over a wide temperature range [253].
In another study, peroxidases extracted from an invasive plant (Prosopis juliflora) were used for phenol remediation with an initial phenol concentration of approximately 40 mg/L, where crude peroxidases were found capable of degrading phenol in 30 min and with higher efficiencies. to 90% for textile and leather industrial wastewater [254]. This study showed that agricultural residues for different products could be used as an essential source of potent enzymes that are highly efficient in remediating phenolic compounds [255]. In summary, wastewater contains several pollutants that are harmful to life, including phenol in different concentrations [256].
The literature presents several alternatives for removing phenols in wastewater, with advantages and disadvantages. Few are applied on an industrial scale, but some previously presented are promising, as with enzymes. Depending on the concentration of pollutants, the type of treatment method can be selected, considering the cost-effectiveness of the treatment chosen systems. Therefore, it is imperative to analyze the components present in wastewater and understand their levels to use the most efficient treatment for a given sector.
9 Conclusion
The treatment of phenol-containing wastewater has proven to be a fascinating and promising field in modern science and technology. The convergence of unique properties opens up a wide range of applications in different fields, as demonstrated in this study, but also triggers a fascinating synergy between these elements. The bibliometric analysis highlights the growing interest and dynamism surrounding this domain by revealing a notable increase in publications on this topic at an accelerated rate. The publication “The use of zero-valent iron for groundwater remediation and wastewater treatment: A review” was highlighted as the most influential in the field, providing a comprehensive and fundamental overview for investigations. The results of the bibliometric analysis not only point to China as the leader in terms of article production, number of citations, and institutions involved but also highlight the notable collaborations between the prominent authors in this dynamic scenario. The diversity of article publication areas, revealing applications in different contexts and a multidisciplinary approach, highlights the breadth and relevance of treating wastewater containing phenols. Network analysis of the main keywords identified thematic clusters, with “phenol”, “degradation”, and “removal” being the most salient, signalling crucial areas of research. Given these advances and highlighted areas, future research in this area should prioritize wastewater treatment and feedstock diversification. This focus is essential to address significant challenges such as production costs, stability, and durability of treatment processes.
Data availability
For data availability inquiries, you can contact jcs@unilab.edu.br. All data used for this research will be made available to the journal as upon request.
References
Liu Z, Meng H, Zhang H, Cao J, Zhou K, Lian J. Highly efficient degradation of phenol wastewater by microwave induced H2O2-CuOx/GAC catalytic oxidation process. Sep Purif Technol. 2018;193:49–57. https://doi.org/10.1016/j.seppur.2017.11.010.
Shahedi A, Darban AK, Taghipour F, Jamshidi-Zanjani A. A review on industrial wastewater treatment via electrocoagulation processes. Curr Opin Electrochem. 2020;22:154–69. https://doi.org/10.1016/j.coelec.2020.05.009.
Jiang H, Chen H, Duan Z, Huang Z, Wei K. Research progress and trends of biochar in the field of wastewater treatment by electrochemical advanced oxidation processes (EAOPs): a bibliometric analysis. J Hazardous Mater Adv. 2023;10:100305. https://doi.org/10.1016/j.hazadv.2023.100305.
Ajiboye TO, Oyewo OA, Onwudiwe DC. Simultaneous removal of organics and heavy metals from industrial wastewater: a review. Chemosphere. 2021;262:128379. https://doi.org/10.1016/j.chemosphere.2020.128379.
Shukla P, Wang S, Sun H, Ang H-M, Tadé M. Adsorption and heterogeneous advanced oxidation of phenolic contaminants using Fe loaded mesoporous SBA-15 and H2O2. Chem Eng J. 2010;164:255–60. https://doi.org/10.1016/j.cej.2010.08.061.
Wang Y, Cao H, Chen L, Chen C, Duan X, Xie Y, Song W, Sun H, Wang S. Tailored synthesis of active reduced graphene oxides from waste graphite: Structural defects and pollutant-dependent reactive radicals in aqueous organics decontamination. Appl Catal B. 2018;229:71–80. https://doi.org/10.1016/j.apcatb.2018.02.010.
Kavian N, Asadollahfardi G, Hasanbeigi A, Delnavaz M, Samadi A. Degradation of phenol in wastewater through an integrated dielectric barrier discharge and Fenton/photo-Fenton process. Ecotoxicol Environ Saf. 2024;271:115937. https://doi.org/10.1016/j.ecoenv.2024.115937.
Samuel Olugbenga O, Goodness Adeleye P, Blessing Oladipupo S, Timothy Adeleye A, Igenepo John K. Biomass-derived biochar in wastewater treatment- a circular economy approach. Waste Manag Bull. 2024;1:1–14. https://doi.org/10.1016/j.wmb.2023.07.007.
Adeel M, Xu Y, Ren L-F, Shao J, He Y. Improvement of phenol separation and biodegradation from saline wastewater in extractive membrane bioreactor (EMBR). Bioresour Technol Rep. 2022;17:100897. https://doi.org/10.1016/j.biteb.2021.100897.
Montero-Guadarrama I, Balderas-Hernández P, Barrera-Díaz CE, Roa-Morales G. Phenol red degradation using a synergetic method (Electrochemical Oxidation with Ozone) in batch and continuous system. Int J Electrochem Sci. 2020;15:7883–95. https://doi.org/10.20964/2020.08.12.
Yu X, Wang K, Wang X. Electrochemical oxidation of phenol in chloride containing electrolyte using a carbon-coated Ti4O7 anode. Electrochim Acta. 2023;449:142233. https://doi.org/10.1016/j.electacta.2023.142233.
Cifuentes-Cabezas M, Pavani A, Vincent-Vela MC, Mendoza-Roca JA, Álvarez-Blanco S. Concentration of phenolic compounds from olive washing wastewater by forward osmosis using table olive fermentation brine as draw solution. Environ Technol Innov. 2023;30:103054. https://doi.org/10.1016/j.eti.2023.103054.
Su T, Xianyu B, Wang M, Gao W, Gao Y, Gao P, Lu C. Electrocatalytic removal of phenol from coking wastewater using coal based electrode materials. Int J Electrochem Sci. 2023;18:100401. https://doi.org/10.1016/j.ijoes.2023.100401.
Ochando-Pulido JM, González-Hernández R, Martinez-Ferez A. On the effect of the operating parameters for two-phase olive-oil washing wastewater combined phenolic compounds recovery and reclamation by novel ion exchange resins. Sep Purif Technol. 2018;195:50–9. https://doi.org/10.1016/j.seppur.2017.11.075.
Muñoz Sierra JD, Oosterkamp MJ, Spanjers H, van Lier JB. Effects of large salinity fluctuations on an anaerobic membrane bioreactor treating phenolic wastewater. Chem Eng J. 2021;417:129263. https://doi.org/10.1016/j.cej.2021.129263.
García Rea VS, Muñoz Sierra JD, El-Kalliny AS, Cerqueda-García D, Lindeboom REF, Spanjers H, van Lier JB. Syntrophic acetate oxidation having a key role in thermophilic phenol conversion in anaerobic membrane bioreactor under saline conditions. Chem Eng J. 2023;455:140305. https://doi.org/10.1016/j.cej.2022.140305.
Dionisi D, Etteh CC. Effect of process conditions on the aerobic biodegradation of phenol and paracetamol by open mixed microbial cultures. J Environ Chem Eng. 2019;7:103282. https://doi.org/10.1016/j.jece.2019.103282.
Wu Z, Jing J, Zhang K, Li W, Yang J, Shen J, Zhang S, Xu K, Zhang S, Zhu Y. Epitaxial BiP5O14 layer on BiOI nanosheets enhancing the photocatalytic degradation of phenol via interfacial internal-electric-field. Appl Catal B. 2022;307:121153. https://doi.org/10.1016/j.apcatb.2022.121153.
Elayadi F, Boumya W, Achak M, Chhiti Y, Alaoui FEM, Barka N, El Adlouni C. Experimental and modeling studies of the removal of phenolic compounds from olive mill wastewater by adsorption on sugarcane bagasse. Environ Challenges. 2021;4:100184. https://doi.org/10.1016/j.envc.2021.100184.
Xue Y, Chen Y, Shi L, Wu H, Zhang C, Cheng M, Li H, Li W, Niu Y. Lignite-based N-doped porous carbon as an efficient adsorbent for phenol adsorption. Processes. 2022. https://doi.org/10.3390/pr10091746.
Zango ZU, Sambudi NS, Jumbri K, Ramli A, Bakar NHHA, Saad B, Rozaini MNH, Isiyaka HA, Osman AM, Sulieman A. An overview and evaluation of highly porous adsorbent materials for polycyclic aromatic hydrocarbons and phenols removal from wastewater. Water (Switzerland). 2020;12:1–40. https://doi.org/10.3390/w12102921.
Al-Ananzeh N, Bani-Melhem K, Khasawneh HE, Tawalbeh M, Al-Qodah Z, Al-Bodour A. Investigating the potential of using solid waste generated from stone cutting factories for phenol removal from wastewater: a study of adsorption kinetics and isotherms. Results Eng. 2023;20:101404. https://doi.org/10.1016/j.rineng.2023.101404.
Cifuentes-Cabezas M, María Sanchez-Arévalo C, Antonio Mendoza-Roca J, Cinta Vincent-Vela M, Álvarez-Blanco S. Recovery of phenolic compounds from olive oil washing wastewater by adsorption/desorption process. Sep Purif Technol. 2022;298:121562. https://doi.org/10.1016/j.seppur.2022.121562.
Melo RLF, Sales MB, de Castro Bizerra V, de Sousa Junior PG, Cavalcante ALG, Freire TM, Neto FS, Bilal M, Jesionowski T, Soares JM, Fechine PBA, dos Santos JCS. Recent applications and future prospects of magnetic biocatalysts. Int J Biol Macromol. 2023;253:126709. https://doi.org/10.1016/j.ijbiomac.2023.126709.
Rodrigues AFS, da Silva AF, da Silva FLB, dos Santos KM, de Oliveira MP, Nobre MMR, Catumba BD, Sales MB, Silva ARM, Braz AKS, Cavalcante ALG, Alexandre JYNH, Junior PGS, Valério RBR, de Castro Bizerra V, dos Santos JCS. A scientometric analysis of research progress and trends in the design of laccase biocatalysts for the decolorization of synthetic dyes. Process Biochem. 2023;126:272–91. https://doi.org/10.1016/j.procbio.2023.01.014.
Catumba BD, Sales MB, Borges PT, Ribeiro Filho MN, Lopes AAS, de Sousa Rios MA, Desai AS, Bilal M, dos Santos JCS. Sustainability and challenges in hydrogen production: an advanced bibliometric analysis. Int J Hydrogen Energy. 2023;48:7975–92. https://doi.org/10.1016/j.ijhydene.2022.11.215.
Sales MB, Neto JGL, De Sousa Braz AK, De Sousa Junior PG, Melo RLF, Valério RBR, de Serpa JF, Da Silva Lima AM, De Lima RKC, Guimarães AP, de Souza MCM, Lopes AAS, de Rios MAS, Serafim LF, dos Santos JCS. Trends and opportunities in enzyme biosensors coupled to metal-organic frameworks (MOFs): an advanced bibliometric analysis. Electrochem. 2023;4:181–211. https://doi.org/10.3390/electrochem4020014.
Neto FS, de MeloNeta MMF, Sales MB, de Oliveira FAS, de CastroBizerra V, Lopes AAS, de SousaRios MA, dos Santos JCS. Research progress and trends on utilization of lignocellulosic residues as supports for enzyme immobilization via advanced bibliometric analysis. Polymers (Basel). 2023. https://doi.org/10.3390/polym15092057.
Nogueira RC, Neto FS, de Sousa Junior PG, Valério RBR, de França Serpa J, da Silva Lima AM, de Souza MCM, de Lima RKC, Lopes AAS, Guimarães AP, Melo RLF, de Sousa Rios MA, dos Santos JCS. Research trends and perspectives on hydrothermal gasification in producing biofuels. Energy Nexus. 2023;10:100199. https://doi.org/10.1016/j.nexus.2023.100199.
Ferreira VC, Ampese LC, Sganzerla WG, Colpini LMS, Forster-Carneiro T. An updated review of recent applications and future perspectives on the sustainable valorization of pitaya (Hylocereus spp) by-products. Sustain Chem Pharm. 2023;33:101070. https://doi.org/10.1016/j.scp.2023.101070.
Sales MB, Borges PT, Ribeiro Filho MN, Miranda da Silva LR, Castro AP, Sanders Lopes AA, Chaves de Lima RK, de Sousa Rios MA, dos Santos JCS. Sustainable feedstocks and challenges in biodiesel production: an advanced bibliometric analysis. Bioengineering. 2022. https://doi.org/10.3390/bioengineering9100539.
Guimarães CEC, Neto FS, de Castro Bizerra V, do Nascimento JGA, Valério RBR, de SousaJunior PG, de SousaBraz AK, Melo RLF, de FrançaSerpa J, de Lima RKC, Guimarães AP, de Souza MCM, Lopes AAS, de SousaRios MA, Desai AS, Bilal M, Smułek W, Jesionowski T, dos Santos JCS. Sustainable bioethanol production from first- and second-generation sugar-based feedstocks: advanced bibliometric analysis. Bioresour Technol Rep. 2023;23:101543. https://doi.org/10.1016/j.biteb.2023.101543.
Nabgan W, Ikram M, Alhassan M, Owgi AHK, Van Tran T, Parashuram L, Nordin AH, Djellabi R, Jalil AA, Medina F, Nordin ML. Bibliometric analysis and an overview of the application of the non-precious materials for pyrolysis reaction of plastic waste. Arab J Chem. 2023;16:104717. https://doi.org/10.1016/j.arabjc.2023.104717.
Borges PT, Sales MB, César Guimarães CE, de França Serpa J, de Lima RKC, Sanders Lopes AA, de Sousa Rios MA, Desai AS, da Silva Lima AM, Lora EES, dos Santos JCS. Photosynthetic green hydrogen: advances, challenges, opportunities, and prospects. Int J Hydrogen Energy. 2024;49:433–58. https://doi.org/10.1016/j.ijhydene.2023.09.075.
Melo RLF, Neto FS, Dari DN, Fernandes BCC, Freire TM, Fechine PBA, Soares JM, dos Santos JCS. A comprehensive review on enzyme-based biosensors: advanced analysis and emerging applications in nanomaterial-enzyme linkage. Int J Biol Macromol. 2024;264:130817. https://doi.org/10.1016/j.ijbiomac.2024.130817.
da Silva JL, Sales MB, de Castro Bizerra V, Nobre MMR, de Sousa Braz AK, da Silva Sousa P, Cavalcante ALG, Melo RLF, De SousaJunior PG, Neto FS, da Fonseca AM, dos Santos JCS. Lipase from yarrowia lipolytica: prospects as an industrial biocatalyst for biotechnological applications. Fermentation. 2023. https://doi.org/10.3390/fermentation9070581.
Chen Y, Lin M, Zhuang D. Wastewater treatment and emerging contaminants: bibliometric analysis. Chemosphere. 2022;297:133932. https://doi.org/10.1016/j.chemosphere.2022.133932.
Konur O. The scientometric evaluation of the research on the production of bioenergy from biomass. Biomass Bioenergy. 2012;47:504–15. https://doi.org/10.1016/j.biombioe.2012.09.047.
Jou C-JG, Huang G-C. A pilot study for oil refinery wastewater treatment using a fixed-film bioreactor. Adv Environ Res. 2003;7:463–9. https://doi.org/10.1016/S1093-0191(02)00016-3.
Eisner DA. Reproducibility of science: fraud, impact factors and carelessness. J Mol Cell Cardiol. 2018;114:364–8. https://doi.org/10.1016/J.YJMCC.2017.10.009.
Guo X, Li X, Yu Y. Publication delay adjusted impact factor: the effect of publication delay of articles on journal impact factor. J Informetr. 2021;15:101100. https://doi.org/10.1016/J.JOI.2020.101100.
Panigrahy N, Priyadarshini A, Sahoo MM, Verma AK, Daverey A, Sahoo NK. A comprehensive review on eco-toxicity and biodegradation of phenolics: recent progress and future outlook. Environ Technol Innov. 2022. https://doi.org/10.1016/j.eti.2022.102423.
Liao Z, Chen Z, Wu Y, Xu A, Liu J, Hu HY. Identification of development potentials and routes of wastewater treatment and reuse for Asian countries by key influential factors and prediction models. Resour Conserv Recycl. 2021. https://doi.org/10.1016/j.resconrec.2020.105259.
Fu F, Dionysiou DD, Liu H. The use of zero-valent iron for groundwater remediation and wastewater treatment: a review. J Hazard Mater. 2014;267:194–205. https://doi.org/10.1016/j.jhazmat.2013.12.062.
Daneshvar N, Salari D, Khataee AR. Photocatalytic degradation of azo dye acid red 14 in water: investigation of the effect of operational parameters. J Photochem Photobiol A Chem. 2003;157:111–6. https://doi.org/10.1016/S1010-6030(03)00015-7.
Chen D, Cheng Y, Zhou N, Chen P, Wang Y, Li K, Huo S, Cheng P, Peng P, Zhang R, Wang L, Liu H, Liu Y, Ruan R. Photocatalytic degradation of organic pollutants using TiO2-based photocatalysts: a review. J Clean Prod. 2020;268:121725. https://doi.org/10.1016/j.jclepro.2020.121725.
Torres RA, Torres W, Peringer P, Pulgarin C. Electrochemical degradation of p-substituted phenols of industrial interest on Pt electrodes: attempt of a structure–reactivity relationship assessment. Chemosphere. 2003;50:97–104. https://doi.org/10.1016/S0045-6535(02)00487-3.
You Y, Huang S, Zhao X, Li H, Cheng F, Wu J, Zhang Y, Zhou S. Hybrid microbial electrolytic/UV system for highly efficient organic pollutants removal. J Environ Sci (China). 2019;83:39–45. https://doi.org/10.1016/j.jes.2019.03.017.
Gallegos-Alcaíno A, Robles-Araya N, Avalos C, Alfonso-Alvarez A, Rodríguez CA, Valdés H, Sánchez-Flores NA, Durán-Alvarez JC, Bizarro M, Romero-Salguero FJ, Mera AC. Synthesis of BiOI/mordenite composites for photocatalytic treatment of organic pollutants present in agro-industrial wastewater. Nanomaterials. 2022;2022(12):1161. https://doi.org/10.3390/nano.
Saputera WH, Pranata JJ, Jonatan R, Widiatmoko P, Sasongko D. Photocatalytic simulation of phenol waste degradation using titanium dioxide (TiO2) P25-based photocatalysts. J Eng Technol Sci. 2023;55:419–33. https://doi.org/10.5614/j.eng.technol.sci.2023.55.4.6.
Ban F, Ye C, Yang S, Li M. Degradation of phenol by UV light-assisted electrocatalytic treatment. J Iran Chem Soc. 2023;20:2031–41. https://doi.org/10.1007/s13738-023-02821-x.
Suresh R, Rajendran S, Kumar PS, Dutta K, Vo DVN. Current advances in microbial fuel cell technology toward removal of organic contaminants—a review. Chemosphere. 2022. https://doi.org/10.1016/j.chemosphere.2021.132186.
Usman M, Monfort O, Gowrisankaran S, Hameed BH, Hanna K, Al-Abri M. Dual functional materials capable of integrating adsorption and Fenton-based oxidation processes for highly efficient removal of pharmaceutical contaminants. J Water Process Eng. 2023. https://doi.org/10.1016/j.jwpe.2023.103566.
El Shahawy A, Mohamadien RH, El-Fawal EM, Moustafa YM, Dawood MMK. Hybrid Photo-Fenton oxidation and biosorption for petroleum wastewater treatment and optimization using box-behnken design. Environ Technol Innov. 2021. https://doi.org/10.1016/j.eti.2021.101834.
Sarvothaman VP, Velisoju VK, Subburaj J, Panithasan MS, Kulkarni SR, Castaño P, Turner J, Guida P, Roberts WL, Nagarajan S. Is cavitation a truly sensible choice for intensifying photocatalytic oxidation processes?—implications on phenol degradation using ZnO photocatalysts. Ultrason Sonochem. 2023. https://doi.org/10.1016/j.ultsonch.2023.106548.
Deena S, Vedanayaki S, Sathish T, Dao MU, Rajasimman M, Saravanan R, Prakash P. Magnetic Co/CoOx@NCNT catalysts for activation of potassium peroxymonosulfate to deteriorate phenol from wastewater. Environ Res. 2023. https://doi.org/10.1016/j.envres.2022.114763.
Cavalcante IO, Neto FS, da Sousa PS, da Aires FIS, Dari DND, de Lima RKC, dos Santos JCS. Evolving sustainable energy technologies and assessments through global research networks: advancing the role of blue hydrogen for a cleaner future. RSC Sustainability. 2024. https://doi.org/10.1039/d3su00309d.
de Castro Bizerra V, Melo RLF, Neto FS, de Castro EC, Sales MB, de Sousa Junior PG, Dari DN, da Silva Aires FI, dos Santos KM, de França Serpa J, Kim TH, Fernández-Lucas J, de Souza MCM, dos Santos JCS. Exploring the synergy of ionic liquids and lipase in sustainable enzymatic engineering. J Mol Liq. 2024;399:124373. https://doi.org/10.1016/j.molliq.2024.124373.
de Castro Bizerra V, Sales MB, Melo RLF, do Nascimento JGA, Junior JB, França MP, Silva KM, dos Santos, da SilvaSousa P, da Fonseca AM, de Souza MCM, dos Santos JCS. Opportunities for cleaner leather processing based on protease enzyme: current evidence from an advanced bibliometric analysis. Renew Sustain Energy Rev. 2024;191:114162. https://doi.org/10.1016/j.rser.2023.114162.
Luo S, Zhu X, Gong M, Mo R, Yang S. Coupling the recovery of spent lithium-ion batteries and the treatment of phenol wastewater: a “treating waste with waste” strategy. Chemosphere. 2023. https://doi.org/10.1016/j.chemosphere.2023.140018.
Mumtaz F, Li B, Al Shehhia MR, Feng X, Wang K. Treatment of phenolic-wastewater by hybrid technologies: a review. J Water Proc Eng. 2024;57:104695. https://doi.org/10.1016/j.jwpe.2023.104695.
Mohamad Said KA, Ismail AF, Abdul Karim Z, Abdullah MS, Hafeez A. A review of technologies for the phenolic compounds recovery and phenol removal from wastewater. Proc Safety Environ Protect. 2021;151:257–89. https://doi.org/10.1016/j.psep.2021.05.015.
Davidson RS. The photodegradation of some naturally occurring polymers. J Photochem Photobiol B Biol. 1996. https://doi.org/10.1016/1011-1344(95)07262-4.
Zhang G, Chen W, Dou M, Su D, Zhang J, Zhao Z. Application of iron oxides modified volcanic rock in continuous phenol wastewater anaerobic treatment: performance and underlying mechanisms. Renew Energy. 2023. https://doi.org/10.1016/j.renene.2023.119214.
Kang Y, Chen Y, Zhou M, Xu Y, Feng H, Chen R. Identification of regional industrial priority pollutants in surface water: a field study in Taihu Lake Basin. Front Environ Sci. 2022. https://doi.org/10.3389/fenvs.2022.1077430.
Pan Y, Zhang Y, Hou M, Xue J, Qin R, Zhou M, Zhang Y. Properties of polyphenols and polyphenol-containing wastewaters and their treatment by Fenton/Fenton-like reactions. Sep Purif Technol. 2023. https://doi.org/10.1016/j.seppur.2023.123905.
Kundu A, Cherwoo L, Kumar B. Leveraging microorganisms for phenol and cyanide degradation in coke oven industry effluent treatment: current advances and future potential. Bioresour Technol Rep. 2023. https://doi.org/10.1016/j.biteb.2023.101586.
Mainali K. Phenolic Compounds contaminants in water: a glance. Curr Trends Civil Struct Eng. 2020. https://doi.org/10.33552/ctcse.2020.04.000593.
Otitoju O, Alfred M, Ogunlaja O, Olorunnisola C, Olukanni O, Ogunlaja A, Omorogie M, Unuabonah E. Pollution and risk assessment of phenolic compounds in drinking water sources from South-Western Nigeria. Environ Sci Pollut Res. 2022. https://doi.org/10.21203/rs.3.rs-2233618/v1.
Mahmoud AED, Franke M, Braeutigam P. Experimental and modeling of fixed-bed column study for phenolic compounds removal by graphite oxide. J Water Proc Eng. 2022. https://doi.org/10.1016/j.jwpe.2022.103085.
Li X, Liu M, Wu N, Sharma VK, Qu R. Enhanced removal of phenolic compounds by ferrate(VI): Unveiling the Bi(III)-Bi(V) valence cycle with in situ formed bismuth hydroxide as catalyst. Water Res. 2024. https://doi.org/10.1016/j.watres.2023.120827.
Ladeia Ramos R, Rezende Moreira V, Santos Amaral MC. Phenolic compounds in water: review of occurrence, risk, and retention by membrane technology. J Environ Manage. 2024. https://doi.org/10.1016/j.jenvman.2023.119772.
Grace Pavithra K, Sundar Rajan P, Arun J, Brindhadevi K, Le Hoang Q, Pugazhendhi A. A review on recent advancements in extraction, removal and recovery of phenols from phenolic wastewater: challenges and future outlook. Environ Res. 2023;237:117005. https://doi.org/10.1016/j.envres.2023.117005.
Xue Y, Hu X, Sun Q, Yang Wang H, Long Wang H, MeiHou X. Review of electrochemical degradation of phenolic compounds. Int J Miner Metall Mater. 2021;28:1413–28. https://doi.org/10.1007/s12613-020-2241-7.
Savić BG, Stanković DM, Živković SM, Ognjanović MR, Tasić GS, Mihajlović IJ, Brdarić TP. Electrochemical oxidation of a complex mixture of phenolic compounds in the base media using PbO2-GNRs anodes. Appl Surf Sci. 2020. https://doi.org/10.1016/j.apsusc.2020.147120.
Seid L, Lakhdari D, Berkani M, Belgherbi O, Chouder D, Vasseghian Y, Lakhdari N. High-efficiency electrochemical degradation of phenol in aqueous solutions using Ni-PPy and Cu-PPy composite materials. J Hazard Mater. 2022. https://doi.org/10.1016/j.jhazmat.2021.126986.
Min SJ, Kim JG, Baek K. Role of carbon fiber electrodes and carbonate electrolytes in electrochemical phenol oxidation. J Hazard Mater. 2020. https://doi.org/10.1016/j.jhazmat.2020.123083.
Mohamed A, Yousef S, Nasser WS, Osman TA, Knebel A, Sánchez EPV, Hashem T. Rapid photocatalytic degradation of phenol from water using composite nanofibers under UV. Environ Sci Eur. 2020;32:160. https://doi.org/10.1186/s12302-020-00436-0.
Asencios YJO, Lourenço VS, Carvalho WA. Removal of phenol in seawater by heterogeneous photocatalysis using activated carbon materials modified with TiO2. Catal Today. 2022;388–389:247–58. https://doi.org/10.1016/j.cattod.2020.06.064.
Zhai R, Hu J, Jin M. Towards efficient enzymatic saccharification of pretreated lignocellulose: enzyme inhibition by lignin-derived phenolics and recent trends in mitigation strategies. Biotechnol Adv. 2022;61:108044. https://doi.org/10.1016/j.biotechadv.2022.108044.
Liu S, Huang B, Zheng G, Zhang P, Li J, Yang B, Chen Y, Liang L. Nanocapsulation of horseradish peroxidase (HRP) enhances enzymatic performance in removing phenolic compounds. Int J Biol Macromol. 2020;150:814–22. https://doi.org/10.1016/j.ijbiomac.2020.02.043.
Sellami K, Couvert A, Nasrallah N, Maachi R, Tandjaoui N, Abouseoud M, Amrane A. Bio-based and cost effective method for phenolic compounds removal using cross-linked enzyme aggregates. J Hazard Mater. 2021;403:124021. https://doi.org/10.1016/j.jhazmat.2020.124021.
Mei X, Li J, Jing C, Fang C, Liu Y, Wang Y, Liu J, Bi S, Chen Y, Xiao Y, Yang X, Xiao Y, Wu S, Ding Y. Separation and recovery of phenols from an aqueous solution by a green membrane system. J Clean Prod. 2020;251:119675. https://doi.org/10.1016/j.jclepro.2019.119675.
Tian H, Xu X, Qu J, Li H, Hu Y, Huang L, He W, Li B. Biodegradation of phenolic compounds in high saline wastewater by biofilms adhering on aerated membranes. J Hazard Mater. 2020. https://doi.org/10.1016/j.jhazmat.2020.122463.
Zagklis DP, Vavouraki AI, Kornaros ME, Paraskeva CA. Purification of olive mill wastewater phenols through membrane filtration and resin adsorption/desorption. J Hazard Mater. 2015;285:69–76. https://doi.org/10.1016/j.jhazmat.2014.11.038.
Bódalo A, Gómez E, Hidalgo AM, Gómez M, Murcia MD, López I. Nanofiltration membranes to reduce phenol concentration in wastewater. Desalination. 2009;245:680–6. https://doi.org/10.1016/j.desal.2009.02.037.
Mahmoud AED, Mostafa E. Nanofiltration membranes for the removal of heavy metals from aqueous solutions: preparations and applications. Membranes (Basel). 2023. https://doi.org/10.3390/membranes13090789.
Acero JL, Benítez FJ, Leal I, Real FJ. Removal of phenolic compounds in water by ultrafiltration membrane treatments. J Environ Sci Health Part A. 2005;40:1585–603. https://doi.org/10.1081/ESE-200060651ss.
Jiang B, Zeng Q, Liu J, Hou Y, Xu J, Li H, Shi S, Ma F. Enhanced treatment performance of phenol wastewater and membrane antifouling by biochar-assisted EMBR. Bioresour Technol. 2020. https://doi.org/10.1016/j.biortech.2020.123147.
Ren L-F, Ngo HH, Bu C, Ge C, Ni S-Q, Shao J, He Y. Novel external extractive membrane bioreactor (EMBR) using electrospun polydimethylsiloxane/polymethyl methacrylate membrane for phenol-laden saline wastewater. Chem Eng J. 2020;383:123179. https://doi.org/10.1016/j.cej.2019.123179.
Xu J, Duan W, Zhou X, Zhou J. Extraction of phenol in wastewater with annular centrifugal contactors. J Hazard Mater. 2006;131:98–102. https://doi.org/10.1016/j.jhazmat.2005.09.011.
Dehmani Y, Alrashdi AA, Lgaz H, Lamhasni T, Abouarnadasse S, Chung I-M. Removal of phenol from aqueous solution by adsorption onto hematite (α-Fe2O3): Mechanism exploration from both experimental and theoretical studies. Arab J Chem. 2020;13:5474–86. https://doi.org/10.1016/j.arabjc.2020.03.026.
Farhod Chasib K. Extraction of phenolic pollutants (Phenol and p-Chlorophenol) from industrial wastewater. J Chem Eng Data. 2013;58:1549–64. https://doi.org/10.1021/je4001284.
Chen Y, Zhang X, Wang L, Cheng X, Shang Q. Rapid removal of phenol/antibiotics in water by Fe-(8-hydroxyquinoline-7-carboxylic)/TiO2 flower composite: Adsorption combined with photocatalysis. Chem Eng J. 2020;402:126260. https://doi.org/10.1016/j.cej.2020.126260.
Polaert I, Wilhelm AM, Delmas H. Phenol wastewater treatment by a two-step adsorption–oxidation process on activated carbon. Chem Eng Sci. 2002;57:1585–90. https://doi.org/10.1016/S0009-2509(02)00034-9.
Ma Y, Gao N, Chu W, Li C. Removal of phenol by powdered activated carbon adsorption. Front Environ Sci Eng. 2013;7:158–65. https://doi.org/10.1007/s11783-012-0479-7.
Roostaei N, Tezel FH. Removal of phenol from aqueous solutions by adsorption. J Environ Manage. 2004;70:157–64. https://doi.org/10.1016/j.jenvman.2003.11.004.
Romanini E, Colangelo D, Lucini L, Lambri M. Identifying chemical parameters and discriminant phenolic compounds from metabolomics to gain insight into the oxidation status of bottled white wines. Food Chem. 2019;288:78–85. https://doi.org/10.1016/j.foodchem.2019.02.073.
Rubalcaba A, Suárez-Ojeda ME, Stüber F, Fortuny A, Bengoa C, Metcalfe I, Font J, Carrera J, Fabregat A. Phenol wastewater remediation: advanced oxidation processes coupled to a biological treatment. Water Sci Technol. 2007;55:221–7. https://doi.org/10.2166/wst.2007.412.
Yao J, Liu S, Huang Y, Ren S, Lv Y, Kong M, Li G. Acyl-chloride functionalized graphene oxide chemically grafted with hindered phenol and its application in anti-degradation of polypropylene. Prog Nat Sci Mater Int. 2020;30:328–36. https://doi.org/10.1016/j.pnsc.2020.05.010.
Dwinandha D, Zhang B, Fujii M. Prediction of reaction mechanism for OH radical-mediated phenol oxidation using quantum chemical calculation. Chemosphere. 2022. https://doi.org/10.1016/j.chemosphere.2021.132763.
Mohammadi S, Kargari A, Sanaeepur H, Abbassian K, Najafi A, Mofarrah E. Phenol removal from industrial wastewaters: a short review, desalination. Water Treat. 2015;53:2215–34. https://doi.org/10.1080/19443994.2014.883327.
Pratarn W, Pornsiri T, Thanit S, Tawatchai C, Wiwut T. Adsorption and ozonation kinetic model for phenolic wastewater treatment. Chin J Chem Eng. 2011;19:76–82. https://doi.org/10.1016/S1004-9541(09)60180-4.
Kujawski W, Warszawski A, Ratajczak W, Porębski T, Capała W, Ostrowska I. Removal of phenol from wastewater by different separation techniques. Desalination. 2004;163:287–96. https://doi.org/10.1016/S0011-9164(04)90202-0.
Zhao Y. Composition of phenolic compounds in wastewater from the fixed-bed gasification of Baishihu coal. Int J Coal Sci Technol. 2021;8:1461–7. https://doi.org/10.1007/s40789-021-00425-5.
Peñaranda PA, Noguera MJ, Florez SL, Husserl J, Ornelas-Soto N, Cruz JC, Osma JF. Treatment of wastewater, phenols and dyes using novel magnetic torus microreactors and laccase immobilized on magnetite nanoparticles. Nanomaterials. 2022. https://doi.org/10.3390/nano12101688.
Mohd A. Presence of phenol in wastewater effluent and its removal: an overview. Int J Environ Anal Chem. 2022;102:1362–84. https://doi.org/10.1080/03067319.2020.1738412.
Nakhli SAA, Ahmadizadeh K, Fereshtehnejad M, Rostami MH, Safari M, Borghei SM. Biological removal of phenol from saline wastewater using a moving bed biofilm reactor containing acclimated mixed consortia. Springerplus. 2014;3:112. https://doi.org/10.1186/2193-1801-3-112.
Moussavi G, Mahmoudi M, Barikbin B. Biological removal of phenol from strong wastewaters using a novel MSBR. Water Res. 2009;43:1295–302. https://doi.org/10.1016/j.watres.2008.12.026.
Pradeep NV, Anupama S, Navya K, Shalini HN, Idris M, Hampannavar US. Biological removal of phenol from wastewaters: a mini review. Appl Water Sci. 2015;5:105–12. https://doi.org/10.1007/s13201-014-0176-8.
Baig N, Kammakakam I, Falath W, Kammakakam I. Nanomaterials: a review of synthesis methods, properties, recent progress, and challenges. Mater Adv. 2021;2:1821–71. https://doi.org/10.1039/d0ma00807a.
Taghipour S, Hosseini SM, Ataie-Ashtiani B. Engineering nanomaterials for water and wastewater treatment: review of classifications, properties and applications. New J Chem. 2019;43:7902–27. https://doi.org/10.1039/C9NJ00157C.
Baig N. Two-dimensional nanomaterials: a critical review of recent progress, properties, applications, and future directions. Compos Part A Appl Sci Manuf. 2023;165:107362. https://doi.org/10.1016/j.compositesa.2022.107362.
Rana K, Kaur H, Singh N, Sithole T, Siwal SS. Graphene-based materials: unravelling its impact in wastewater treatment for sustainable environments. Next Mater. 2024;3:100107. https://doi.org/10.1016/j.nxmate.2024.100107.
Iravani S, Varma RS. Nanosponges for water treatment: progress and challenges. Appl Sci. 2022. https://doi.org/10.3390/app12094182.
Zahmatkesh S, Bokhari A, Karimian M, Zahra MMA, Sillanpää M, Panchal H, Alrubaie AJ, Rezakhani Y. A comprehensive review of various approaches for treatment of tertiary wastewater with emerging contaminants: what do we know? Environ Monit Assess. 2022;194:884. https://doi.org/10.1007/s10661-022-10503-z.
Rathod S, Preetam S, Pandey C, Bera SP. Exploring synthesis and applications of green nanoparticles and the role of nanotechnology in wastewater treatment. Biotechnol Reports. 2024;41:e00830. https://doi.org/10.1016/j.btre.2024.e00830.
Nti EK, Cobbina SJ, Attafuah EE, Senanu LD, Amenyeku G, Gyan MA, Forson D, Safo A-R. Water pollution control and revitalization using advanced technologies: uncovering artificial intelligence options towards environmental health protection. Sustain Water Secur Heliyon. 2023;9:e18170. https://doi.org/10.1016/j.heliyon.2023.e18170.
Safeer S, Pandey RP, Rehman B, Safdar T, Ahmad I, Hasan SW, Ullah A. A review of artificial intelligence in water purification and wastewater treatment: recent advancements. J Water Proc Eng. 2022;49:102974. https://doi.org/10.1016/j.jwpe.2022.102974.
Wang Y, Cheng Y, Liu H, Guo Q, Dai C, Zhao M, Liu D. A review on applications of artificial intelligence in wastewater treatment. Sustainability. 2023. https://doi.org/10.3390/su151813557.
Zhang M, Zhang Z, Liu S, Peng Y, Chen J, Yoo Ki S. Ultrasound-assisted electrochemical treatment for phenolic wastewater. Ultrason Sonochem. 2020;65:105058. https://doi.org/10.1016/j.ultsonch.2020.105058.
Razmi R, Ramavandi B, Ardjmand M, Heydarinasab A. Efficient phenol removal from petrochemical wastewater using biochar-La/ultrasonic/persulphate system: characteristics, reusability, and kinetic study. Environ Technol. 2019;40:822–34. https://doi.org/10.1080/09593330.2017.1408694.
Cai Y, Li J, Qu G, Ren N, Zou H, Hu Y, Qiu W. Research on dynamics and mechanism of treatment on phenol simulated wastewater by the ultrasound cooperated electro-assisted micro-electrolysis. Water Environ Res. 2021;93:1243–53. https://doi.org/10.1002/wer.1533.
Vernès L, Vian M, Chemat F. Chapter 12—ultrasound and microwave as green tools for solid-liquid extraction. In: Poole CF, editor. Liquid-phase extraction. Amsterdam: Elsevier; 2020. p. 355–74. https://doi.org/10.1016/B978-0-12-816911-7.00012-8.
Dehghani MH, Karri RR, Koduru JR, Manickam S, Tyagi I, Mubarak NM. Recent trends in the applications of sonochemical reactors as an advanced oxidation process for the remediation of microbial hazards associated with water and wastewater: a critical review. Ultrason Sonochem. 2023;94:106302. https://doi.org/10.1016/j.ultsonch.2023.106302.
Son Y. Advanced oxidation processes using ultrasound technology for water and wastewater treatment, in: handbook of ultrasonics and sonochemistry. Singapore: Springer Singapore; 2016. p. 711–32.
Servant G, Laborde J-L, Hita A, Caltagirone J-P, Gérard A. Spatio-temporal dynamics of cavitation bubble clouds in a low frequency reactor: comparison between theoretical and experimental results. Ultrason Sonochem. 2001;8:163–74. https://doi.org/10.1016/S1350-4177(01)00074-8.
Antonov DV, Fedorenko RM, Strizhak PA. Micro-explosion phenomenon: conditions and benefits. Energies (Basel). 2022. https://doi.org/10.3390/en15207670.
Kyriazis N, Koukouvinis P, Gavaises M. Numerical investigations on bubble-induced jetting and shock wave focusing: application on a needle-free injection. Proc Royal Soc A Math Phys Eng Sci. 2019. https://doi.org/10.1098/rspa.2018.0548.
Luo C, Gu J, Xu X, Ma P, Zhang H, Ren X. Impact of solid particles on cavitation behaviors and laser-induced degradation in aqueous suspension. Ultrason Sonochem. 2021. https://doi.org/10.1016/j.ultsonch.2021.105632.
Shokoohi R, Rahmani A, Asgari G, Ashrafi M, Ghahramani E. The effect of the combined system of hydrodynamic cavitation, ozone, and hydrogen peroxide on chlorophyll a and organic substances removal in the raw water. Sci Rep. 2023. https://doi.org/10.1038/s41598-023-37167-0.
Kennelly TR, Eshraghi J, Dabiri S, Vlachos PP. An experimentally validated cavitation inception model for spring-driven autoinjectors. Int J Pharm. 2024;652:123753. https://doi.org/10.1016/j.ijpharm.2023.123753.
Wang N, Li L, Wang K, Huang X, Han Y, Ma X, Wang M, Lv X, Bai X. Study and application status of ultrasound in organic wastewater treatment. Sustainability (Switzerland). 2023. https://doi.org/10.3390/su152115524.
Yuan S, Li C, Zhang Y, Yu H, Xie Y, Guo Y, Yao W. Ultrasound as an emerging technology for the elimination of chemical contaminants in food: a review. Trends Food Sci Technol. 2021;109:374–85. https://doi.org/10.1016/j.tifs.2021.01.048.
Pui D, Sheng W, Bilad MR, Shamsuddin N. Assessment and optimization of coagulation process in water treatment plant: a review. ASEAN J Sci Eng. 2022. https://doi.org/10.17509/xxxx.xxxx.
Massima Mouele ES, Tijani JO, Badmus KO, Pereao O, Babajide O, Zhang C, Shao T, Sosnin E, Tarasenko V, Fatoba OO, Laatikainen K, Petrik LF. Removal of pharmaceutical residues from water and wastewater using dielectric barrier discharge methods—a review. Int J Environ Res Public Health. 2021;18:1–42. https://doi.org/10.3390/ijerph18041683.
Heidarinejad Z, Dehghani MH, Heidari M, Javedan G, Ali I, Sillanpää M. Methods for preparation and activation of activated carbon: a review. Environ Chem Lett. 2020;18:393–415. https://doi.org/10.1007/s10311-019-00955-0.
Lima L, Baêta BEL, Lima DRS, Afonso RJCF, de Aquino SF, Libânio M. Comparison between two forms of granular activated carbon for the removal of pharmaceuticals from different waters. Environ Technol. 2016;37:1334–45. https://doi.org/10.1080/09593330.2015.1114030.
Begum MHA, Rahman ASMA, Molla MR, Rahman MA. Preparation and characterization of activated carbon from paper mill sludge by chemical activation: a waste management approach. Int J Environ Sci Technol. 2023;20:2757–70. https://doi.org/10.1007/s13762-022-04169-w.
Kårelid V, Larsson G, Björlenius B. Pilot-scale removal of pharmaceuticals in municipal wastewater: Comparison of granular and powdered activated carbon treatment at three wastewater treatment plants. J Environ Manage. 2017;193:491–502. https://doi.org/10.1016/j.jenvman.2017.02.042.
Rämö R, Bonaglia S, Nybom I, Kreutzer A, Witt G, Sobek A, Gunnarsson JS. Sediment remediation using activated carbon: effects of sorbent particle size and resuspension on sequestration of metals and organic contaminants. Environ Toxicol Chem. 2022;41:1096–110. https://doi.org/10.1002/etc.5292.
Abd AA, Othman MR, Kim J. A review on application of activated carbons for carbon dioxide capture: present performance, preparation, and surface modification for further improvement. Environ Sci Pollut Res. 2021;28:43329–64. https://doi.org/10.1007/s11356-021-15121-9.
Almahbashi NMY, Kutty SRM, Jagaba AH, Al-nini A, Al-Dhawi BNS, Rathnayake U. Phenol removal from aqueous solutions using rice stalk-derived activated carbon: equilibrium, kinetics, and thermodynamics study. Case Stud Chem Environ Eng. 2023;8:100471. https://doi.org/10.1016/j.cscee.2023.100471.
Aksu Z, Tatlı Aİ, Tunç Ö. A comparative adsorption/biosorption study of Acid Blue 161: effect of temperature on equilibrium and kinetic parameters. Chem Eng J. 2008;142:23–39. https://doi.org/10.1016/j.cej.2007.11.005.
P Jin, X Jin, X Wang, Y Feng, XC., Biological activated carbon treatment process for advanced water and wastewater treatment, in: biomass now cultivation and utilization. InTech. 2013. https://doi.org/10.5772/52021.
Alves AT, Lasmar DJ, de Andrade Miranda IP, da Silva Chaar J, dos Santos Reis J. The potential of activated carbon in the treatment of water for human consumption, a study of the state of the art and its techniques used for its development. Adv Biosci Biotechnol. 2021;12:143–53. https://doi.org/10.4236/abb.2021.126010.
Belhachemi M. Chapter 14—Adsorption of organic compounds on activated carbons. In: Núñez-Delgado A, editor. Sorbents materials for controlling environmental pollution. Amsteram: Elsevier; 2021. p. 355–85. https://doi.org/10.1016/B978-0-12-820042-1.00006-7.
Mestre AS, Campinas M, Viegas RMC, Mesquita E, Carvalho AP, Rosa MJ. Chapter 17—Activated carbons in full-scale advanced wastewater treatment. In: Giannakoudakis D, Meili L, Anastopoulos I, editors. Advanced materials for sustainable environmental remediation. Amsterdam: Elsevier; 2022. p. 433–75.
Sacco NA, Zoppas FM, Devard A, del González Muñoz MP, García G, Marchesini FA. Recent advances in microplastics removal from water with special attention given to photocatalytic degradation: review of scientific research. Microplastics. 2023;2:278–303. https://doi.org/10.3390/microplastics2030023.
Mishra RK, Mentha SS, Misra Y, Dwivedi N. Emerging pollutants of severe environmental concern in water and wastewater: a comprehensive review on current developments and future research. Water-Energy Nexus. 2023;6:74–95. https://doi.org/10.1016/j.wen.2023.08.002.
Hu Y, Gong M, Xing X, Wang H, Zeng Y, Xu CC. Supercritical water gasification of biomass model compounds: a review. Renew Sustain Energy Rev. 2020. https://doi.org/10.1016/j.rser.2019.109529.
Bogdan VI, Koklin AE, Bogdan TV, Mishanin II, Kalenchuk AN, Laptinskaya TV, Kustov LM. Hydrogen generation by gasification of phenol and alcohols in supercritical water. Int J Hydrogen Energy. 2020;45:30178–87. https://doi.org/10.1016/j.ijhydene.2020.08.086.
Wei N, Xu D, Hao B, Guo S, Guo Y, Wang S. Chemical reactions of organic compounds in supercritical water gasification and oxidation. Water Res. 2021. https://doi.org/10.1016/j.watres.2020.116634.
Honarmandrad Z, Javid N, Malakootian M. Removal efficiency of phenol by ozonation process with calcium peroxide from aqueous solutions. Appl Water Sci. 2021. https://doi.org/10.1007/s13201-020-01344-7.
Lim S, Shi JL, von Gunten U, McCurry DL. Ozonation of organic compounds in water and wastewater: a critical review. Water Res. 2022. https://doi.org/10.1016/j.watres.2022.118053.
Malik SN, Ghosh PC, Vaidya AN, Mudliar SN. Hybrid ozonation process for industrial wastewater treatment: Principles and applications: a review. J Water Proc Eng. 2020. https://doi.org/10.1016/j.jwpe.2020.101193.
Zhang S, Zhou L, Li Z, Esmailpour AA, Li K, Wang S, Liu R, Li X, Yun J. Efficient treatment of phenol wastewater by catalytic ozonation over micron-sized hollow MgO rods. ACS Omega. 2021;6:25506–17. https://doi.org/10.1021/acsomega.1c03497.
Wang J, Chen H. Catalytic ozonation for water and wastewater treatment: recent advances and perspective. Sci Total Environ. 2020. https://doi.org/10.1016/j.scitotenv.2019.135249.
El-Saadony MT, Saad AM, El-Wafai NA, Abou-Aly HE, Salem HM, Soliman SM, Abd El-Mageed TA, Elrys AS, Selim S, Abd El-Hack ME, Kappachery S, El-Tarabily KA, AbuQamar SF. Hazardous wastes and management strategies of landfill leachates: a comprehensive review. Environ Technol Innov. 2023;31:103150. https://doi.org/10.1016/j.eti.2023.103150.
Tóth AJ, Fózer D, Mizsey P, Varbanov PS, Klemeš JJ. Physicochemical methods for process wastewater treatment: powerful tools for circular economy in the chemical industry. Rev Chem Eng. 2023;39:1123–51. https://doi.org/10.1515/revce-2021-0094.
Ali I, Asim M, Khan TA. Low cost adsorbents for the removal of organic pollutants from wastewater. J Environ Manage. 2012;113:170–83. https://doi.org/10.1016/j.jenvman.2012.08.028.
Rahmani A, Asgari G, Barjasteh Asgari F, Hedayati Kamran E, Alijani F. Investigation of phenol removal from aqueous solutions using copper-impregnated pumice. Umsha. 2011;17:50–6.
Nishu Kumar S. Smart and innovative nanotechnology applications for water purification, Hybrid. Advances. 2023;3:100044. https://doi.org/10.1016/j.hybadv.2023.100044.
Pérez H, García OJQ, Amezcua-Allieri MA, Vázquez RR. Nanotechnology as an efficient and effective alternative for wastewater treatment: an overview. Water Sci Technol. 2023;87:2971–3001. https://doi.org/10.2166/wst.2023.179.
Rios NS, Morais EG, dos Santos Galvão W, Andrade Neto DM, dos Santos JCS, Bohn F, Correa MA, Fechine PBA, Fernandez-Lafuente R, Gonçalves LRB. Further stabilization of lipase from Pseudomonas fluorescens immobilized on octyl coated nanoparticles via chemical modification with bifunctional agents. Int J Biol Macromol. 2019. https://doi.org/10.1016/j.ijbiomac.2019.09.003.
Melo RLF, Freire TM, Valério RBR, Neto FS, de Castro Bizerra V, Fernandes BCC, de Sousa Junior PG, da Fonseca AM, Soares JM, Fechine PBA, dos Santos JCS. Enhancing biocatalyst performance through immobilization of lipase (Eversa® Transform 2.0) on hybrid amine-epoxy core-shell magnetic nanoparticles. Int J Biol Macromol. 2024. https://doi.org/10.1016/j.ijbiomac.2024.130730.
Monteiro RRC, Dos Santos JCS, Alcántara AR, Fernandez-Lafuente R. Enzyme-coated micro-crystals: an almost forgotten but very simple and elegant immobilization strategy. Catalysts. 2020;10:1–27. https://doi.org/10.3390/catal10080891.
Monteiro RRC, Neto DMA, Fechine PBA, Lopes AAS, Gonçalves LRB, Dos Santos JCS, de Souza MCM, Fernandez-Lafuente R. Ethyl butyrate synthesis catalyzed by lipases a and b from candida antarctica immobilized onto magnetic nanoparticles. Improvement of biocatalysts’ performance under ultrasonic irradiation. Int J Mol Sci. 2019. https://doi.org/10.3390/ijms20225807.
Lima P, Silva J, Melo R, Neto F, Fechine P, Rocha M, Gonçalves L, dos Santos J. Dimensioning of vinylsulfonic supports from cashew apple bagasse biomass in the immobilization of lipases. Quim Nova. 2024. https://doi.org/10.21577/0100-4042.20230132.
Hairuddin MN, Mubarak NM, Khalid M, Abdullah EC, Walvekar R, Karri RR. Magnetic palm kernel biochar potential route for phenol removal from wastewater. Environ Sci Pollut Res. 2019;26:35183–97. https://doi.org/10.1007/s11356-019-06524-w.
Abdelwahab O, Amin NK. Adsorption of phenol from aqueous solutions by Luffa cylindrica fibers: Kinetics, isotherm and thermodynamic studies. Egypt J Aquat Res. 2013;39:215–23. https://doi.org/10.1016/j.ejar.2013.12.011.
Viet NM, Mai Huong NT, Thu Hoai PT. Enhanced photocatalytic decomposition of phenol in wastewater by using La–TiO2 nanocomposite. Chemosphere. 2023;313:137605. https://doi.org/10.1016/j.chemosphere.2022.137605.
Ren L-F, Chen R, Zhang X, Shao J, He Y. Phenol biodegradation and microbial community dynamics in extractive membrane bioreactor (EMBR) for phenol-laden saline wastewater. Bioresour Technol. 2017;244:1121–8. https://doi.org/10.1016/j.biortech.2017.08.121.
Eisenreich SJ, Looney BB, David J. Thornton, Airborne organic contaminants in the Great Lakes ecosystem. Environ Sci Technol. 1981;15:30–8. https://doi.org/10.1021/es00083a002.
Breton RL, Teed RS, Moore DRJ. An ecological risk assessment of phenol in the aquatic environment. Human Ecol Risk Assess Int J. 2003;9:549–68. https://doi.org/10.1080/713609922.
Dalela RC, Rani S, Rani S, Verma SR. Influence of pH on the toxicity of phenol and its two derivatives pentachlorophenol and dinitrophenol to some fresh water teleosts. Acta Hydrochimica et Hydrobiol. 1980;8:623–9. https://doi.org/10.1002/j.1521-401X.1980.tb00074.x.
Saarikoski J, Viluksela M. Relation between physicochemical properties of phenols and their toxicity and accumulation in fish. Ecotoxicol Environ Saf. 1982;6:501–12. https://doi.org/10.1016/0147-6513(82)90032-X.
Green DWJ, Williams KA, Hughes DRL, Shaik GAR, Pascoe D. Toxicity of phenol to asellus aquaticus (L.)—effects of temperature and episodic exposure. Water Res. 1988;22:225–31. https://doi.org/10.1016/0043-1354(88)90082-6.
Muñoz-Palazon B, Gorrasi S, Rosa-Masegosa A, Pasqualetti M, Braconcini M, Fenice M. Treatment of high-polyphenol-content waters using biotechnological approaches: the latest update. Molecules. 2023. https://doi.org/10.3390/molecules28010314.
Raza W, Lee J, Raza N, Luo Y, Kim K-H, Yang J. Removal of phenolic compounds from industrial waste water based on membrane-based technologies. J Industrial Eng Chem. 2019;71:1–18. https://doi.org/10.1016/j.jiec.2018.11.024.
Al Ajmi ASS, Bosu S, Rajamohan N. Biomass—metal oxide nano composite for the decontamination of phenol from polluted environment—parametric, kinetics and isotherm studies. Environ Res. 2024;240:117467. https://doi.org/10.1016/j.envres.2023.117467.
Dubey M, Kumar R, Srivastava SK, Verma A, Mathur A, Chawla V, Rubahn H-G, Joshi M, Mishra YK. ZnO/α-MnO2 hybrid 1D nanostructure-based sensor for point-of-care monitoring of chlorinated phenol in drinking water. Mater Today Chem. 2022;26:101098. https://doi.org/10.1016/j.mtchem.2022.101098.
Yi Z, Kun-Lin Y. Quantitative detection of phenol in wastewater using square wave voltammetry with pre-concentration. Anal Chim Acta. 2021;1178:338788. https://doi.org/10.1016/j.aca.2021.338788.
Al Hashemi W, Maraqa MA, Rao MV, Hossain MM. Characterization and removal of phenolic compounds from condensate-oil refinery wastewater. Desalinat Water Treat. 2015;54:660–71. https://doi.org/10.1080/19443994.2014.884472.
Ely C, de Lourdes Borba M, Magalhães CHL, Soares ES. Optimization of phenol removal from biorefinery effluent using horseradish peroxidase. J Environ Eng. 2017;143:4017075. https://doi.org/10.1061/(ASCE)EE.1943-7870.0001279.
Bosu S, Rajamohan N, Rajasimman M. Enhanced remediation of lead (II) and cadmium (II) ions from aqueous media using porous magnetic nanocomposites—a comprehensive review on applications and mechanism. Environ Res. 2022;213:113720. https://doi.org/10.1016/j.envres.2022.113720.
Rajamohan N, Bosu S, Rajasimman M, Varjani S. Environmental remediation of selenium using surface modified carbon nano tubes—characterization, influence of variables, equilibrium and kinetic analysis. Environ Res. 2023;216:114629. https://doi.org/10.1016/j.envres.2022.114629.
Cai J, Zhao D, Varis O. Match words with deeds: curbing water risk with the sustainable development goal 6 index. J Clean Prod. 2021;318:128509. https://doi.org/10.1016/j.jclepro.2021.128509.
Tortajada C. Contributions of recycled wastewater to clean water and sanitation sustainable development goals. NPJ Clean Water. 2020;3:22. https://doi.org/10.1038/s41545-020-0069-3.
Al-Sakkaf MK, Basfer I, Iddrisu M, Bahadi SA, Nasser MS, Abussaud B, Drmosh QA, Onaizi SA. An up-to-date review on the remediation of dyes and phenolic compounds from wastewaters using enzymes immobilized on emerging and nanostructured materials: promises and challenges. Nanomaterials. 2023. https://doi.org/10.3390/nano13152152.
Bhandari G, Chaudhary P, Gangola S, Gupta S, Gupta A, Rafatullah M, Chen S. A review on hospital wastewater treatment technologies: current management practices and future prospects. J Water Proc Eng. 2023;56:104516. https://doi.org/10.1016/j.jwpe.2023.104516.
Kalavrouziotis IK, Arambatzis C, Kalfountzos D, Varnavas SP. Wastewater reuse planning in agriculture: the case of aitoloakarnania, western greece. Water (Switzerland). 2011;3:988–1004. https://doi.org/10.3390/w3040988.
Morote ÁF, Olcina J, Hernández M. The use of non-conventional water resources as a means of adaptation to drought and climate change in semi-arid regions: South-eastern Spain. Water (Switzerland). 2019. https://doi.org/10.3390/w11010093.
Jodar-Abellan A, López-Ortiz MI, Melgarejo-Moreno J. Wastewater treatment and water reuse in Spain. Current situation and perspectives. Water (Switzerland). 2019. https://doi.org/10.3390/w11081551.
Prats-Rico D. Reuse of Purifi ed regenerated water worldwide analyzes and projections. Agua Y Territorio Water Landscape. 2016. https://doi.org/10.17561/at.v0i8.3292.
Chojnacka K, Moustakas K. Anaerobic digestate management for carbon neutrality and fertilizer use: a review of current practices and future opportunities. Biomass Bioenergy. 2024;180:106991. https://doi.org/10.1016/j.biombioe.2023.106991.
Al Hamedi FH, Kandhan K, Liu Y, Ren M, Jaleel A, Alyafei MAM. Wastewater irrigation: a promising way for future sustainable agriculture and food security in the United Arab Emirates. Water (Switzerland). 2023. https://doi.org/10.3390/w15122284.
Zhang W, Jia H, Wang Y, Gao F, Yang G, Wang J. Review in application of blast furnace dust in wastewater treatment: material preparation, integrated process, and mechanism. Environ Sci Pollut Res. 2024. https://doi.org/10.1007/s11356-024-32631-4.
Bender AF, de Souza JB, C.M. de S. Vidal,. Tecnologias avançadas de tratamento visando à remoção de cor e fenol de efluente de indústria de celulose e papel. Ciência Florestal. 2019;29:571–82. https://doi.org/10.5902/1980509832503.
Patel J, Desai H. Removal of phenol by liquid-liquid extraction from pharmaceutical wastewater. Mater Today Proc. 2022;57:2396–9. https://doi.org/10.1016/j.matpr.2021.12.435.
Liu Z, Xie W, Li D, Peng Y, Li Z, Liu S. Biodegradation of phenol by bacteria strain Acinetobacter Calcoaceticus PA isolated from phenolic wastewater. Int J Environ Res Public Health. 2016. https://doi.org/10.3390/ijerph13030300.
Afzal M, Iqbal S, Rauf S, Khalid ZM. Characteristics of phenol biodegradation in saline solutions by monocultures of Pseudomonas aeruginosa and Pseudomonas pseudomallei. J Hazard Mater. 2007;149:60–6. https://doi.org/10.1016/j.jhazmat.2007.03.046.
Wei G, Yu J, Zhu Y, Chen W, Wang L. Characterization of phenol degradation by Rhizobium sp. CCNWTB 701 isolated from Astragalus chrysopteru in mining tailing region. J Hazard Mater. 2008;151:111–7. https://doi.org/10.1016/j.jhazmat.2007.05.058.
Saraji M, Bakhshi M. Determination of phenols in water samples by single-drop microextraction followed by in-syringe derivatization and gas chromatography–mass spectrometric detection. J Chromatogr A. 2005;1098:30–6. https://doi.org/10.1016/j.chroma.2005.08.063.
Melaibari AA, Elamoudi AS, Mostafa ME, Abu-Hamdeh NH. Waste-to-Energy in Saudi Arabia: Treatment of petroleum wastewaters utilizing zeolite structures in the removal of phenol pollutants by using the power of molecular dynamics method. Eng Anal Bound Elem. 2023;148:317–23. https://doi.org/10.1016/J.ENGANABOUND.2023.01.003.
Elhamji S, Haydari I, Sbihi K, Aziz K, Elleuch J, Kurniawan TA, Chen Z, Yap PS, Aziz F. Uncovering applicability of Navicula permitis algae in removing phenolic compounds: a promising solution for olive mill wastewater treatment. J Water Proc Eng. 2023;56:104313. https://doi.org/10.1016/J.JWPE.2023.104313.
Shi J, Huang W, Wan N, Wang J. Effects of sodium acetate, glucose, and Chlorella powder as carbon source on enhanced treatment of phenolic compounds and NO2–-N in coal pyrolysis wastewater. Fuel. 2023;339:126974. https://doi.org/10.1016/J.FUEL.2022.126974.
Zeng Q, Xu J, Hou Y, Li H, Du C, Jiang B, Shi S. Effect of Fe3O4 nanoparticles exposure on the treatment efficiency of phenol wastewater and community shifts in SBR system. J Hazard Mater. 2021;407:124828. https://doi.org/10.1016/J.JHAZMAT.2020.124828.
Chaudhry A, Bashir F, Adil SF, Saif S, Shaik MR, Hatshan MR, Shaik B. Ascorbic acid-mediated Fe/Cu nanoparticles and their application for removal of COD and phenols from industrial wastewater. J King Saud Univ Sci. 2022;34:101927. https://doi.org/10.1016/J.JKSUS.2022.101927.
Abdelfattah I, Ismail AA. Reduction of COD concentration and complete removal of phenol in industrial wastewater utilizing mesoporous TiO2 nanoparticles under UVA illumination. Opt Mater (Amst). 2023;145:114410. https://doi.org/10.1016/J.OPTMAT.2023.114410.
Li Y, Kong W, Liu H, Hong Y, Huang T. Enhanced degradation of phenolic compounds in coal gasification wastewater by activated carbon-Fe3O4 nanoparticles coupled with anaerobic co-metabolism. Biochem Eng J. 2022;189:108717. https://doi.org/10.1016/J.BEJ.2022.108717.
Shah SSA, Sohail M, Murtza G, Waseem A, Rehman A, Hussain I, Bashir MS, Alarfaji SS, Hassan AM, Nazir MA, Javed MS, Najam T. Recent trends in wastewater treatment by using metal-organic frameworks (MOFs) and their composites: a critical view-point. Chemosphere. 2024;349:140729. https://doi.org/10.1016/J.CHEMOSPHERE.2023.140729.
Ambaye AD, Kebede TG, Ntsendwana B, Nxumalo EN. Fe-MOF derived graphitic carbon nitride nanocomposites as novel electrode materials for the electrochemical sensing of 2,4-dichlorophenol in wastewater. Synth Met. 2023;299:117452. https://doi.org/10.1016/J.SYNTHMET.2023.117452.
Yuan L, Chai J, Wang S, Li T, Yan X, Wang J, Yin H. Biomimetic Laccase-Cu2O@MOF for synergetic degradation and colorimetric detection of phenolic compounds in wastewater. Environ Technol Innov. 2023;30:103085. https://doi.org/10.1016/J.ETI.2023.103085.
Feng CY, Wang KH, Li S, Liu DS, Yang Z. Use of tyrosinase-inorganic salt hybrid nanoflowers and tyrosinase-MOF hybrid composites for elimination of phenolic pollutants from industrial wastewaters. Chemosphere. 2023;317:137933. https://doi.org/10.1016/J.CHEMOSPHERE.2023.137933.
Zhang J, Qin L, Yang Y, Liu X. Porous carbon nanospheres aerogel based molecularly imprinted polymer for efficient phenol adsorption and removal from wastewater. Sep Purif Technol. 2021;274:119029. https://doi.org/10.1016/J.SEPPUR.2021.119029.
Abdullah NH, Borhan A, Saadon SZAH. Biosorption of wastewater pollutants by chitosan-based porous carbons: a sustainable approach for advanced wastewater treatment. Bioresour Technol Rep. 2024;25:101705. https://doi.org/10.1016/J.BITEB.2023.101705.
Qu Y, Qin L, Yang Y, Liu X, Huang Y. ZIF-8-derived N-doped hierarchical porous carbon coated with imprinted polymer as magnetic absorbent for phenol selective removal from wastewater. J Colloid Interface Sci. 2023;630:573–85. https://doi.org/10.1016/J.JCIS.2022.10.040.
Li D, Sun T, Wang L, Wang N. Enhanced electro-catalytic generation of hydrogen peroxide and hydroxyl radical for degradation of phenol wastewater using MnO2/Nano-G|Foam-Ni/Pd composite cathode. Electrochim Acta. 2018;282:416–26. https://doi.org/10.1016/J.ELECTACTA.2018.06.075.
Liu H, Kim GE, Hong CO, Song YC, Lee WK, Liu D, Jang SH, Park YK. Treatment of phenol wastewater using nitrogen-doped magnetic mesoporous hollow carbon. Chemosphere. 2021;271:129595. https://doi.org/10.1016/J.CHEMOSPHERE.2021.129595.
Garcia-Ivars J, Iborra-Clar MI, Alcaina-Miranda MI, Mendoza-Roca JA, Pastor-Alcañiz L. Treatment of table olive processing wastewaters using novel photomodified ultrafiltration membranes as first step for recovering phenolic compounds. J Hazard Mater. 2015;290:51–9. https://doi.org/10.1016/J.JHAZMAT.2015.02.062.
Adebayo MA, Areo FI. Removal of phenol and 4-nitrophenol from wastewater using a composite prepared from clay and Cocos nucifera shell: Kinetic, equilibrium and thermodynamic studies. Resour Environ Sustain. 2021;3:100020. https://doi.org/10.1016/J.RESENV.2021.100020.
Al-Ghouti MA, Sayma J, Munira N, Mohamed D, Da’na DA, Qiblawey H, Alkhouzaam A. Effective removal of phenol from wastewater using a hybrid process of graphene oxide adsorption and UV-irradiation. Environ Technol Innov. 2022;27:102525. https://doi.org/10.1016/J.ETI.2022.102525.
Tawabini B, Zubair A. Bromate control in phenol-contaminated water treated by UV and ozone processes. Desalination. 2011;267:16–9. https://doi.org/10.1016/J.DESAL.2010.08.039.
Franco E, Camargo J, Aguilar N, Silva D, Rodrigues J, Pádua V, Libânio M, Giani A, Sierau LD. Validity of the analytical method of LLE-GC-MS for the detection of haloacetics acids in environmental samples. Engenharia Sanitaria e Ambiental. 2021;26:711–20. https://doi.org/10.1590/S1413-415220200050.
Hashim HS, Fen YW, Omar NAS, Fauzi NIM, Daniyal WMEMM. Recent advances of priority phenolic compounds detection using phenol oxidases-based electrochemical and optical sensors. Measurement. 2021;184:109855. https://doi.org/10.1016/J.MEASUREMENT.2021.109855.
Han Y, Xu H, Zhang L, Ma X, Man Y, Su Z, Wang J. An internal circulation iron–carbon micro-electrolysis reactor for aniline wastewater treatment: parameter optimization, degradation pathways and mechanism. Chin J Chem Eng. 2023;63:96–107. https://doi.org/10.1016/J.CJCHE.2023.05.009.
Fu W, Liu Z, Yang Z, Li Y, Pan B. Confined iron-based nanomaterials for water decontamination: fundamentals, applications, and challenges. Fundamental Res. 2023. https://doi.org/10.1016/J.FMRE.2023.07.011.
de Farias MB, Prediger P, Vieira MGA. Conventional and green-synthesized nanomaterials applied for the adsorption and/or degradation of phenol: a recent overview. J Clean Prod. 2022;367:132980. https://doi.org/10.1016/J.JCLEPRO.2022.132980.
Nayak T, Pathan A. Environmental remediation and application of carbon-based nanomaterials in the treatment of heavy metal-contaminated water: a review. Mater Today Proc. 2023;92:1659–70. https://doi.org/10.1016/J.MATPR.2023.06.227.
Sathish T, Ahalya N, Thirunavukkarasu M, Senthil TS, Hussain Z, Haque Siddiqui MI, Panchal H, Kumar Sadasivuni K. A comprehensive review on the novel approaches using nanomaterials for the remediation of soil and water pollution. Alexandria Eng J. 2024;86:373–85. https://doi.org/10.1016/J.AEJ.2023.10.038.
de Sousa CFB, Mota LTM, Valente RKM, Gomes WE, dos Silva VCS. IImplementation of portable biosensor using wi-fi technology for detection of phenolic compounds in urban environment. Brazilian J Dev. 2020;6:59469–80. https://doi.org/10.34117/BJDV6N8-387.
Kusworo TD, Kumoro AC, Utomo DP. Phenol and ammonia removal in petroleum refinery wastewater using a poly(vinyl) alcohol coated polysulfone nanohybrid membrane. J Water Proc Eng. 2021;39:101718. https://doi.org/10.1016/J.JWPE.2020.101718.
Mohammadi S, Kargari A, Sanaeepur H, Abbassian K, Najafi A, Mofarrah E. Phenol removal from industrial wastewaters: a short review. Desalinat Water Treat. 2015;53:2215–34. https://doi.org/10.1080/19443994.2014.883327.
Poi G, Aburto-Medina A, Mok PC, Ball AS, Shahsavari E. Bioremediation of phenol-contaminated industrial wastewater using a bacterial consortium—from laboratory to field. Water Air Soil Pollut. 2017;228:89. https://doi.org/10.1007/s11270-017-3273-0.
Fajardo AS, Rodrigues RF, Martins RC, Castro LM, Quinta-Ferreira RM. Phenolic wastewaters treatment by electrocoagulation process using Zn anode. Chem Eng J. 2015;275:331–41. https://doi.org/10.1016/j.cej.2015.03.116.
Zawadzki P. Visible light-driven advanced oxidation processes to remove emerging contaminants from water and wastewater: a review. Water Air Soil Pollut. 2022;233:374. https://doi.org/10.1007/s11270-022-05831-2.
Cuerda-Correa EM, Alexandre-Franco MF, Fernández-González C. Advanced oxidation processes for the removal of antibiotics from water. An overview. Water (Switzerland). 2020. https://doi.org/10.3390/w12010102.
Malakootian M, Nasiri A, Khatami M, Mahdizadeh H, Karimi P, Ahmadian M, Asadzadeh N, Heidari MR. Experimental data on the removal of phenol by electro-H2O2 in presence of UV with response surface methodology. MethodsX. 2019;6:1188–93. https://doi.org/10.1016/j.mex.2019.05.004.
Saputera WH, Putrie AS, Esmailpour AA, Sasongko D, Suendo V, Mukti RR. Technology advances in phenol removals: current progress and future perspectives. Catalysts. 2021. https://doi.org/10.3390/catal11080998.
Bibi A, Bibi S, Abu-Dieyeh M, Al-Ghouti MA. Towards sustainable physiochemical and biological techniques for the remediation of phenol from wastewater: a review on current applications and removal mechanisms. J Clean Prod. 2023;417:137810. https://doi.org/10.1016/j.jclepro.2023.137810.
Mosur Nagarajan A, Subramanian A, Prasad Gobinathan K, Mohanakrishna G, Sivagami K. Electrochemical-based approaches for the treatment of pharmaceuticals and personal care products in wastewater. J Environ Manage. 2023;344:118385. https://doi.org/10.1016/j.jenvman.2023.118385.
Qiao J, Xiong Y. Electrochemical oxidation technology: A review of its application in high-efficiency treatment of wastewater containing persistent organic pollutants. J Water Proc Eng. 2021;44:102308. https://doi.org/10.1016/j.jwpe.2021.102308.
Tawfik A, Alalm MG, Awad HM, Islam M, Qyyum MA, Al-Muhtaseb AH, Osman AI, Lee M. Solar photo-oxidation of recalcitrant industrial wastewater: a review. Environ Chem Lett. 2022;20:1839–62. https://doi.org/10.1007/s10311-022-01390-4.
Villegas LGC, Mashhadi N, Chen M, Mukherjee D, Taylor KE, Biswas N. A short review of techniques for phenol removal from wastewater. Curr Pollut Rep. 2016;2:157–67. https://doi.org/10.1007/s40726-016-0035-3.
Torres E, Bustos-Jaimes I, Le Borgne S. Potential use of oxidative enzymes for the detoxification of organic pollutants. Appl Catal B. 2003;46:1–15. https://doi.org/10.1016/S0926-3373(03)00228-5.
Robinson PK. Enzymes: principles and biotechnological applications. Essays Biochem. 2015;59:1–41. https://doi.org/10.1042/BSE0590001.
Yu Y, Dong H, Lian L, Guan X. Selective and rapid degradation of organic contaminants by Mn(V) generated in the Mn(II)-nitrilotriacetic acid/periodate process. Chem Eng J. 2022;443:136387. https://doi.org/10.1016/j.cej.2022.136387.
Karume I. Nature of support system and enzyme as key factors in immobilized biocatalyzed processes. Sustain Chem Environ. 2023;4:100048. https://doi.org/10.1016/j.scenv.2023.100048.
Alshabib M, Onaizi SA. A review on phenolic wastewater remediation using homogeneous and heterogeneous enzymatic processes: current status and potential challenges. Sep Purif Technol. 2019;219:186–207. https://doi.org/10.1016/j.seppur.2019.03.028.
Patidar R, Khanna S, Moholkar VS. Physical features of ultrasound assisted enzymatic degradation of recalcitrant organic pollutants. Ultrason Sonochem. 2012;19:104–18. https://doi.org/10.1016/j.ultsonch.2011.06.005.
Bansal N, Kanwar SS. Peroxidase(s) in environment protection. Sci World J. 2013. https://doi.org/10.1155/2013/714639.
Kurnik K, Treder K, Skorupa-Kłaput M, Tretyn A, Tyburski J. Removal of phenol from synthetic and industrial wastewater by potato pulp peroxidases. Water Air Soil Pollut. 2015;226:254. https://doi.org/10.1007/s11270-015-2517-0.
Garg S, Kumar P, Singh S, Yadav A, Dumée LF, Sharma RS, Mishra V. Prosopis juliflora peroxidases for phenol remediation from industrial wastewater—an innovative practice for environmental sustainability. Environ Technol Innov. 2020;19:100865. https://doi.org/10.1016/j.eti.2020.100865.
Langsdorf A, Volkmar M, Ulber R, Hollmann F, Holtmann D. Peroxidases from grass clippings for the removal of phenolic compounds from wastewater. Bioresour Technol Rep. 2023;22:101471. https://doi.org/10.1016/j.biteb.2023.101471.
Sas OG, Sánchez PB, González B, Domínguez Á. Removal of phenolic pollutants from wastewater streams using ionic liquids. Sep Purif Technol. 2020;236:116310. https://doi.org/10.1016/j.seppur.2019.116310.
Acknowledgements
The authors would like to thank the support of the Instituto de Engenharias e Desenvolvimento Sustentável (IEDS) of the Universidade da Integração Internacional da Lusofonia Afro-Brasileira (Unilab). We also thank the following Brazilian agencies for scientific and technological development: Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico (FUNCAP) (PS1-0186-00216.01.00/21; 31052.000417/2023; UNI-0210-00537.01.00/23), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (311062/2019-9; 440891/2020-5; 307454/2022-3), and Coordenação de Aperfeiçoamento de Ensino Superior (CAPES) (finance code 001).
Author information
Authors and Affiliations
Contributions
All authors have contributed equally to the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
da Silva Aires, F.I., Dari, D.N., Freitas, I.S. et al. Advanced and prospects in phenol wastewater treatment technologies: unveiling opportunities and trends. Discov Water 4, 20 (2024). https://doi.org/10.1007/s43832-024-00076-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43832-024-00076-y