Abstract
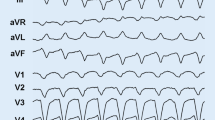
This study aimed to determine the prevalence and clinical features of Arrhythmogenic Right Ventricular Cardiomyopathy (ARVC) caused by pathogenic mutations in the Phospholamban (PLN) gene. The study included 170 patients who had a confirmed diagnosis of ARVC and underwent PLN genetic screening using next-generation sequencing. The findings of this study provide valuable insights into the association between PLN mutations and ARVC, which can aid in the development of more effective diagnostic and treatment strategies for ARVC patients. Out of the patients evaluated, six had a rare pathogenic mutation in PLN with the same p.R14del variant. Family screening revealed that heterozygous carriers of p.R14del exhibited a definite ARVC phenotype. In clinical studies, individuals with the p.R14del mutation experienced a similar rate of malignant arrhythmia events as those with classic desmosome mutations. After adjusting for covariates, individuals with PLN mutations had a two point one seven times greater likelihood of experiencing transplant-related risks compared to those who did not possess PLN mutations (95% CI 1.08–6.82, p = 0.035). The accumulation of left ventricular fat and fibers is a pathological marker for ARVC patients with p.R14del mutations. In a cohort of 170 Chinese ARVC patients, three point five percent of probands had the PLN pathogenic variant (p.R14del) and all were female. Our data shows that PLN-related ARVC patients are at high risk for ventricular arrhythmias and heart failure, which requires clinical differentiation from classic ARVC. Furthermore, carrying the p.R14del mutation can be an independent prognostic risk factor in ARVC patients.



Similar content being viewed by others
Data Availability
Data relating to this article are available in the article itself or in its Supplementary material online.
Abbreviations
- ARVC:
-
Arrhythmogenic right ventricular cardiomyopathy
- PLN :
-
Phospholamban
- DCM:
-
Dilated cardiomyopathy
- HCM:
-
Hypertrophic cardiomyopathy
- SERCA2a:
-
Sarcoplasmic/endoplasmic reticulum Ca2+-dependent ATPase 2a
- ICD:
-
Implantable cardioverter defibrillator
- RFCA:
-
Radiofrequency current catheter ablation
- NYHA:
-
The New York Heart Association
- HT:
-
Heart transplantation
- NV-HT:
-
None variants-heart transplantation
- LV:
-
Left ventricular
- RV:
-
Right ventricular
- LVEDD:
-
Left ventricle end diastolic dimension
- RVEDD:
-
Right ventricle end diastolic dimension
- LVEF:
-
Left ventricle ejection fraction
- MACE:
-
Major arrhythmic cardiovascular events
- SCD:
-
Sudden cardiac death
- P:
-
Pathogenic
- LP:
-
Likely pathogenic
- VUS:
-
Uncertain significance
- LB:
-
Likely benign
- B:
-
Benign
- ACMG:
-
American College of Medical Genetics and Genomics
- ALV:
-
Anterior free wall of the left ventricle
- LLV:
-
Lateral free wall of left ventricular
- PLV:
-
Posterior free wall of the left ventricular
- ARV:
-
Anterior free wall of right ventricular
- PRV:
-
Posterior free wall of the right ventricular
- IVS:
-
The interventricular septum
- HGVS:
-
Human genome variation society
- COX:
-
Proportional hazards
References
Bhonsale A, Te Riele A, Sawant AC et al (2017) Cardiac phenotype and long-term prognosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia patients with late presentation. Heart Rhythm 14(6):883–891. https://doi.org/10.1016/j.hrthm.2017.02.013
Calkins H, Corrado D, Marcus F (2017) Risk stratification in arrhythmogenic right ventricular cardiomyopathy. Circulation 136(21):2068–2082. https://doi.org/10.1161/CIRCULATIONAHA.117.030792
Castanos Gutierrez SL, Kamel IR, Zimmerman SL (2016) current concepts on diagnosis and prognosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia. J Thorac Imaging 31(6):324–335. https://doi.org/10.1097/RTI.0000000000000171
Chen K, Rao M, Guo G et al (2019a) Recessive variants in plakophilin-2 contributes to early-onset arrhythmogenic cardiomyopathy with severe heart failure. Europace 21(6):970–977. https://doi.org/10.1093/europace/euz026
Chen L, Rao M, Chen X et al (2019b) A founder homozygous DSG2 variant in East Asia results in ARVC with full penetrance and heart failure phenotype. Int J Cardiol 274:263–270. https://doi.org/10.1016/j.ijcard.2018.06.105
Chen L, Song J, Chen X et al (2019c) A novel genotype-based clinicopathology classification of arrhythmogenic cardiomyopathy provides novel insights into disease progression. Eur Heart J 40(21):1690–1703. https://doi.org/10.1093/eurheartj/ehz172
Choudhary N, Tompkins C, Polonsky B et al (2016) Clinical presentation and outcomes by sex in arrhythmogenic right ventricular cardiomyopathy: findings from the north american arvc registry. J Cardiovasc Electrophysiol 27(5):555–562. https://doi.org/10.1111/jce.12947
Corrado D, Basso C, Judge DP (2017a) Arrhythmogenic cardiomyopathy. Circ Res 121(7):784–802. https://doi.org/10.1161/CIRCRESAHA.117.309345
Corrado D, Link MS, Calkins H (2017b) Arrhythmogenic right ventricular cardiomyopathy. N Engl J Med 376(1):61–72. https://doi.org/10.1056/NEJMra1509267
Corrado D, van Tintelen PJ, McKenna WJ et al (2020) Arrhythmogenic right ventricular cardiomyopathy: evaluation of the current diagnostic criteria and differential diagnosis. Eur Heart J 41(14):1414–1429. https://doi.org/10.1093/eurheartj/ehz669
Engelhardt S, Hein L, Dyachenkow V et al (2004) Altered calcium handling is critically involved in the cardiotoxic effects of chronic beta-adrenergic stimulation. Circulation 109(9):1154–1160. https://doi.org/10.1161/01.CIR.0000117254.68497.39
Fu F, Tao X, Jiang Z et al (2022) Identification of germline mutations in east-asian young never-smokers with lung adenocarcinoma by whole-exome sequencing. Phenomics. https://doi.org/10.1007/s43657-022-00062-1
Gandjbakhch E, Redheuil A, Pousset F et al (2018) Clinical diagnosis, imaging, and genetics of arrhythmogenic right ventricular cardiomyopathy/dysplasia: JACC state-of-the-art review. J Am Coll Cardiol 72(7):784–804. https://doi.org/10.1016/j.jacc.2018.05.065
Haghighi K, Kolokathis F, Pater L et al (2003) Human phospholamban null results in lethal dilated cardiomyopathy revealing a critical difference between mouse and human. J Clin Invest 111(6):869–876. https://doi.org/10.1172/JCI17892
Haghighi K, Kolokathis F, Gramolini AO et al (2006) A mutation in the human phospholamban gene, deleting arginine 14, results in lethal, hereditary cardiomyopathy. Proc Natl Acad Sci USA 103(5):1388–1393. https://doi.org/10.1073/pnas.0510519103
Heinis FI, Andersson KB, Christensen G et al (2013) Prominent heart organ-level performance deficits in a genetic model of targeted severe and progressive SERCA2 deficiency. PLoS One 8(11):e79609. https://doi.org/10.1371/journal.pone.0079609
Hoorntje ET, Te Rijdt WP, James CA et al (2017) Arrhythmogenic cardiomyopathy: pathology, genetics, and concepts in pathogenesis. Cardiovasc Res 113(12):1521–1531. https://doi.org/10.1093/cvr/cvx150
Jiang Y, Li X, Guo T et al (2022) Ranolazine rescues the heart failure phenotype of PLN-deficient human pluripotent stem cell-derived cardiomyocytes. Stem Cell Reports 17(4):804–819. https://doi.org/10.1016/j.stemcr.2022.02.016
Kranias EG, Hajjar RJ (2012) Modulation of cardiac contractility by the phospholamban/SERCA2a regulatome. Circ Res 110(12):1646–1660. https://doi.org/10.1161/CIRCRESAHA.111.259754
Marcus FI, Fontaine GH, Guiraudon G et al (1982) Right ventricular dysplasia: a report of 24 adult cases. Circulation 65(2):384–398. https://doi.org/10.1161/01.cir.65.2.384
Muona M, Berkovic SF, Dibbens LM et al (2015) A recurrent de novo mutation in KCNC1 causes progressive myoclonus epilepsy. Nat Genet 47(1):39–46. https://doi.org/10.1038/ng.3144
Murray B, Hoorntje ET, Te Riele A et al (2018) Identification of sarcomeric variants in probands with a clinical diagnosis of arrhythmogenic right ventricular cardiomyopathy (ARVC). J Cardiovasc Electrophysiol 29(7):1004–1009. https://doi.org/10.1111/jce.13621
Quarta G, Muir A, Pantazis A et al (2011) Familial evaluation in arrhythmogenic right ventricular cardiomyopathy: impact of genetics and revised task force criteria. Circulation 123(23):2701–2709. https://doi.org/10.1161/CIRCULATIONAHA.110.976936
Richards S, Aziz N, Bale S et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American college of medical genetics and genomics and the association for molecular pathology. Genet Med 17(5):405–424. https://doi.org/10.1038/gim.2015.30
Saguner AM, Ganahl S, Baldinger SH et al (2014) Usefulness of electrocardiographic parameters for risk prediction in arrhythmogenic right ventricular dysplasia. Am J Cardiol 113(10):1728–1734. https://doi.org/10.1016/j.amjcard.2014.02.031
Schmitt JP, Kamisago M, Asahi M et al (2003) Dilated cardiomyopathy and heart failure caused by a mutation in phospholamban. Science 299(5611):1410–1413. https://doi.org/10.1126/science.1081578
Stadiotti I, Catto V, Casella M et al (2017) Arrhythmogenic cardiomyopathy: the guilty party in adipogenesis. J Cardiovasc Transl Res 10(5–6):446–454. https://doi.org/10.1007/s12265-017-9767-8
Towbin JA, McKenna WJ, Abrams DJ et al (2019) 2019 HRS expert consensus statement on evaluation, risk stratification, and management of arrhythmogenic cardiomyopathy. Heart Rhythm 16(11):e301–e372. https://doi.org/10.1016/j.hrthm.2019.05.007
van der Zwaag PA, van Rijsingen IA, Asimaki A et al (2012) Phospholamban R14del mutation in patients diagnosed with dilated cardiomyopathy or arrhythmogenic right ventricular cardiomyopathy: evidence supporting the concept of arrhythmogenic cardiomyopathy. Eur J Heart Fail 14(11):1199–1207. https://doi.org/10.1093/eurjhf/hfs119
van der Zwaag PA, van Rijsingen IA, de Ruiter R et al (2013) Recurrent and founder mutations in the Netherlands-Phospholamban p Arg14del mutation causes arrhythmogenic cardiomyopathy. Neth Heart J 21(6):286–293. https://doi.org/10.1007/s12471-013-0401-3
Acknowledgements
We gratefully appreciate the numerous sample donors for making this work possible.
Funding
This work was Supported by the National Natural Science Foundation for Distinguished Young Scholars of China (Grant No. 82125004), the Shenzhen Science and Technology Innovation Commission (Grant No. JCYJ20220818103414030), the National Natural Science Foundation of China (Grant No. 82300397), the key project of Shenzhen Basic Research Program (Natural Science Foundation of Shenzhen, Grant No. 20220241), and the Program for Guangdong Introducing Innovative and Enterpreneurial Teams (Grant No. 2019ZT08Y481).
Author information
Authors and Affiliations
Contributions
Conceived and designed the study: JS, XH and HM; Performed the experiments: MB, XC and ZS; Analyzed the data: HM, XH and MX; Prepared the manuscript: JS, XH and HM.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare in relation to this study.
Ethical approval
All clinical investigations were conducted in accordance with the principles expressed in the Declaration of Helsinki. The study protocol was approved by the Ethics Committee Review Board of Fuwai Hospital.
Consent to participate
All the participants provided written informed consent.
Consent to publication
The participant's consent to have their data published.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mo, H., Hua, X., Bao, M. et al. A Heterozygous Phospholamban Variant (p.R14del) Leads to Left Ventricular Involvement and Heart Failure Phenotypes in Arrhythmogenic Right Ventricular Cardiomyopathy. Phenomics 4, 13–23 (2024). https://doi.org/10.1007/s43657-023-00126-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43657-023-00126-w