Abstract
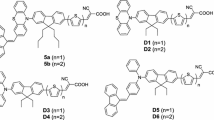
A series of phenothiazine-C60/70 dyads containing fulleropyrrolidine tethered to C-3 position (C60-PTZ and C70-PTZ) or to the heteroatom N-position via either phenyl (C60-Ph–PTZ and C70-Ph-PTZ) or phenoxyethyl linkers (C60-PhOEt-PTZ and C70-PhOEt-PTZ) of the phenothiazine were synthesized and light-induced electron transfer events were explored. Optimized studies suggested that the highest molecular orbital (HOMO) resides on donor phenothiazine moiety while lowest molecular orbital (LUMO) on the acceptor fulleropyrrolidine moiety of the dyads. Optical and electrochemical properties suggested no electronic communication between the donor and acceptor moieties in the ground state. However, steady-state emission studies in solvents of varied polarity, involving selective excitation of C60/C70, disclosed that the emission intensity of C60/C70 was quenched in the dyads in the increasing order, C60/70-PTZ > C60/70-Ph-PTZ > C60/70-PhOEt-PTZ as a consequence of the donor–acceptor distance resulted due to spacer lengths. Also, the emission quenching is more pronounced in polar solvents such as DMF compared to a non-polar solvent, toluene. With the support of parallel electrochemical studies, the emission quenching is attributed to intramolecular photo-induced electron transfer occurring from PTZ to (C60/C70)* generating a radical ion pair, PTZ+⋅–C60−⋅/PTZ+⋅–C70−⋅. Finally, bulk heterojunction (BHJ) solar cells devices inverted fashion prepared by employing the dyads as acceptors, and PTB7 as donor, suggested that the devices prepared from C70 derivatives i.e., PTB7:C70-PTZ and PTB7:C70-PhOEt-PTZ exhibited better power conversion efficiency of 2.66% and 2.15%, respectively over C60 derivatives i.e., PTB7:C60-PTZ and PTB7:C60-PhOEt-PTZ with the efficiencies of 1.80 and 1.72%, respectively. AFM studies revealed that the poor performance of PTB7:C60-PTZ- and PTB7:C60-PhOEt-PTZ-based devices can be ascribed to the lower solubility of the dyads in 1,2-DCB solvent leading to rough morphology.
Graphical abstract










Similar content being viewed by others
References
Nazeerruddin, M. K., Baranoff, E., & Grätzel, M. (2011). Dye-sensitized solar cells: A brief overview. Solar Energy, 85, 1172–1178.
Grätzel, M. (2011). Photoelectrochemical cells. Nature, 414, 338–344.
Nayak, P. K., Mahesh, S., Snaith, H. J., & Cahen, D. (2019). Photovoltaic solar cell technologies: Analyzing the state of the art. Nature Reviews Materials, 4, 269–285.
Doumon, N. Y., Yang, L., & Rosei, F. (2020). Ternary organic solar cells: A review of the role of the third element. Nano Energy, 94, 106915.
Zhao, Y., Yu, L., & Sun, M. (2021). Recent progress in emerging 2D layered materials for organic solar cells. Solar Energy, 218, 621–638.
Ma, L., Zhang, S., Wang, J., Xu, Y., & Hou, J. (2020). Recent advances in non-fullerene organic solar cells: From lab to fab. Chemical Communications, 56, 14337–14352.
Chen, L. X. (2019). Organic solar cells recent progress and challenges. ACS Energy Letters, 4, 2537–2539.
Zhang, Y., Liu, K., Huang, J., Xia, X., Cao, J., Zhao, G., Fong, P. W. K., Zhu, Y., Yan, F., Yang, Y., Lu, X., & Li, G. (2021). Graded bulk heterojuction enables 17% binary organic solar cells via nonhalogenated open air coating. Nature Communications, 12, 4815.
Wibowo, F. T. A., Krishna, N. V., Sinaga, S., Lee, S. J., Hadmojo, W. T., Do, Y. R., & Jang, S.-Y. (2021). High efficiency organic solar cells prepared using a halogen-free solution process. Cell Reports Physical Science, 2, 100517.
Zhan, L., Li, S., Lau, T., Cui, Y., Lu, X., Shi, M., Li, C.-Z., Li, H., Hou, J., & Chen, H. (2020). Over 17% efficiency ternary organic solar cells enabled by two non-fullerene acceptors working in an alloy-like model. Energy & Environmental Science, 13, 635–645.
Cui, Y., Yao, H., Zhang, J., Xian, K., Zhang, T., Hong, L., Wang, Y., Xu, Y., Ma, K., An, C., He, Ch., Wei, Z., Gao, F., & Hou, J. (2020). Single junction organic photovoltaic cells with approaching 18% efficiency. Advanced Materials, 31, 1908205.
Bagui, A., Garg, A., Tyagi, B., Gupta, V., & Singh, S. P. (2018). A fluorine-core-based for fullerene-free BHJ organic solar cells-towards power conversion efficiency over 10%. Chemical Communications, 54, 4001–4004.
Singh, S. P., Kumar, C. P., Nagarjuna, P., Kandhadi, J., Giribabu, L., Chadrasekharam, M., Biswas, S., & Sharma, G. (2016). Efficient solution processable polymer solar cells using newly designed and synthesized fullerene derivatives. Journal of Physical Chemistry C, 120, 19493–19503.
Mikroyannidis, J. A., Kabanakis, A. N., Sharma, S., & Sharma, G. D. (2011). A simple and effective modification of PCBM for use an electron acceptor in efficient bulk heterojunction solar cells. Advanced Functional Materials, 21, 746–755.
Yu, J., Zheng, Y., & Huang, J. (2014). Towards high performance organic photovoltaic cells: A review of recent development in organic solar cells. Polymers, 6, 2473–2509.
Wang, T., Pearson, A. J., & Lidzey, D. G. (2013). Correlating molecular morphology with optoelectronic function in solar cells based on low band-gap copolymer:fullerene blends. Journal of Materials Chemistry C, 1, 7266–7293.
Uchida, S., Xue, J., Rand, B. P., & Forrest, S. R. (2004). Organic small molecular solar cells with a homogenously mixed copper phthalocyanine: C60 active layer. Applied Physics Letters, 84, 4218.
Ganesamoorthy, R., Sathiya, G., & Sakthivel, P. (2017). Review: Fullerene based acceptors for efficient bulkheterojunction organic solar cell applications. Solar Energy Materials & Solar Cells, 161, 102–148.
Liu, Y., Zhao, J., Li, Z., Mu, C., Ma, W., Hu, H., Jiang, K., Lin, H., Ade, H., & Yan, H. (2014). Aggregation and morphology control enables multiple cases of high-efficiency polymer solar cells. Nature Communications, 5, 5293.
Karakawa, M., Nagai, T., Adachi, K., Le, Y., & Aso, Y. (2014). N-phenyl[C60]fulleropyrrolidine: Alternative acceptor materials to PC61BM for high performance organic photovoltaic cells. Journal of Materials Chemistry A, 2, 20889–20895.
Nagarjuna, P., Bagui, A., Gupta, V., & Singh, S. P. (2017). A highly efficient PTB7-Th polymer donor bulk hetero-junction solar cell with increased open circuit voltage using fullerene acceptor CN-PC70BM. Organic Electronics, 43, 262–267.
Blanco, G. D., Hiltunen, A. J., Lim, G. N., Kc, C. B., Kaunisto, K. M., Vuorinen, T. K., Nesterov, V. N., Lemmetyinen, H. J., & D’Souza, F. (2016). Synthesis, charge separation, and inverted bulk heterojunction solar cell application of phenothiazine-fullerene dyads. ACS Applied Materials & Interfaces, 8, 8481–8490.
Brabec, C. J., Gowrisanker, S., Halls, J. J., Laird, D., Jia, S., & Williams, S. P. (2010). Polymer–fullerene bulk-heterojunction solar cells. Advanced Materials, 22, 3839–3856.
Duvva, N., Eom, Y. K., Reddy, G., Schazne, K. K., & Giribabu, L. (2020). Bulk phenanthroimidazole-phenothiazine D-π-A based organic sensitizers for application in efficient dye-sensitized solar cells. ACS Applied Energy Materials, 3, 6758–6767.
Mi, D., Kim, J.-H., Xu, F., Lee, S.-H., Yoon, S. C., Shin, W. S., Moon, S.-J., Lee, C., & Hwang, D.-H. (2011). Synthesis and characterization of a novel fullerene derivative for use in organic solar cells. Solar Energy Materials & Solar Cells, 95, 1182–1187.
Mi, D., Kim, H.-U., Kim, J.-H., Xu, F., Jin, S.-H., & Hwang, D.-H. (2012). Synthesis of soluble fulleropyrrolidine derivative for use as an electron acceptor in bulk-heterojunction polymer solar cells. Synthetic Metals, 162, 483–489.
Maggini, M., Scorrano, G., & Prato, M. (1993). Addition of azomethineylides to C60: Synthesis, characterization, and functionalization of fullerene pyrrolidines. Journal of American Chemical Society, 115, 9798–9799.
Jain, K., Duvva, N., Roy, T. K., Giribabu, L., & Chitta, R. (2021). Porphyrin bearing phenothiazine pincers as host for fullerene binding via concave-convex complementarity: Synthesis and complexation study. New Journal of Chemistry, 45, 19691–19703.
Yanai, T., Tew, D., & Handy, N. (2004). A new hybrid exchange-correlation functional using the coulomb-attenuating method (CAM-B3LYP). Chemical Physics Letters, 393, 51–57.
Frisch, M. J., Trucks, G. W., Schlegel, H. B., Scuseria, G. E., Robb, M. A., Cheeseman, J. R., et al. (2016). Gaussian 16, Revision C.01. Gaussian, Inc., Wallingford, CT.
Langa, F., de la Cruz, P., de la Hoz, A., Espíldora, E., Cossio, F. P., & Lecea, B. (2000). Modification of regioselctivity in cycloaddition to C70 under microwave irradiation. Journal of Organic Chemistry, 65, 2499–2507.
Duvva, N., Sudhakar, K., Badgurjar, D., Chitta, R., & Giribabu, L. (2015). Space controlled photoinduced intramolecular electron transfer in a series of phenothiazine-boron dipyrromethane donor-acceptor dyads. Journal of Photochemistry and Photobiology A Chemistry, 312, 8–19.
Acknowledgements
Author RC is grateful to Department of Science and Technology (DST, CRG/2018/002661), and also DST-FIST grants, SR/FST/CSi-257/2014(C) and SR/FST/CSII/2018/65 awarded to Department of Chemistry, Central University of Rajasthan and Department of Chemistry, National Institute of Technology Warangal for the financial support of this work. The authors LG is grateful to Department of Science and Technology (DST, SB/S1/IC-14/2014), for the financial support. The authors RP is grateful to Department of Science and Technology (DST, ECR/2018/002346, EEQ/2019/000656). Author DB acknowledges Council of Scientific and Industrial Research (CSIR) for Senior Research Fellowship and Pooja acknowledges NITW for the Research Fellowship. We thank the Director CSIR-IICT for support (IICT/Pubs./2022/142).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Badgurjar, D., Duvva, N., Bagui, A. et al. Phenothiazine functionalized fulleropyrrolidines: synthesis, charge transport and applications to organic solar cells. Photochem Photobiol Sci 22, 379–393 (2023). https://doi.org/10.1007/s43630-022-00322-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43630-022-00322-z